Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 8, August 2014, Pages 122-127
Original researchOpen Access
Breast cancer receptor status assessment and clinicopathological association in Nigerian women: A retrospective analysis
-
Samira BL. Makanjuola1,*
 ,
Sanni D. Ayodele2,
Farideh A. Javid3,
John O. Obafunwa2,
Mobolaji A. Oludara4 and
Abiodun O. Popoola5
,
Sanni D. Ayodele2,
Farideh A. Javid3,
John O. Obafunwa2,
Mobolaji A. Oludara4 and
Abiodun O. Popoola5
- 1 Department of Pharmacology, Lagos State University, College of Medicine (LASUCOM), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 2 Department of Pathology and Forensic Medicine, Lagos State University Teaching Hospital (LASUTH), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 3 Department of Pharmacy, School of Applied Sciences, University of Huddersfield, Queensgate, Huddersfield HD1 3DH, United Kingdom
- 4 Department of Surgery, Lagos State University Teaching Hospital (LASUTH), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 5 Department of Oncology, Lagos State University Teaching Hospital (LASUTH), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
*Corresponding author: Samira Lobo Makanjuola, Department of Pharmacology, Lagos State University, College of Medicine (LASUCOM), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria. Tel.: +234 80 96633500; Email: sbatistalobo@hotmail.com
Received 4 May 2014 Revised 16 July 2014 Accepted 24 July 2014 Published 29 July 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-19
Copyright: © 2014 Makanjuola SBL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Background: Breast cancer markers are becoming increasingly important in breast cancer research due to their impact on prognosis, treatment and survival. The present retrospective study was carried out to quantify the proportion of estrogen (ER), progesterone (PR), and human epithelial receptor 2 (HER2) expressions and their association with tumour grade, age, and tumour size in breast cancer patients in Nigeria. Materials and methods: The paraffin embedded tissue sections were analysed for breast cancer markers using monoclonal antibody SP1 for ER and SP2 for PR and polyclonal antibody ErbB2 for HER2. Results: A total of 286 breast cancer paraffin wax tissue sections were analysed for ER, PR and HER2 expression. Of all the tissue samples examined, 20 (7%) were ER-positive, 6 (2.1%) were PR-positive, 11 (3.8%) were HER2-positive whereas 248 (87%) were triple-negative breast carcinoma. ER- and PR-positivity was associated with early grade I and II tumours (P < 0.010-0.009) and tumour sizes of < 50mm (p < 0.001). HER2-positivity was positively (P < 0.009) associated with grade II tumours and negatively (P < 0.0001) associated with grade III tumours. Triple-negative breast cancer was associated with grade III tumours (P < 0.0001) and larger tumour sizes of > 50mm (P < 0.0001). Conclusion: A small proportion of Nigerian women with breast cancer are ER/PR-positive which are associated with less aggressive, better prognosis and benefit from endocrine therapy. An even smaller proportion of patients with aggressive tumors were HER2-posivite but responsive to Herceptin treatment. Unfortunately, a very high proportion of cases were triple-negative which is associated with very aggressive tumours and no targeted treatment, which may explain the high mortality rates from breast cancer in Nigeria.
Keywords: estrogen; progesterone; HER2; breast carcinoma; Nigerian; tumour grade; tumour size
IntroductionTop
Breast cancer is the most common neoplasia both in the developed and developing countries. The incidence rates vary greatly worldwide from 19.3 per 100,000 in Eastern Africa, to 89.7 per 100,000 in Western Europe [1]. Although the lowest incidence rates of breast cancer are found in African countries, the incidence is increasing. In Nigeria, breast cancer occupies number 12th position among 20 causes of death in women [1]. Breast cancer survival rates also vary greatly among countries, ranging from 80% and over in Northern America, Sweden and Japan to around 60% in middle-income countries and below 40% in low-income countries [2-3]. The low survival rates in less developed countries are associated with ineffective early detection programmes resulting in late presentation as well as the lack of adequate diagnosis and treatment facilities.
The occurrence, progression and prognosis of breast cancer depend on complex processes involving various genes, pathways and protein expression and regulation. Therefore it is difficult to determine the biological behaviour and prognosis of breast cancer based on the assessment of a single factor. Immunohistochemistry (IHC) has become essential to many malignancies and plays a key role in tumour diagnosis, treatment and prognosis assessment [4].
