Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 6, June 2014, Pages 84–88
Original researchOpen Access
Histopathological investigation of differences between female and male rats on radiation-induced acute lung toxicity
- 1 Katip Celebi University, Ataturk Training and Research Hospital, Department of Radiation Oncology, Izmir, Turkey
- 2 Trakya University Department of Radiation Oncology, Edirne, Turkey
- 3 Trakya University Department of Pathology, Edirne, Turkey
*Corresponding author: Dr. Mihriban Kocak, Katip Celebi University, Ataturk Training and Research Hospital, Department of Radiation Oncology, Basin Sitesi, Karabaglar, Izmir, Turkey. Fax: 90232-243-44-43; Email: mihribankocak@hotmail.com
Received 31 March 2014 Revised 12 May 2014 Accepted 20 May 2014 Published 28 May 2014
Citation: Bilal BU, Kocak Z, Cosar R, Altaner S, Kocak M (2014) Histopathological investigation of differences between female and male rats on radiation-induced acute lung toxicity. J Cancer Res Ther 2: 84-88.
DOI: http://dx.doi.org/10.14312/2052-4994.2014-12
Copyright: © 2014 Bilal BU, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Radiation-induced acute and chronic lung toxicity is one of the important adverse events that has a negative influence on quality of life. It is widely accepted today that there are important factors such as personal sensibility and genetic differences between patients which effect both treatment response and treatment toxicity. In this study, the influence of gender differences on acute lung toxicity due to radiotherapy is investigated. Accordingly, it is examined if there is a relation between gender and histopathologically proven vasculitis and pneumonia in the lung tissues of male and female rats that are sacrificed 6 weeks after radiotherapy. It is concluded that the gender differences in rats have no effect on lung toxicity.
Keywords: radiotherapy; radiation-induced lung toxicity; lung radiotherapy; radiation pneumonitis
IntroductionTop
Radiotherapy (RT) plays an important role in the treatment of many tumors which exist in and around thorax. Radiation pneumonia is the main dose-limiting toxicity in thoracic radiotherapy. Radiation pneumonia usually develops within 4–12 weeks after RT and it can cause hypoxia, cough, dyspnea, fever and exhaustion, and can be fatal [1].
Early histopathological findings seen after radiotherapy are defined as “diffuse alveolar damage” [2]. In this period, besides endothelial changes which cause an increase in capillary vascular permeability, protein exudation in alveolar spaces and alveolar wall oedema are observed. Thrombosis and consecutively intra-alveolar haemorrhage can develop in vessels that have focal necrosis. Inflammatory cell infiltration is distinctive [3, 4]. Fibroblastic proliferation and collagen accumulation occur both in intra-alveolar spaces and insterstitium. Alveolar septum thickening and fibrosis become obvious in a couple of weeks [5]. Acute changes that occur in lungs after radiotherapy are summarized in Table 1.
| Type 1 Alveolar cells | Degenerative changes or decline in normal counting. |
| Type 2 Alveolar cells | Normal or early degenerative changes become more distinctive. |
| Basal Membrane | Swelled up, disorder began. |
| Interstitial gap | Increase in the number of inflammatory cells, increase in loose nexus tissue. |
| Alveolar gap | Filled with hemorragical fibrin and cell debris. Increase in the number of alveolar macrophages. |
Many risk factors has been described in literature for the development of radiation pneumonia. These are low performance score, low lung capacity, smoking history, other genetic factors such as changes in plasma TGF-β levels, radiotherapy fractionation schema, practice of chemotheraphy simultaneous with radiotherapy, fraction dose and radiotherapy field size [7-19]. When 18-20 Gy applied on the whole lung in standart fraction and doses (1.5/2 Gy/ fraction/ day), radiation pneumonia is observed at least in 5% phenomenon [20, 21]. However, no studies are found in literature that investigate the effect of gender difference on radiotherapy-dependent lung toxicity.
Materials and methodsTop
This study is performed by Trakya University Faculty of Medicine (TÜTF), Department of Radiation Oncology, Department of Pathology and Department of Biophysics in Experimental Animal Laboratory of the Trakya University. Our study is approved by Trakya University Animal Experiment Local Ethics Committee (TÜHDYEK) with the protocol number TÜHDYEK – 2011/09 (Ap. 1).
