Journal of Neurology and Therapeutics
An International Peer-Reviewed Open Access Journal
ISSN 2397-1304


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Neurology and Therapeutics
Volume 2, Issue 2, March 2016, Pages 4–7
Case reportOpen Access
Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids: Case report and review of the literature
- 1 Department of Neurology, University of Texas Medical Branch, Galveston, Texas, USA
- 2 Department of Pathology, University of Texas Southwestern Medical Center, Dallas, Texas, USA
*Corresponding author: Xiang Fang, M.D., Ph.D, Department of Neurology, University of Texas Medical Branch, Galveston, Texas 77555, USA. Tel.: 409-772-2646; Fax: 409-772-6940; E-mail: sxfang@utmb.edu
Received 7 December 2015 Revised 8 February 2016 Accepted 18 February 2016 Published 27 February 2016
DOI: http://dx.doi.org/10.14312/2397-1304.2016-2
Copyright: © 2016 Fang X, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS) is a brainstem-predominant encephalomyelitis first described by Pittock and colleagues in 2010. CLIPPERS presents with cranial nerve symptoms as well as some cognitive and spinal features. CLIPPERS is a diagnosis of exclusion. Characteristic radiographic and histopathologic findings along with clinical response to steroid suggest the diagnosis. Awareness of the features of CLIPPERS may aid in its prompt recognition, early initiation of treatment, and reduction in its associated morbidity and mortality. We present a case of CLIPPERS in a 48-year-old male patient with initial response to steroid therapy, subsequent relapse, and eventual recovery after therapy augmented with disease-modifying agents.
Keywords: Lymphocytic inflammation; brain stem; steroids; disease modifying therapy
IntroductionTop
Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS) is a newly identified clinical entity, first described in a report of 8 cases by Pittock et al. in 2010. Reported cases of this rare CNS disorder share similar clinical, radiographic, and pathologic findings: Clinical findings include diplopia, dysarthria, nystagmus, facial paresthesia, and cerebellar ataxia, with features of cognitive, spinal, and brainstem pathology in some patients. Laboratory findings include CD3+/CD4+ T-cell predominant lymphocytic infiltration. Radiographic findings include pontine and cerebellar perivascular lesions with a punctate pattern of enhancement. Patients with CLIPPERS show marked response to Glucocorticosteroids (GCS) and immunosuppressive therapy [1-5].
Here we report a case of a 48-year-old male patient who presented to our hospital with clinical, radiologic, and histologic features responsive to GCS and disease-modifying therapy (DMT). In light of our case, we briefly discuss the diagnostic features, pathogenesis, treatment, and challenges facing CLIPPERS.
Case reportTop
A 48-year-old Caucasian male with a past medical history of hypertension, hyperlipidemia, hypothyroidism, and type 2 diabetes mellitus presented with a two-month history of worsening diplopia, dysarthria, facial tingling, and gait ataxia. Neurological examination revealed limited upgaze extraocular movements, lateral gaze nystagmus, left-sided facial droop, right-handed intention tremor, left plantar up-going toe, and abnormal cerebellar signs that included bilateral finger-to-nose and heel-to-shin dysmetria. Of note, the patient was treated two years prior for previous symptoms at an outside facility, at which time he underwent extensive workup. Brain MRI revealed FLAIR hyper-intensity involving the bilateral posterior pons, middle cerebellar peduncles and right posterior lateral medulla. Our initial differential diagnoses included primary CNS tumors, metastatic disease, demyelinating disease, infection, and autoimmune encephalitis. Biopsy revealed polyclonal lymphocytic infiltration with macrophages and plasma cells. His symptoms responded well to long-term oral dexamethasone (initially 4 mg, every 6 hours, then 2 mg, every 6 hours) therapy, but clinical relapse occurred during the two months after discontinuation of therapy by his primary care physician, and which preceded his readmission.
The patient underwent an extensive laboratory workup soon after admission. His serum inflammatory markers (ACE, RPR, ESR and RF) were unremarkable. Additional tests for anti-Ro (SSA), anti-La (SSB), anti-nuclear, anti-NMO autoantibodies, and paraneoplastic panel were also unremarkable. Cerebrospinal fluid glucose and protein measured 108 mg/ml and 57 mg/ml, respectively. CSF WBC count was 29/mm3. Further workup for infectious etiologies including HTLV I/II, HHV-6, JC Virus, Parvovirus B19, HIV I/II, EBV PCR, HSV PCR, VZV PCR, Coccidioides, Lyme, and Histoplasma were negative. In addition, the CSF IgG index was not indicative of intrathecal IgG synthesis, and no oligoclonal bands were identified. CT Angiogram of the head and neck, MRI of the cervical, thoracic, and lumbar spine, and CT of the thorax, abdomen, and pelvis were similarly unremarkable.