Several tumours are hormone-dependent and breast cancer is a typical example. Estrogen (ER) and progesterone (PR) play important roles in the growth and differentiation of breast cancers making them important prognostic markers [5]. Human epithelial receptor 2 (HER2), a proto-oncogene also known as ErbB2-neu, located on chromosome 17q21 is also considered to be closely associated with occurrence and development of breast cancer [6].
Different expression patterns of ER, PR and HER2 have been identified, making knowledge of the receptor content of breast carcinoma essential in planning the management of disease [7-8]. ER over expression has been predominantly observed in lower grade, smaller size-tumours, more likely to be node negative, and shows better survival outcome than ER-negative cancers [7, 9-10]. PR over expression is also associated with well differentiated tumours with good overall survival [11]. The over expression of ER is reported to occur in approximately 70-80% of invasive breast carcinoma at the point of diagnosis. Over expression of HER2 is associated with higher grade [12], and ER-negative tumours [13], which demonstrate poor overall survival [14]. The HER2 over expression is reported to occur in 10-30% of invasive breast cancers [15]. Another subtype usually identified in breast cancer classification is the triple-negative. Triple-negative breast cancers are tumours characterized by their lack of hormone receptors (ER and PR) and HER2. They are the most aggressive form and account for 10-17% of all breast cancers [17].
The Lagos State University Teaching Hospital (LASUTH), Nigeria receives an average of 150 patients diagnosed with breast cancer annually. Majority of patients diagnosed with invasive breast carcinoma undergo surgery, followed by chemotherapy and radiotherapy as well as hormonal therapy.
In Nigeria, receptor status assessment for breast cancer patients is currently unavailable in many centers. However, tamoxifen is prescribed based on evidence suggesting that ER/ PR-positivity rates in Nigerians are the same as in Western countries [17]. Therefore, the aim of this study was first to carry out retrospective investigation to study the reliability of routinely processed paraffin wax sections in the assessment of ER, PR, and HER2 in a series of breast carcinomas. Second, to evaluate the association between ER-, PR-, HER2-positivity, and triple-negative tumours with other tumour characteristics including age at presentation, size of the tumour, and grade of the tumour.
Materials and methodsTop
Data collection
Approved by the Faculty Research Board of Nigeria, the Lagos State University Teaching Hospital Cancer Registry was queried from 1 January 2009 to 31 December 2011 and provided the following data on each invasive breast cancer subject: age, cancer site, tumour characteristics (morphology, grade, and size), and nodal disease status.
Two hundred and eighty six formalin fixed, paraffin embedded tissue block samples from the breast lesions were investigated. Data related from the studied subjects were retrieved from Oncology Centre, LASUTH, Lagos, Nigeria. All samples were obtained from females with breast lesions, their ages ranging from 23 to 82 years with a mean age of 48.9 years old.
Sample processing
Serial sections on HistoBond slides for Immunohistochemistry (IHC) and one section on a regular slide for haematoxylin and eosin (H&E) procedure were prepared for each case. The Immunohistochemistry staining was performed as per standard protocols. Briefly, slides were heated overnight at 60˚C, followed by deparaffinization in xylene and through graded ethyl alcohols and rehydration using the aqueous buffer. Before immunostaining with antibodies, the deparaffinized slides were incubated in 0.3% hydrogen peroxide in methanol for 20 min to inhibit endogenous peroxidise activity, then washed 3 times with IHC wash buffer (Bethyl Lab., UK). The tissues were subsequently treated with IHC retrieval buffer (PH 6.0) (Bethyl Lab., UK) at 96˚C for 20 min for antigenic retrieval, and then washed 3 times with IHC wash buffer. Further slides were incubated in IHC blocking buffer (Bethyl Lab., UK) for 15 min to prevent non specific binding of antibodies. The IHC blocking agent was then drained and slides were incubated with the primary antibody for 1 h in a humidity chamber using the following dilutions: ER (clone SP1, titer 1:50, Abcam), PR (clone SP2, titer 1:50, Abcam), HER2/neu (ErbB2, titer 1:150, Bethyl Lab, UK). After rinsing the primary antibody thrice using IHC wash buffer for 5 min in each, the slides were incubated in secondary antibody labelled with HRP (Bethyl Lab., UK) for 1 h in the same chamber. Detection of labelled secondary antibody was detected using the DAB substrate ABC (Bethyl Lab., UK) according to manufacturer’s instructions. The sections were counter stained using haematoxylin, dehydrated using ethyl alcohol, cleared using xylene, and mounted in DPX then examined under light microscope. All sections were performed at the same time and submitted to standard methods. Known positive and negative cases were used as external controls. Two investigators evaluated the sections independently. Positive expression for each tumour marker was defined as in the literature: ER and PR, were considered positive when >10 of the nuclei were stained in 10 high power field (HPF) [18]. HER2/neu was considered negative when scored 0 and +1, positive with score +2 and +3. Cellular membrane should be completely stained in >10% tumour cells for it to be considered as +2, +3. Cells with no staining, or weak staining in part of the cell membrane and in <10% of the tumour cells were considered negative [19].