Protocol of experiment
Fourty Wistar Albino type rats provided from Trakya University Laboratory Animals Research Laboratory were used in the study (Baseline, 20 females, average weight 200 gr and 20 males, average weight 260 gr). Four groups each including 10 rats were constituted in the study. 1) Control 1 group onsisted of only female rats; 2) Control 2 group, only male rats; 3) Experiment 1 group, RT applied female rats; and 4) Experiment 2 group, RT applied male rats.
Rats were followed for 6 weeks during the procedure. 50 mg/kg bw ketamine (Ketasol, Richter Farma AG, Austria) and 3.9 mg/kg bw xylazine (Rompun, Bayer Turkish Chemical Industry Limited Company, Turkey) were used for general anesthesia throughout the experiment.
For irradiation, rats in the radiotherapy group were immobilised in prone position on blue foam after anesthesia was applied (Figure 1). Radiotherapy fields of lungs of 6x4 cm size for females and 6x5 cm size for males were simulated. Simulation radiography was taken for the field viewing of first rats simulated. Front and back thickness of rats were measured by a ruler and half-thickness of d=1.5 cm for females and d=1.75 cm for males were detected. In the 0 day of our study, 10 Gy radiotherapy was applied in one fraction to right and left lungs of all rats with Varian Linak Device (linear accelerator) to the field determined by measuring dose in half-thickness in the manner that source – skin distance would be 100 cm.
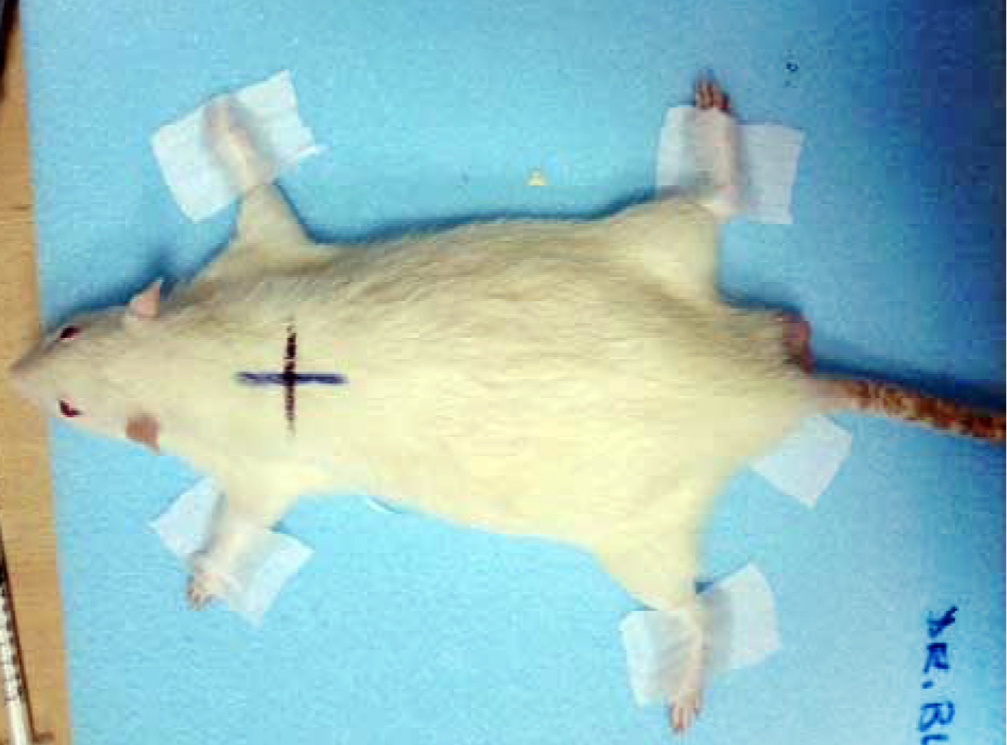
Figure 1 Rats simulated in prone position.