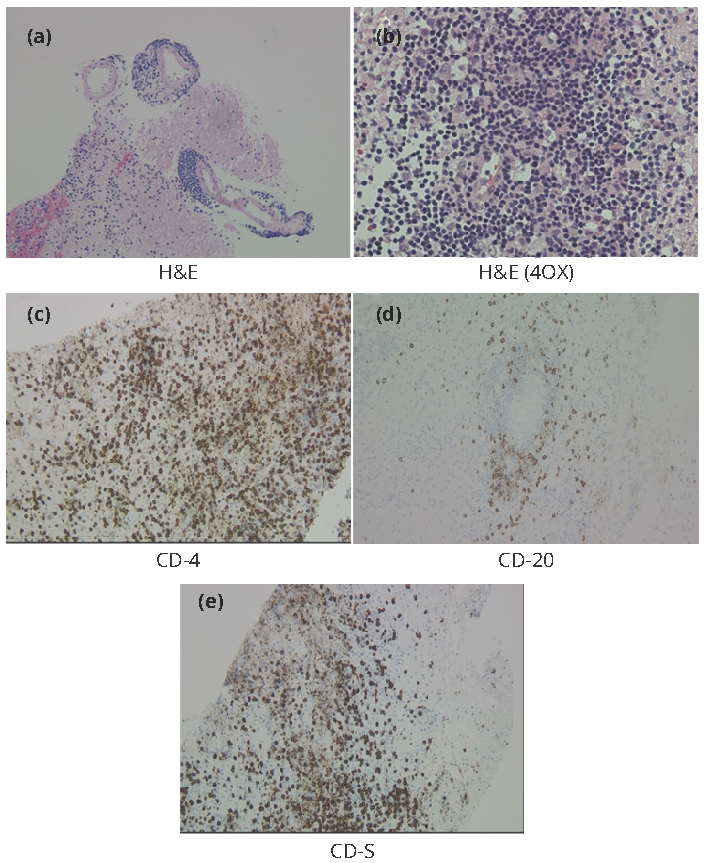
MRI of the brain was performed on admission with and without contrast (Figure 1, panels A&B). MRI findings included a patchy area of T2/FLAIR hyper-intensity seen in the posterior pons, superior medulla, middle cerebellar peduncles, and cerebellar hemispheres. These areas of abnormal T2 signal intensities demonstrated homogeneous gadolinium enhancement and appeared to show increased involvement in the cerebellar hemispheres, though no focus of restricted diffusion was noted. C-spinal MRI was unremarkable. CT-guided stereotactic biopsy of the left middle cerebellar peduncle was performed. Histologic sections of fragments of the left middle cerebellar peduncle (Figure 2, panels A&B) showed collections of chronic inflammatory cells, predominantly B and T lymphocytes, with relatively smaller numbers of plasma cells and macrophages. The inflammatory cells surrounded vessels and, in places, infiltrated the nearby parenchyma. Immunohistochemical stains demonstrated expression of CD3, CD4, CD5, CD8, CD20, CD43, bcl-2, and bcl-6 in the lymphocytes. The lymphocytes did not express CD10. In situ hybridization demonstrated expression of kappa and lambda immunoglobulin light chains in the plasma cells (Figure 2, panels C to E).