Statistical analysis
SPSS Software version 17.0 was used for all statistical analysis and p-value of 0.05 or less was regarded as statistically significant. Differences in subjects and tumour characteristics between the various breast cancer subtypes were analysed using analysis of variance for continuous variables and Chi-square test for categorical variables.
Ethical consent
The studied protocol was submitted and approved by the Faculty Research Board of Nigeria in Collaboration with the Department of Pathology and Forensic Medicine, Lagos state Teaching Hospital, Nigeria.
ResultsTop
Two hundred and eighty six formalin fixed, paraffin embedded tissue block samples from the breast lesions were investigated. These included 262 (92%) invasive ductal carcinomas, 13 (4.6%) invasive lobular carcinomas, 6 (2.1%) invasive mucinous carcinomas, 2 (0.7%) invasive papillary carcinoma, 1 (0.4%) medullary carcinoma and 1 (0.4%) mixed invasive ductal and lobular carcinoma (Table 1).
Eighty eight (31.1%) cases presented a tumour size of < 20mm, 102 (36.0%) presented a tumour size between 20-50mm, and 93 (32.9%) presented a tumour size of > 50mm (Table 1). Thirty six (12.7%) cases were tumour grade I, 136 (48.0%) were tumour grade II, and 111 (39.3%) were tumour grade III (Table 1). Only 51 (17.8%) cases were examined for lymph node positivity, with 42 (82.4%) being positive and 9 (17.6%) being negative (Table 1).
| Clinicopathological features | Frequency | Percentage |
| Histological type (available for 285 (99.6%)) | ||
| Invasive ductal carcinoma | 262 | 92% |
| Invasive lobular carcinoma | 13 | 4.6% |
| Invasive mucinous carcinoma | 6 | 2.1% |
| invasive papillary carcinoma | 2 | 0.7% |
| Medullary carcinoma | 1 | 0.4% |
| Mixed Invasive ductal & lobular carcinoma | 1 | 0.4% |
| Total | 285 | 100% |
| Tumour size (available for 283 (98. 9%)) | ||
| <2 cm | 88 | 31.1% |
| 2-5 cm | 102 | 36.0% |
| >5 cm | 93 | 32.9% |
| Total | 283 | 100% |
| Tumour grade (available for 283 (98. 9%)) | ||
| Grade I | 36 | 12.7% |
| Grade II | 136 | 48.0% |
| Grade III | 111 | 39.3% |
| Total | 283 | 100% |
| Lymph node status (available for 51 (17.8%)) | ||
| Negative | 9 | 17.6% |
| Positive | 42 | 82.4% |
| Total | 51 | 100% |
| Body mass index (BMI) (available for 42 (14.7%)) | ||
| Underweight <18.5 | 5 | 11.9% |
| Normal 18.5-24.9 | 14 | 33.3% |
| Overweight 25-29.9 | 13 | 31.0% |
| Obese >30 | 10 | 23.80% |
| Total | 42 | 100% |
Other factors including BMI were available for 42 (14.8%) cases, with 5 (11%) being underweight, and larger proportions being either of normal weight 14 (33.3%), overweight 13 (31%) or obese 10 (23.3%). The triple-negative phenotype was observed in 248 (87%) of eligible breast cancer cases, 20 (7%) were classified as ER-positive, 6 (2.1%) were classified as PR-positive, and 11 (3.9%) were classified as HER2-positive (Table 2) (Figure 1).