For sacrification, after radiotherapy applied for 6 weeks, heart blood was taken from deeply anesthetised rats and sacrification was done. Lung tissue samples were taken and fixed in formaldehyde for histopathological study. After 24 h fixation, one piece from each lung tissue was taken for tissue processing; embedded into parafine; 4 micron sections were taken dyed with hematoxylin – eosinand examined under light microscope. Tissues were dyed with Sirius red as well, to evaluate fibrosis. Samples were evaluated separately in control and RT groups for pneumonia and vasculitis, and scored as 0 (none) and 1 (exists).
If histologically, haematogenous exudates that fill the alveoli and hyaline membranes composed of fibrin besides inflammatory cells were observed, the case was classified as pneumonia “exists”. If vasculer damage and collagen deposition were apparent as well as fibrin deposition, necrosis and inflammatory cell infiltration on vessel walls, the case was classified as vasculitis “exists”.
Statistical analysis
Data on histopathological findings are expressed as “none – exists” and “p ratio”. Kruskal Wallis test is used to determine inter-group differences. Bonferroni and Mann Whitney-U tests are used to determine from which group or groups this difference is derived, when a significant difference is detected. p<0.05 ratio is accepted as statistically significance limit.
ResultsTop
The development of RT-induced changes were statistically higher in the RT-male and RT-female groups as compared to the control groups (RT-induced pneumonitis: RT-male and control male, p=0.004, RT-female and control female, p=0.01; Vasculitis: RT-male and control male, p=0.009, RT-female and control female, p=0.08), as expected (Tables 2 & 3).
| RT-male | Control-male | p | |
| Pneumonitis: yes/no | 6/4 | 0/10 | 0.004 |
| Vasculitis: yes/no | 8/2 | 2/10 | 0.009 |
p<0.05= statistical meaningful; p = Kruskal Wallis test (Bonferroni arranged Mann Whitney U test is used for multiple comparisons).
| RT-female | Control-female | p | |
| Pneumonitis: yes/no | 5/5 | 0/10 | 0.01 |
| Vasculitis: yes/no | 7/3 | 1/10 | 0.08 |
p<0.05= statistical meaningful; p = Kruskal Wallis test (Bonferroni arranged Mann Whitney U test is used for multiple comparisons).
No significant difference between RT-male group and RT-female group was detected in the development of RT-induced pneumonitis (60% vs. 50%, respectively, p=0.66) and vasculitis (80% vs. 70%, respectively, p=0.61) (Table 4). The histopathological changes induced by irradiation in the lung tissue are shown in Figure 2 (pneumonitis) and Figure 3 (vasculitis) in the RT-male, RT -female, and control groups.
| RT-male | RT-female | p | |
| Pneumonitis: yes/no | 6/4 | 5/5 | 0.66 |
| Vasculitis: yes/no | 8/2 | 7/3 | 0.61 |
p<0.05= statistical meaningful; p = Kruskal Wallis test (Bonferroni arranged Mann Whitney U test is used for multiple comparisons).

Figure 2 Inflammatory cell infiltration around the bronchioles and edema in the alveoli for the RT-male (a) and RT-female (b) groups. The regular vascular structure, lung parenchyma, and the alveoli, for the control-male group (c) (H&Ex200).