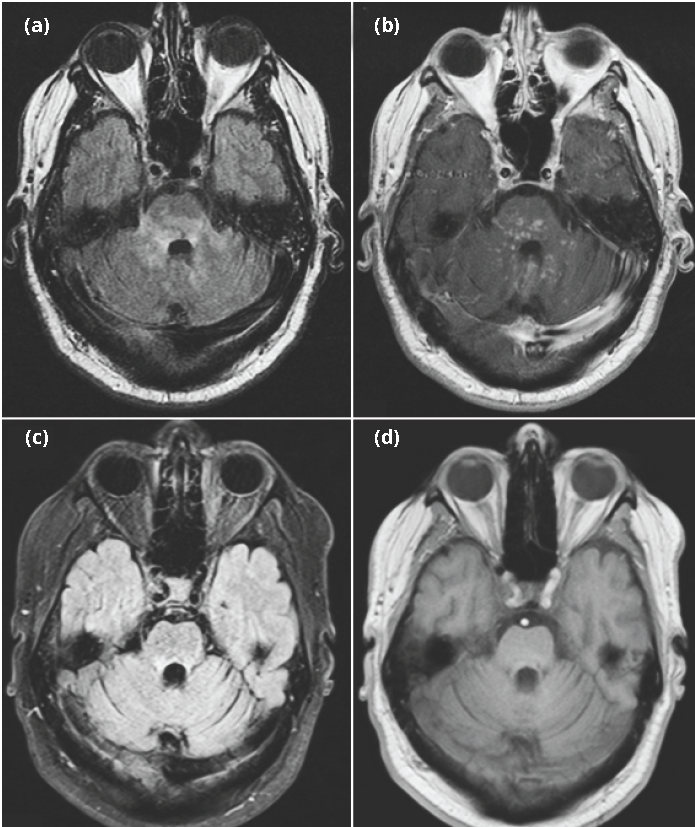
A repeat MRI of the brain (Figure 1, panels C&D) on day 3 of a 5-day course of treatment with a daily dose of 1000 mg IV methylprednisolone showed response to treatment. This finding coincided with gradual clinical improvement in dysmetria and lateral gaze nystagmus. The patient’s symptoms of dysarthria and diplopia subsequently resolved and he was able to ambulate with support. He was diagnosed with CLIPPERS and discharged on a daily 30 mg maintenance dose of oral prednisone. At clinical follow-up four months later, the patient reported a slightly worsening gait ataxia. However, repeated brain MRI revealed improvement of T2/Flair hyper-intensity in the brainstem and cerebellum as compared to the previous imaging at the time of admission (data not shown). The patient was subsequently treated with a daily 30 mg dose of prednisone and a twice-daily 50 mg dose of azathioprine. His brain MRI was stable at follow-up six months after maintenance therapy with prednisone and azathioprine.
DiscussionTop
CLIPPERS is a newly identified disease entity
CLIPPERS syndrome is a newly-identified chronic encephalomyelitis with marked responsiveness to steroid therapy [1-6]. It is characterized by episodic, progressive cerebellar ataxia with cranial nerve symptoms such as diplopia, dysarthria, and facial paresthesia [6]. Clinical findings correlate with radiographic findings of bilateral, multifocal gadolinium-enhancing lesions with punctate or curvilinear patterns on MRI, and histological findings of white matter CD4+ predominant perivascular inflammation. Although CLIPPERS was initially described as having a predilection for the hindbrain [2], specifically the pons and cerebellum, recent reports have identified lesions throughout the hindbrain and extending to the spinal cord, thalamus, internal capsule, basal ganglia, and cerebrum [7, 8]. An isolated case of suspected CLIPPERS demonstrated lesions primarily in the spinal cord [7]. Despite its evolving phenotype, CLIPPERS is a diagnosis of exclusion and should only be presumed after thoughtful exclusion of alternative diagnoses [2, 9]. In our case, the spinal cord was completely spared. Our patient was diagnosed with CLIPPERS after ruling out central infectious diseases, vasculitis, demyelination disease (MS, NMO) and CNS tumors (metastasis vs. primary), as well as corroborating clinical and radiographic evidence of marked response to steroids. Early recognition of CLIPPERS’ phenotypic spectrum followed by prompt initiation of appropriate steroids and immunotherapy are important steps in both reducing mortality and morbidity associated with CLIPPERS and avoiding unnecessary brain biopsy.
Neuroimage diagnosis
Clinical suspicion of CLIPPERS is supported by characteristic neuroimaging, namely brain and spinal cord MRI imaging. Numerous punctate (e.g., “peppered” or “speckled”) lesions of the pons and adjacent cerebellum with “curvilinear” patterns are indicative of pontine perivascular enhancement [10, 11]. As these lesions may extend rostrally to the midbrain or caudally to the medulla and cervical cord, some authors suggest that “pontocerebellar” perivascular enhancement replace “pontine” perivascular enhancement [12]. Moreover, similar lesions have also been found throughout the basal ganglia, corpus callosum, internal capsule, thalamus, and white matter [2]. An important correlate of this distribution is that the density of punctate lesions decreases with increasing distance from the pons. Additionally, lesions may coalesce to form larger lesions without any appreciable mass effect [2]. Consistent with our patient’s radiographic findings, CLIPPERS’ lesions are noteworthy for their normal cerebral angiography, marked hyper-intensity on post-T2 weighted imaging, and lack of restricted diffusion on diffusion-weighted images. Decreased gadolinium-enhancement in response to immunosuppressive therapy may aid clinicians in long-term monitoring.