| Marker | Positive | Negative | ||
| Cases (286) | Frequency | Percentage | Frequency | Percentage |
| ER | 20 | 7.0% | 266 | 93.0% |
| PR | 6 | 2.1% | 280 | 97.9% |
| HER2/neu | 11 | 3.9% | 275 | 96.2% |
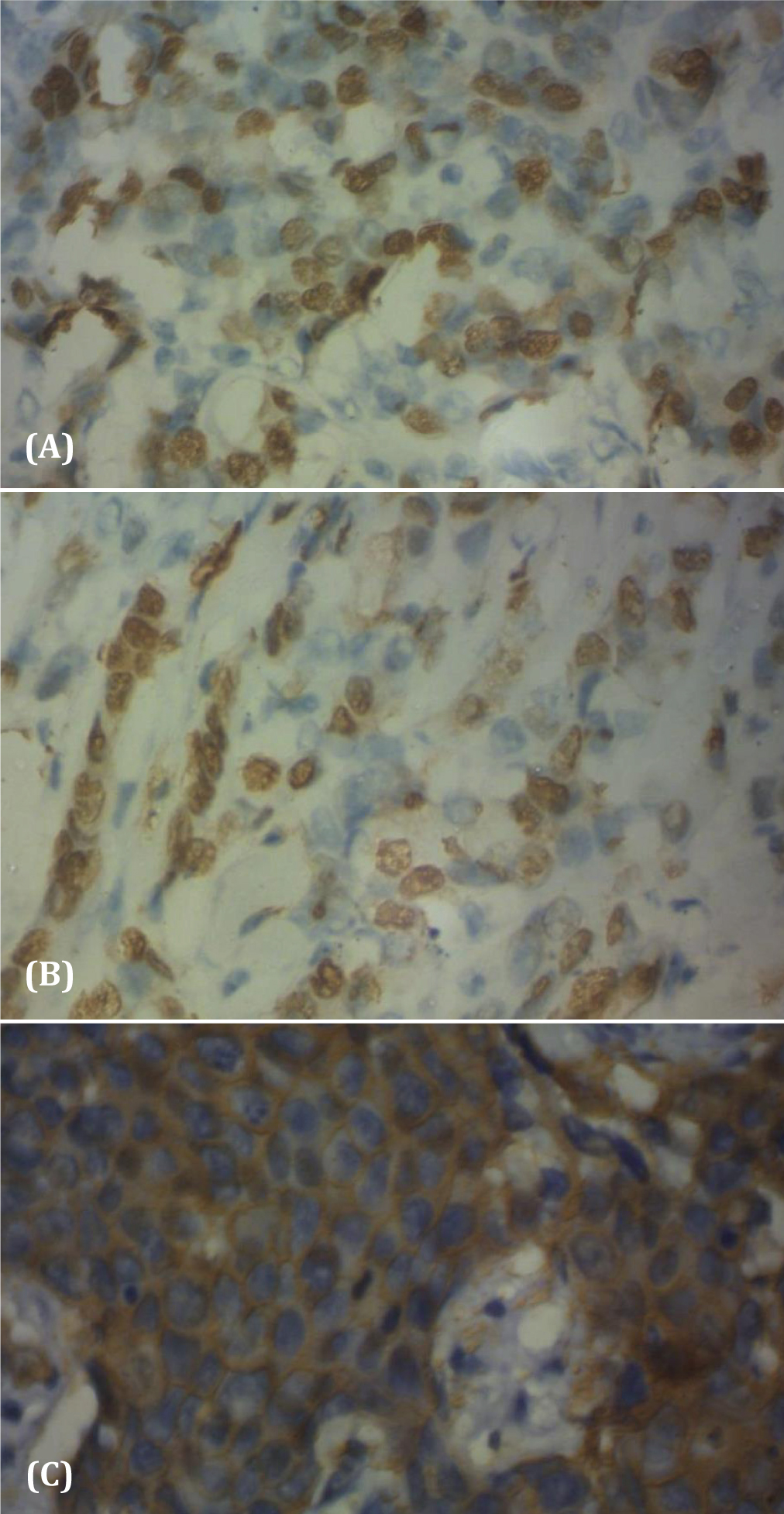
Figure 1 Positive expression of Immunohistochemical profiles in invasive breast cancer tissues. ER (A) and PR (B) are only present in the nucleus and HER2 (C) is mainly present in the cytoplasm. Magnification x100.