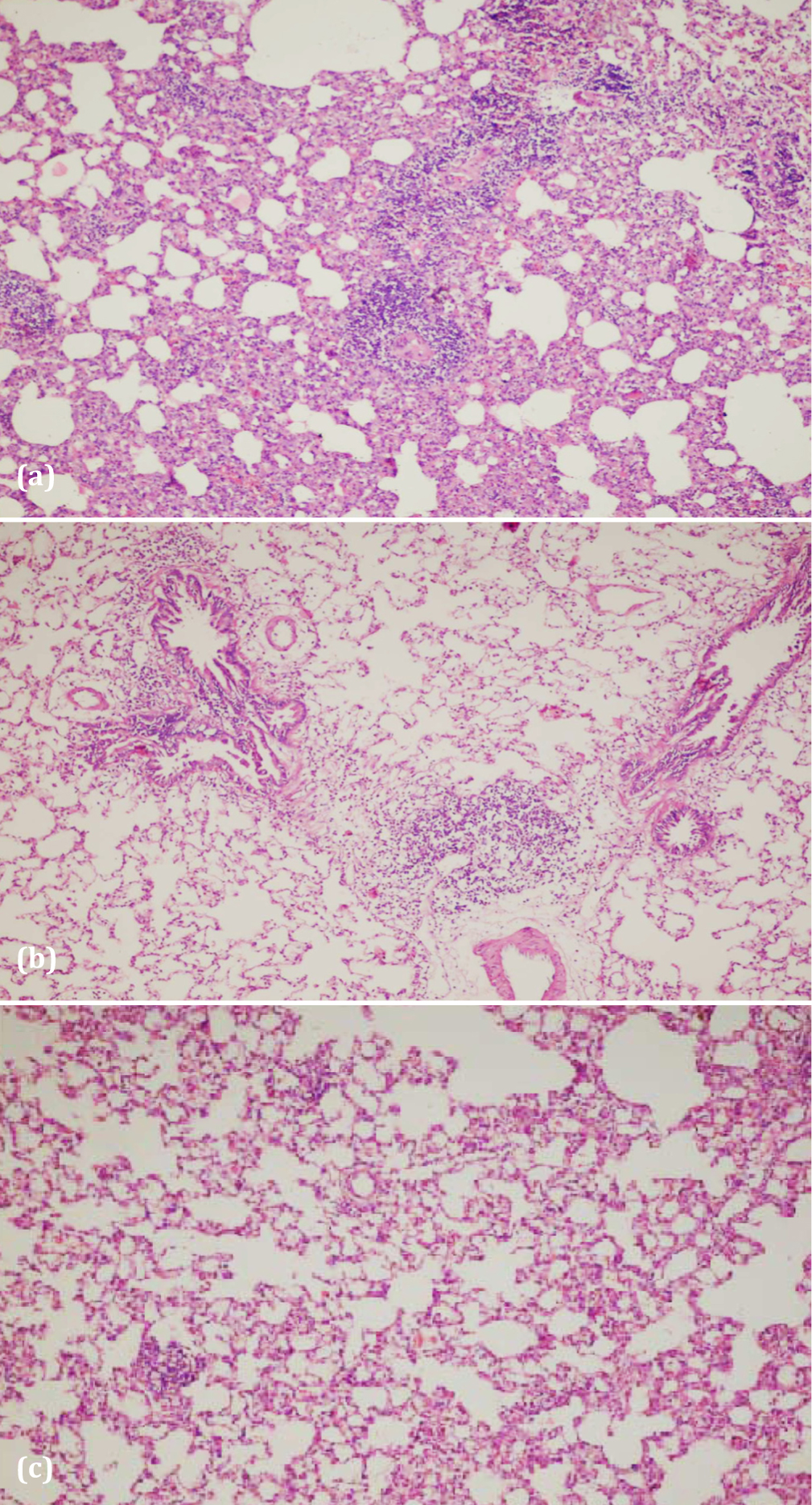
Figure 3 The mix type inflammatory cell infiltration around the vascular structure for the RT-male (a) and RT-female (b) groups and the regular vascular structure for the control-female group (c), (H&Ex200).
DiscussionTop
Current data about histopathological changes after irradiation of human lung are limited since it is not possible for patients to go into diagnostic thoracotomy after lung RT and explanatory autopsies are done rarely. Therefore, current data are obtained mostly from animal models.
In our study, we intended to explore that if radiation-induced acute lung toxicity had a relation to gender or not. For this purpose, we applied 10 Gy radiotherapy to the lung of male and female rats which have its own control groups. The dose we gave was enough to show radiation damage in lung tissue. Gender difference in cancer treatment is recently a subject of interest in literature. Besides evaluating treatment effectiveness in both prospective and retrospective clinical studies, the influence of gender difference on treatment response and in occurence of adverse events are also investigated. In literature, data is focused mostly upon colorectal cancer [22-25]. Kaminski et al. investigated side effects in rectal cancer treatment and showed that therapy had more toxic effects on females [22].