Laboratory and histopathological characteristics
In patients with CLIPPERS, there are perivascular lymphohistocytic infiltrates that sometimes extend into the nearby surrounding parenchyma. The lymphocytes are mature and polyclonal. By immunohistochemistry, they usually express CD3, CD4 and CD20 [2, 11]. Importantly, the infiltrates are not granulomatous, there is no necrosis of the vessel walls and there is no surrounding demyelination [2]. CSF protein and serum IgE levels may be elevated [4], and may have mild lymphocytosis dominated by T cells. Additionally, CSF oligoclonal bands have also been reported in a patient with CLIPPERS [13]. Unlike typical CSF findings, WBC was elevated in this case. Therefore, it was critical to rule out any infectious etiologies before confirming the diagnosis of CLIPPERS.
Pathogenesis
Although the pathogenic mechanism of CLIPPERS is still unknown, neuropathological findings together with the characteristic response to GCS suggest an autoimmune or other inflammatory-mediated process; gadolinium-enhancement may be suggestive of a vascular process. We do not know why these lesions show predominantly CD4+/CD3+ expression, or why they preferentially target the pontine and peripontine structures. We observed expression of CD5, CD8, CD20, CD43, bcl-2, and bcl-6 in the lymphocytes of our patient. Nonetheless, the hindbrain is implicated as a target of immunologic attack in other CNS inflammatory disorders. Moreover, CLIPPERS’ predilection for pontine and hindbrain perivascular structures may be explained by an immune-mediated process specifically against a yet unknown auto-antigenic epitope found in those structures. The predominance of CD4+ expression may also suggest a response specific to major histocompatibility class II-restricted (MHC-II) presentation. Elevations of serum IgE and CSF protein levels suggest CLIPPERS may be a response to exogenous pathogens versus intracellular pathogens [2]. Furthermore, Taieb et al. has proposed that CLIPPERS may be a pre-lymphoma state [14].
Treatment
Responsiveness to treatment is perhaps the most promising feature of CLIPPERS. Clinical evidence to date suggests that CLIPPERS is extremely responsive to steroids, specifically intravenous steroids followed by GCS immunotherapy. Therapy includes initially high doses of steroids, followed by a gradual taper to the smallest maintenance dose necessary to prevent relapse. Our patient’s treatment included an initial five-day course of high-dose intravenous steroids (e.g., methylprednisolone 1g) followed by long-term daily 30 mg dose of oral prednisone. It is suggested that a daily 20 mg maintenance dose of oral steroids for at least 2 years could effectively prevent relapses. To be sure, early initiation of appropriate immunotherapy does not always result in complete resolution without relapse. As our case further suggests, immunomodulators and immunosupressants (e.g., azathioprine, azathioprine, methotrexate, cyclophosphamide, rituximab, or intravenous immunoglobulin) administered with GCSs may prove more beneficial than steroids alone [2]. Although there is insufficient evidence of the efficacy of these agents as monotherapy, concurrent use may lessen the dose of immunosuppressant needed, resulting in fewer side effects of therapy. This was particularly desirable in the present case, as our patient developed diabetes mellitus in the setting of long-term GCS administration. Moreover, GCS taper or cessation may provoke relapse. Thus, long-term steroid therapy and GCS-sparing immunosuppressants are the mainstays of treatment. Radiologic evidence is important not only in the early stages of cases of suspicion for CLIPPERS, but also during stages of recovery, as clinical relapse correlates with the reappearance of gadolinium-enhancing lesions.
Challenges
The diagnosis of CLIPPERS is still based primarily on clinical suspicion, guided by clinical and radiographic correlation. Patients with steroid-responsive chronic encephalomyelitis suggestive of CLIPPERS experience an average six-month delay in treatment. Moreover, no specific inflammatory biomarker or lesion is yet attributed to the disease, supporting the need for large-scale systematic studies of the entity to identify its full spectrum of clinical and pathologic manifestations. Patients presenting with cranial nerve and cerebellar symptoms should raise suspicion for CLIPPERS. CLIPPERS exists among a large number of differential diagnoses including: neurosarcoidosis, CNS lymphoma, lymphomatoid granulomatosis, CNS vasculitis, Bickerstaff brainstem encephalitis, paraneoplastic disease, chronic perivascular infectious process, glioma, CNS demyelinating disease, and CNS Behcet’s disease, Langerhans cell histiocytosis, Sjögren’s syndrome with CNS involvement, multiple sclerosis, and acute demyelinating encephalomyelitis. Furthermore, a central nervous system B-cell lymphoma was reported in a patient two years after initial diagnosis of CLIPPERS [15] and another patient was diagnosed with CLIPPERS after treatment for Hodgkin's lymphoma [16]. Exclusion of these conditions together with the lack of pathognomonic findings poses perhaps the greatest diagnostic challenge to CLIPPERS. Therefore, clinical monitoring and neuroimage surveillance are also warranted in patients with CLIPPERS.