The average age of presentation for ER/PR-positive cases was 51.3 years old whereas for HER2-positive and triple-negative breast cancer was 48.1 and 48.8, respectively. Although ER/PR-positive breast cancer cases were associated with older age and HER2-positive and triple-negative with younger age, the differences between the groups were not significant (Table 3). Tumour grade was significantly associated with breast cancer receptor subtypes. A significant (P < 0.019) proportion of grade I tumours were ER/PR-positive whereas a significant (P < 0.009) proportion of grade II tumours were HER2-positive (Table 3).
| ER/PR+ (26) | HER2+ (12) | ER/PR-, HER2- (248) | p-value | ||
| Age (years) | 51.3± 11.2 | 48.1±7.9 | 48.8± 12.7 | 0.593 | |
| Tumour grade | |||||
| I | 6 (23.1%)* | 1 (9.1%) | 29 (11.7%) | *<0.019 | |
| II | 11 (42.3%) | 8 (72.7%)* | 119 (48.0%) | *<0.009 | |
| III | 9 (34.6%) | 2 (18.2%)* | 100 (40.3%) | *<0.0001 /td> | |
| Tumour size | |||||
| <20mm | 9 (34.6%) | 4 (33.3%) | 75 (30.2%) | 0.336 | |
| 20-50mm | 15 (57.7%)* | 4 (33.3%) | 85 (34.3%) | *<0.001 | |
| >50mm | 2 (7.7%)* | 3 (27.2%) | 91 (36.7%) | *<0.0001 | |
Tumour size was also significantly associated with breast cancer receptor subtypes. Although there was no statistically significant differences in expression for ER/PR and HER2 in smaller tumour sizes (< 20mm), there was a significant (p < 0.001) proportion of ER-positive tumours that were between 20-50mm and a significant (P > 0.0001) proportion of HER2-positive and triple-negative were associated with larger tumour sizes (>50mm) (Table 3).
DiscussionTop
Our findings revealed that a large number of breast cancer cases (87%) in Nigeria are triple-negative. Only 7% of cases account for ER-positive, 2.1% account for PR-positive, and 3.8% for HER2-positive breast cancers. The immediate realisation of a possible misclassification in these samples arises from the high proportion of triple-negative (87%) and low proportion of ER (7%), PR (2.1%), and HER2 (3.8%). However, studies of breast cancer markers in Sub-Saharan Africa have had extremely variable findings with reported percentage of ER-negative tumours ranging from 40% [17, 20] to >70% [21, 22]. In comparison, corresponding percentages in the black American population were 35% in breast cancer patients aged 40 and 15% to 20% by age 70 [23]. A study carried out at the University of Michigan evaluated the pattern of ER, PR, HER2, and triple-negative breast cancers in white American, black American, Ghanaian/African background and showed a high prevalence of triple-negative breast cancers in Ghanaian women (82.2%) followed by black American (26.4%) and white American (16%) women [22].
The high prevalence of triple-negative breast cancer in the present study (87.0%) is in concordance with the reported data for the Ghanaian/African background in the Michigan study (82.2%) [22]. The prevalence of ER-positive and/or PR-positive, and HER2-positive breast cancer in the Michigan study was 61.9% for white American, 49.4% for black American, and 13.3% for Ghanaian population [22]. Results for ER-positive and/or PR-positive, and HER2-positive breast cancer in our study were 13.0%. Another study evaluating the hormone expression in East African breast cancer cases also showed poor ER and PR-positivity (24% and 10%, respectively) and high prevalence of triple-negative (66%) breast cancer [22].
The IHC-based classification systems are still considered useful in clinical practice, especially when fresh tissue is not available and has been shown to correlate well with intrinsic classification using gene expression microarrays [24]. It is worth noting, however, that the reliability of the ER/PR and HER2 testing is imperfect. There are substantial intra-laboratory and inter-laboratory variation in results because of fixation, antigen retrieval, and staining methods that may vary among laboratories [18]. The drawback of ER testing is that results are highly sensitive to biopsy-tissue fixation and processing procedures, which leads to false negatives worldwide [19]. Ideally, receptor status should be determined in biopsy specimen obtained before pre-operative neoadjuvant therapy with IHC performed shortly after to avoid antigen degradation [25]. Moreover, if receptor status is determined from mastectomy tissue taken after neoadjuvant chemotherapy, the tumour phenotype may no longer be the same. Substantial discordance has also been reported in HER2 results produced in different laboratories from the same specimen [18, 26]. The present study raises two issues of vital importance in studying the breast cancer subtypes. First, whether cancer tissue samples are processed, preserved and stained in line with international guidelines Having carried out a retrospective study, the second issue would be the lengthy storage of the tissue samples. The latter is a deterrent factor in determining breast cancer subtypes as it is widely known that IHC performs better on fresh samples and it is affected by the fixation methods, reagents used, protocols, antibody sensitivity, and scoring system [27]. Tissue samples used in this study were obtained from archived samples from 2008 to 2011 and we do lack information on how tissues were fixed and processed. Such limitations suggest that possible misclassification may occur. Nonetheless, it is important to evaluate the breast cancer receptor status profile for this sample group in order to understand the possible limitations of this study and exercise ways to assist this Institute in establishing standards for molecular breast cancer diagnosis in the future.