On the other hand, when gender difference was questioned in lung cancer, data did not go beyond epidemiology and information on it was far too limited. Our study is the first in literature that investigates the effect of gender difference in radiation-induced lung toxicity histopathologically, by focusing on development of vasculitis and pneumonia.
It is shown that inflammation plays a lead role in the beginning and development of radiation pneumopathy [26]. Inflammatory leucocytes bind adhesion molecules tightly to endothelial cells of microvascular system. Lymphocytic and macrophage transmigration to interstitium follow it. Production of various cytokines causes the activation of fibroblasts and endothelium and this leads to paracrine and autocrine loop formation among fibroblasts, endothelin and macrophages. In the histopathological examination, pulmonary oedema, mixed inflammatory cell infiltration centered around vessels, mixed inflammatory cell infiltration in focal focuses, and subsequent development of vasculitis and pneumonia are considered as indicators of the damage in lung. In our study we, besides observing signs of pneumonia and vasculitis, also showed a statistically significant difference between control and RT groups both in male and female rats [Tables 2 & 3]. However, there was no difference between male and female genders in terms of neither pneumonia nor vasculitis development.
ConclusionTop
we could not detect histopathologically the effect of gender difference on acute lung toxicity due to radiotherapy. However, gender difference in cancer treatment is a new and open-to-investigation subject in literature. More studies are needed in this subject.
Conflict of interest
The authors wish to express that they have no conflict of interest.
ReferencesTop
[1] Bradley J, Graham MV, Winter K, Purdy JA, Komaki R, et al. (2005) Toxicity and outcome results of RTOG 9311: A phase I-II dose-escalation study using three dimensional conformal radiotherapy in patients with inoperable non-small-cell lung carcinoma. Int J Radiat Oncol Biol Phys 61:318–328. Article Pubmed
[2] Rosai J (1996) Acute pulmonary injury and interstitial pneumonia. In: Rosia J, editor. Ackerman’s surgical pathology. Vol. 1, 8th ed. New York: Mosby. pp. 358–369.
[3] Gross NJ (1980) Experimental radiation pneumonitis: IV. Leakage of circulatory proteins onto the alveolar surface. J Lab Clin Med 95:19–31. Pubmed
[4] Abdollahi A, Li M, Ping G, Plathow C, Domhan S, et al. (2005) Inhibition of plateletderived growth factor signaling attenuates pulmonary fibrosis. J Exp Med 201:925–935. Article Pubmed
[5] McDonald S, Rubin P, Philips TL, Marks LB (1995) Injury to the lung from cancer therapy: clinical syndromes, measurable endpoints, and potential scoring systems. Int J Radiat Oncol Biol Phys 31:1187-1203. Article Pubmed
[6] Coggle JE, Lambert EB, Moores SR (1986) Radiation effects in the lung. Environ Health Perspect 70:261-291. Article
[7] Tsujino K, Hirota S, Endo M, Obayashi K, Kotani Y, et al. (2003) Predictive value of dose–volume histogram parameters for predicting radiation pneumonitis after concurrent chemoradiation for lung cancer. Int J Radiat Oncol Biol Phys 55:110–115. Article Pubmed
[8] Lind PA, Marks LB, Hollis D, Fan M, Zhou SM, et al. (2002) Receiver operating characteristic curves to assess predictors of radiation-induced symptomatic lung injury. Int J Radiat Oncol Biol Phys 54:340–347. Article Pubmed
[9] Kasten-Pisula U, Tastan H, Dikomey E (2005) Huge differences in cellular radiosensitivity due to only very small variations in double-strand break repair capacity. Int J Radiat Biol 81:409–419. Article Pubmed