ConclusionTop
CLIPPERS is a newly identified disease entity, and it should be considered in the differential diagnosis when patients present with clinical symptoms associated with brainstem, cerebellum, spinal cord lesions. CLIPPERS has characteristic neuroimaging findings, and workup for infectious diseases, tumor, autoimmune and demyelinating encephalomyelitis in those patients is warranted. A long-term maintenance dose of steroid therapy for the treatment of CLIPPPERS is recommended. Though disease-modifying immunotherapy may be beneficial, its long-term efficacy warrants further investigation.
Conflicts of interest
The authors declare no conflicts of interest.
ReferencesTop
[1]Pittock SJ, Debruyne J, Krecke KN, Giannini C, van den Ameele J, et al. Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS). Brain. 2010; 133(9):2626–2634.Article Pubmed
[2]Dudesek A, Rimmele F, Tesar S, Kolbaske S, Rommer PS, et al. CLIPPERS: chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids. Review of an increasingly recognized entity within the spectrum of inflammatory central nervous system disorders. Clin Exper Immunol. 2014; 175(3):385–396.Article Pubmed
[3]Gabilondo, I. Saiz A, Graus F, Villoslada P. Response to immunotherapy in CLIPPERS syndrome. J Neurol. 2011; 258(11):2090–2092.Article Pubmed
[4]Biotti D, Deschamps R, Shotar E, Maillart E, Obadia M, et al. CLIPPERS: chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids. Pract Neurol. 2011; 11(6):349–351.Article Pubmed
[5]Tohge R, Nagao M, Yagishita A, Matsubara S. A case of chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS) in East Asia. Intern Med. 2012; 51(9):1115–1119.Article Pubmed
[6]Buttmann M, Metz I, Brecht I, Brück W, Warmuth-Metz M. A typical chronic lymphocytic inflammation with pontocerebellar perivascular enhancement responsive to steroids (CLIPPERS), primary angiitis of the CNS mimicking CLIPPERS or overlap syndrome? A case report. J Neurol Sci. 2013; 324(1-2):183–186.Article Pubmed
[7]Song B, Gao Y, Fang H, Li P, Li Y, et al. Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids with lesions distributed predominantly in spinal cord. Brain. 2015; 138(Pt 12):e397.Article Pubmed
[8]Sempere AP, Mola S, Martin-Medina P, Bernabeu A, Khabbaz E, et al. Response to Immunotherapy in CLIPPERS: Clinical, MRI, and MRS Follow-Up. J Neuroimaging. 2013; 23(2):254–255.Article Pubmed
[9]Tan BL, Agzarian M, Schultz DW. CLIPPERS: Induction and maintenance of remission using hydroxychloroquine. Neurol Neuroimmunol Neuroinflamm. 2014; 2(1):e56.Article Pubmed
[10]Taieb G, Duflos C, Renard D, Audoin B, Kaphan E, et al. Long-term outcomes of CLIPPERS (chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids) in a consecutive series of 12 patients. Arch Neurol. 2012; 69(7):847–855.Article Pubmed
[11]Kastrup O, van de Nes J, Gasser T, Keyvani K. Three cases of CLIPPERS: a serial clinical, laboratory and MRI follow-up study. J Neurol. 2011; 258(12):2140–2146.Article Pubmed
[12]Simon NG, Parratt JD, Barnett MH, Buckland ME, Gupta R, et al. Expanding the clinical, radiological and neuropathological phenotype of chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS). J Neurol Neurosurg and Psychiatry. 2012; 83(1):15–22.Article Pubmed
[13]Kerrn-Jespersen BM, Lindelof M, Illes Z, Blaabjerg M, Lund EL, et al. CLIPPERS among patients diagnosed with non-specific CNS neuroinflammatory diseases. J Neurol Sci. 2014; 343(1-2):224–227.Article Pubmed
[14]Taieb G, Uro-Coste E, Clanet M, Lassmann H, Benouaich-Amiel A, et al. A central nervous system B-cell lymphoma arising two years after initial diagnosis of CLIPPERS. J Neurol Sci. 2014; 344(1–2):224–226.Article Pubmed
[15]Taieb G, Renard D, Labauge P. Should CLIPPERS be considered a prelymphoma state or a new inflammatory disease? JAMA Neurol. 2013; 70(9):1200–1201.Article Pubmed
[16]Mashima K, Suzuki S, Mori T, Shimizu T, Yamada S, et al., Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids (CLIPPERS) after treatment for Hodgkin's lymphoma. Int J Hematol. 2015; 102(6):709–712.Article Pubmed