Indeed, it was acknowledged that there will be misclassification in this retrospective study due to fixation and processing techniques as well as long-term storage and IHC staining methods. As a result, this breast cancer receptor classification study was complemented with other important clinical and prognostic variables for the individuals such as age, tumour size, tumour grade, and lymph node status [28]. The clinicopathological association data further affirms that the high proportion of triple-negative patients may be less associated with fixation, processing and storage techniques and more significantly associated with the biological nature of the tumours.
Stark and colleagues in 2010 demonstrated the impact of the racial/ ethnic composition in determining breast cancer receptor subtypes [22]. This study has demonstrated that the triple-negative subtype is not only associated with black African race but also tumour biology including tumour grade and size. ER/PR-positive breast cancer was significantly (P < 0.019) associated with grade I tumours and tumour sizes < 50mm (P < 0.001). As previously reported ER-positive cancers are associated with a lower grade, smaller size, more likely to be node negative and better survival outcome than ER-negative cancers [7]. It has been demonstrated that ER expression decreased with increasing histological grade, indicating that the lower the tumour cell differentiation, the lower the estrogen dependence, which would in turn affect the sensitivity to the hormone therapy [29-30]. Women with ER-positive breast cancer benefit from endocrine therapy explaining the better survival outcomes. Most evidence regarding the prognostic role of PR is based upon the assumption that PR expression indicates a functioning ER pathway [31]. Therefore, it has been shown that PR-positive and ER-positive tumours have a better response to endocrine therapy than ER-positive and PR-negative cancers [32].
HER2-positive were significantly (P < 0.009) associated with grade II tumours but not with grade I or III; and significantly (P < 0.0001) associated with larger tumours (> 50mm). In a similar study the frequency of HER2 over expression decreased significantly in low grade tumour and also in patients with high grade tumour [33]. More recently, Lal et al., in a study of 3,655 breast cancer cases reported that HER2 amplification and over expression are limited essentially to invasive breast carcinoma of intermediate grade to high grade [34]. Under normal physiological conditions HER2 is inactive; however, once activated it may enhance tumour invasion and metastases and increase degree of malignancy [35], which may explain HER2 association with intermediate to high grade tumours and large tumour sizes.
Triple-negative were more associated with grade II and grade III tumours and significantly (P < 0.0001) associated with larger tumour sizes (> 50mm). These pathological features translate into poorer clinical prognosis and the worst overall and disease-free survival. Histologically, triple-negative breast cancers are poorly differentiated and are characterized by an aggressive clinical history. Since there are no specific treatment guidelines for this subtype, triple-negative breast cancers are managed with standard treatment, which leaves them with a high rate of local and systemic relapse [36]. Therefore, this continues to direct the focus of ongoing research and it should be of great benefit to breast cancer patients of African background.
ConclusionTop
This study has supported the use of Immunohistochemistry classification as a clinical tool in the future as ER/PR and HER2 markers are widely available at a reasonable cost. This information is clinically useful, therapeutically informative and somewhat predictive. Additional efforts to translate the currently standardised research method into reliable and reproducible diagnostic protocols are ongoing.
Funding
This study was fully supported by a grant from the Caverton Offshore Support Group (COSG).
Acknowledgment
The authors would also like to thank the Lagos State University Teaching Hospital (LASUTH) and the Lagos State University College of Medicine (LASUCOM) for the support provided.
Conflict of interest
The authors express no conflict of interest.