[10] Kong FM, Anscher MS, Sporn TA, Washington MK, Clough R, et al. (2001) Loss of heterozygosity at the mannose 6-phosphate insulin-like growth factor 2 receptor (M6P/IGF2R) locus predisposes patients to radiation- induced lung injury. Int J Radiat Oncol Biol Phys 49:35–41. Article Pubmed
[11] Monson JM, Stark P, Reilly JJ, Sugarbaker DJ, Strauss GM, et al. (1998) Clinical radiation pneumonitis and radiographic changes after thoracic radiation therapy for lung carcinoma. Cancer 82:842–850. Article Pubmed
[12] Anscher MS, Murase T, Prescott DM, Marks LB, Reisenbichler H, et al. (1994) Changes in plasma TGF beta levels during pulmonary radiotherapy as a predictor of the risk of developing radiation pneumonitis. Int J Radiat Oncol Biol Phys 30:671–676. Article Pubmed
[13] Segawa Y, Takigawa N, Kataoka M, Takata I, Fujimoto N, et al. (1997) Risk factors for development of radiation pneumonitis following radiation therapy with or without chemotherapy for lung cancer. Int J Radiat Oncol Biol Phys 39:91–98. Article Pubmed
[14] Mah K, Keane TJ, Dyk JV, Braban LE, Poon PY, et al. (1994) Quantitative effect of combined chemotherapy and fractionated radiotherapy on the incidence of radiation-induced lung damage: A prospective clinical study. Int J Radiat Oncol Biol Phys 28:563–574. Article Pubmed
[15] Roach M, Gandara DR, Yuo HS, Swift PS, Kroll S, et al. (1995) Radiation pneumonitis following combined modality therapy for lung cancer: Analysis of prognostic factors. J Clin Oncol 13:2606–2612. Article Pubmed
[16] Dillman RO, Seagren SL, Propert KJ, Guerra J, Eaton WL, et al. (1990) A randomized trial of induction chemotherapy plus high-dose radiation versus radiation alone in stage 3 non-small-cell lung cancer. N Engl J Med 323:940–945. Article Pubmed
[17] Dillman RO, Herndon J, Seagren SL, Eaton WL Jr, Gren MR (1996) Improved survival in stage 3 non-small-cell lung cancer: seven-year follow-up of Cancer and Leukemia Group B (CALGB) 8433 trial. J Natl Cancer Inst 88:1210–1215. Article Pubmed
[18] Yamada M, Kudoh S, Hirata K, Nakajima T, Yoshikawa J (1998) Risk factors of pneumonitis following chemoradiotherapy for lung cancer. Eur J Cancer 34:71–75. Article Pubmed
[19] Byhardt RW, Martin L, Pajak TF, Shin KH, Emami B, et al. (1993) The influence of field size and other treatment factors on pulmonary toxicity following hyperfractionated irradiation for inoperable non–small cell lung cancer (NSCLC)—Analysis of a Radiation Therapy Oncology Group (RTOG) protocol. Int J Radiat Oncol Biol Phys 27:537–544. Article Pubmed
[20] Perez CA, Brady LW (2004) Principles and practice of radiation oncology (4nd ed). Lippincott Company, Philadelphia.
[21] Cox JD, Ang KK (2003) Radiation Oncology Rational, Technique, Results (8th ed), Mosby.
[22] A Kaminski, D Joseph, H Elsaleh (2007) Differences in toxicity across gender in patients treated with chemoradiation for rectal cancer. Australas Radiol 51:283–288. Article Pubmed
[23] Sloan JA, Goldberg RM, Sargent DJ, Vargas-Chanes D, Nair S, et al. (2002) Women experience greater toxicity with fluorouracil-based chemotherapy for colorectal cancer. J Clin Oncol 20:1491–1498. Article Pubmed
[24] Elsaleh H, Joseph D, Grieu F, Zeps N, Spry N, et al. (2000) Association of tumour site and sex with survival benefit from adjuvant chemotherapy in colorectal cancer. Lancet 355:1745–50. Article Pubmed
[25] Pahlman L, Glimelius B (1990) Pre- or postoperative radiotherapy in rectal and rectosigmoid carcinoma. Report from a randomized multicenter trial. Ann Surg 211:187–195. Article Pubmed
[26] Ward PA, Hunninghake GW (1998) Lung inflammation and fibrosis. Am J Respir Crit Care Med 1998;157:S123–S129. Article Pubmed