ReferencesTop
[1] GLOBOCAN (2008) World Health Organization. Article
[2] Coleman MP, Quaresma M, Berrino F, Lutz JM, De Angelis R, et al. (2008) Cancer Survival in five continents: a worldwide population-based study (CONCORD). Lancet Oncol 9:730–756. Article Pubmed
[3] Makanjuola SBL, Popoola AO, Oludara MA (2014) Radiation therapy: A major factor in the five-year survival analysis of women with breast cancer in Lagos, Nigeria. Radiother Oncol 111:321–326. Article Pubmed
[4] Qiao EQ, Ji M, Wu J, Li J, Xu X, et al. (2013) Joint detection of multiple immunohistochemical indices and clinical significance in breast cancer. Mol Clin Oncol 1:703–710. Article Pubmed
[5] Recareanu F, Simionnescu C, Georgescu CV, Pirici E (2011) Ductal invasive mammary carcinoma-clinicopathological prognostic factors related to immunohistochemical expression of hormonal receptors and Her2/neu oncoprotein. Rom J Morphol Embrol 52:1059–1064. Article Pubmed
[6] Gown AM (2008) Current issues in ER and HER-2 testing by IHC in breast cancer. Mod Pathol 21:S8–S15. Article Pubmed
[7] Low SC, Dixon AR, Bell J, Ellis IO, Elston CW, et al. (1992) Tumour estrogen receptor content allows selection of elderly patients with breast cancer for conservative tamoxifen treatment. BrJ Surg 79:1314–1316. Article Pubmed
[8] Sacks NP, Baum M (1993) Primary management of carcinoma of the breast. Lancet 342:1402–1408. Article Pubmed
[9] Grann VR, Troxel AB, Zojwalla NJ, Jacobson JS, Hershman D, et al. (2005) Hormone receptor status and survival in a population-based cohort of patients with breast carcinoma. Cancer 103:2241–2251. Article Pubmed
[10] Fisher B, Redmond C, Fisher ER, Caplan R (1988) Relative worth of estrogen or progesterone receptor and pathologic characteristics of differentiation as indicators of prognosis in node negative breast cancer patients: findings from National Surgical Adjuvant Breast and Bowel Project Protocol B-06. J Clin Oncol 6:1076–1087. Article Pubmed
[11] Reiner A, Neumeister B, Spona J, Reiner G, Schemper M, et al. (1990) Immunocytochemical localisation of estrogen and progesterone receptor and prognosis in human primary breast cancer. Cancer Res 50:7057–7061. Article Pubmed
[12] Sjögren S, Inganäs M, Lindgren A, Holmberg L, Bergh J (1998) Prognostic and predictive value of c-erbB-2 overexpression in primary breast cancer, alone and in combination with other prognostic markers. J Clin Oncol 16:462–469. Article Pubmed
[13] Gago FE, Fanelli MA, Ciocca DR (2006) Co-expression of steroid hormone receptors (estrogen receptor alpha and/or progesterone receptor) and HER2/neu (c-erbB-2) in breast cancer: Clinical outcome following tamoxifen based adjuvant therapy. J Steroid Biochem Mol Biol 98:36–40. Article Pubmed
[14] Yamauchi H, Stearns V, Hayes DF (2001) When is a tumour marker ready for prime time A case study of c-erbB-2 as a predictive factor in breast cancer. J Clin Oncol 19:2334–2356. Article Pubmed
[15] Ciocca DR, Gago FE, Fanelli MA, Calderwood SK (2006) Co-expression of steroid receptors (estrogen receptor alpha and/or progesterone receptors and Her-2/neu: Clinical implications. J Steroid Biochem Mol Biol 102:32–40. Article Pubmed
[16] Nwachukwu C, Grushko T A, Xu J, Khramtsov A, Olopade OI (2009) BRCA1 methylation contributes to the triple-negative breast cancer phenotype. Cancer Res 69:4050. Article
[17] Adebamowo CA, Famooto A, Ogundiran TO, Aniagwu T, Nkwodimmah C, et al. (2008) Immunohistochemical and molecular subtypes of breast cancer in Nigeria. Breast Cancer Res Treat 110:183–188. Article Pubmed
[18] Rhodes A, Jasani B, Barnes DM, Bobrow LG, Miller KD (2000) Reliability of Immunohistochemical demonstration of estrogen receptors in routine practice: interlaboratory variance in the sensitivity of detection and evaluation of scoring systems. J Clin Pathol 53:125–130. Article Pubmed
[19] Yaziji H, Taylor CR, Goldstein NS, Dabbs DJ, Hammond EH, et al. (2008) Consensus recommendations on estrogen receptor testing in breast cancer by immunohistochemistry. Appl Immunohistochem Mol Morphol 16:513–520. Article Pubmed
[20] Awadelkarim KD, Arizzi C, Elamin EO, Hamad HM, De Blasio P, et al. (2008) Pathological, clinical and prognostic characteristics of breast cancer in Central Sudan versus Northern Italy: implications for breast cancer in Africa. Histopathology 52P:445–456. Article Pubmed
[21] Bird PA, Hill AG, Houssami N (2008) Poor hormone receptor expression in East African breast cancer: evidence of a biologically different disease Ann Surg Oncol 15:1983–1988. Article Pubmed
[22] Stark A, Kleer C, Martin I, Awuah B, Nsiah-Asare A, et al. (2010) African Ancestry and Higher Prevalence of Triple-Negative Breast Cancer:Findings From an International Study. Cancer 116: 4926–4932. Article Pubmed
[23] Anderson WF, Katki HA, Rosenberg PS (2011) Incidence of breast cancer in the United States: current and future trends. J Natl Cancer Inst 103:1397–1402. Article Pubmed
[24] Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, et al. (2006) Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 295:2492–2502. Article Pubmed
[25] Fergenbaum JH, Garcia-Closas M, Hewitt SM, Lissowska J, Sakoda LC, et al. (2004) Loss of antigenicity in stored sections of breast cancer tissue microarrays. Cancer Epidemiol Biomarkers Prev 13:667–672. Article Pubmed
[26] Roche PC, Suman VJ, Jenkins RB, Davidson NE, Martino S, et al. (2002) Concordance between local and central laboratory HER2 testing in the breast cancer intergroup trial N9831. J Natl Cancer Inst 94:855–857. Article Pubmed
[27] Shah S, Chen B (2011) Testing for HER2 in breast cancer: a continuing evolution. Patholog Res Int 2011:903202. Article Pubmed
[28] Onitilo AA, Engel JM, Greenlee RT, Mukesh BN (2009) Breast cancer subtypes based on ER/PR and Her2 expression: Comparison of Clinicopathologic features and survival. Clin Med Res 7:4–13. Article Pubmed
[29] Putti TC, El-Rehim DM, Rakha EA, Paish CE, Lee AH, et al. (2005) Estrogen receptor-negative breast carcinomas: a review of morphology and immunophenotypical analysis. Mod Pathol 18:26–35. Article Pubmed
[30] Rosa FE, Caldeira JR, Felipes J, Bertonha FB, Quevedo FC, et al. (2008) Evaluation of estrogen receptor alpha and beta and progesterone receptor expression and correlation with clinicopathologic factors and proliferative marker Ki-67 in breast cancers. Hum Pathol 39:720–730. Article Pubmed
[31] Ravdin PM, Green S, Dorr TM, McGuire WL, Fabian C, et al. (1992) Prognostic significance of progesterone receptor levels in estrogen receptor positive patients with metastatic breast cancer treated with tamoxifen; results of a prospective Southwest oncology group study. J Clin Oncol 10:1284–1291. Article Pubmed
[32] Payne SJ, Bowen RL, Jones JL, Wells CA (2008) Predictive markers in breast cancer - the present. Histopathology 52:82–90. Article Pubmed
[33] Lal P, Tan LK, Chen B (2005) Correlation of HER-2 status with estrogen and progesterone receptors and histologic features in 3655 invasive breast carcinomas. Am J Clin Pathol 123:541–546. Article Pubmed
[34] Huang HJ, Neven P, Drijkoningen M, Paridaens R, Wildiers H, et al. (2005) Association between tumor characteristics and HER-2/neu by immunohistochemistry in 1362 women with primary operable breast cancer. J Clin Pathol 58:611–616. Article Pubmed
[35] Guo H, Bai O (2008) Relationship between the expression of ER, PR, Her -2 in breast cancer and its clinical pathological features. Chin J Lab Diagn 12:1390–1392.
[36] Cleator S, Heller W, Coombes RC (2007) Triple-negative breast cancer: therapeutic options. Lancet Oncol 8:235–244. Article Pubmed


