Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 1, Issue 7, September 2013, Pages 178–185
Original researchOpen Access
Evaluation of serum big endothelin-1 for the diagnosis and prediction of disease recurrence in breast cancer patients
- 1 Applied Medical Chemistry Department, Medical Research Institute, Alexandria University, Alexandria, Egypt
- 2 Experimental and clinical surgery Department, Medical Research Institute, Alexandria University, Alexandria, Egypt
- 3 Cancer Management and Research Department, Medical Research Institute, Alexandria University, Alexandria, Egypt
*Corresponding authors: Medhat M Anwar, Experimental and clinical surgery Department, Medical Research Institute, Alexandria University, Alexandria, Egypt, Tel.: +20 100 5009269. E-mail: medhatanwar@gmail.com(Anwar MM); Neveen A Hussein, Applied Medical Chemistry Department, Medical Research Institute, Alexandria University, Alexandria, Egypt, Tel.: +20 122 3667426. E-mail: neveen39@yahoo.com(Hussein NA).
Received 15 April 2013 Revised 5 August 2013 Accepted 17 August 2013 Published 25 August 2013
DOI: http://dx.doi.org/10.14312/2052-4994.2013-27
Copyright: ©2013 Hussein NA, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Big endothelin-1 (big ET-1) is the biological precursor of endothelin-1 (ET-1) and it is known as an indicator for the degree of activation of endothelin system. Therefore, this study was designed to investigate the serum big ET-1 levels in newly diagnosed primary breast cancer patients; before surgery and after adjuvant therapy. In addition this study was also aimed at examining the correlation between big ET-1 and CA15.3, clinical and pathological criteria of breast cancer, as well as their status during the follow up period after adjuvant therapy. The study consisted of 40 females with newly diagnosed primary breast cancer treated at the Medical Research Institute hospital and 15 healthy females as a control group. Before surgery, big ET-1 serum levels of all breast cancer patients were significantly higher than those of the control group (p<0.001). However, after both surgery and adjuvant therapy, big ET-1 was significantly decreased compared to its level before surgery (p<0.001). On the other hand, there was lack of significant differences in the levels of CA15.3, neither before surgery nor after the adjuvant therapy in comparison to the control group. Interestingly, during the follow up period, nine patients developed metastasis to different organs and their serum big ET-1 and CA15.3 levels significantly increased compared to the levels before surgery and after the adjuvant therapy (p<0.001, 0.008 and <0.001, 0.008 respectively). Inspite of this observation with this specific group of patients, the use of these markers to predict the development of metastasis during the follow up period cannot be generalized. Furthermore, both of these biochemical parameters showed no correlation to any of the clinicopathological parameters and patients characteristics. Therefore, in conclusion this study found that the testing for serum big ET-1 is more useful than CA15.3 for the diagnosis of breast cancer and future trials will be necessary to establish the importance of big ET-1 as a prognostic marker and to formulate a time-line for its measurement in patients with high risk for developing metastasis.
Keywords: breast cancer; big endothelin-1; CA 15.3; metastasis
IntroductionTop
Breast cancer is the most common cancer affecting women worldwide. Globally, about 1.3 million new cases are diagnosed annually, accounting for 23% of the total cancers cases and 14% of all cancer deaths among females [1, 2].
Both non-genetic and genetic factors are involved in the etiology of breast cancer. Non-genetic factors include exposure to female reproductive hormone (both endogenous and exogenous), increasing age, body mass index, alcohol intake, benign breast cancer disease and physical activity. Genetic factors include mutation in breast cancer susceptibility gene 1and 2 (BRCA1 and 2) [3, 4].
The endothelin system (endothelin axis) is a family of peptides (ET-1, ET-2, ET-3 and ET-4) consisting of 21 amino acids (aa), two G- protein-coupled receptors (ETAR and ETBR) and two proteinases [5, 6]. Endothelin-1 is the principle isoform in humans and produced by endothelin cells, vascular smooth muscle cells, epithelial cells, neutrophils, macrophages and fibroblasts [7] .
Human endothelin derives from a 212 aa precursor, preproendothelin-1, which is intracellularly cleaved by endothelin converting enzyme-1 and 2 (ECE-1 and ECE-2) [8] . Removal of single sequence generates the 195 aa proendothelin-1, which is further processed to release the intermediate 38 aa "big ET-1". ECEs hydrolyse big ET-1 to yield the active 21aa ET-1 [6] . Big ET-1 has a circulating half-life of "23min" compared with only "3.5 min" for ET-1 [9] . So, big ET-1 with its long half-life has been implicated as a more accurate indicator of the activation degree of the endothelin system [10] .
Endothelins have numerous potential roles in tumors including modulating angiogenesis, inducing mitogenesis and invasion of tumors cells and protecting cells from apoptosis [6] . In breast cancer, expression of components of the endothelin system has been associated with the transition from normal tissue to progressively invasive lesions [11] . A large number of serum tumor markers have been proposed for breast cancer, among them, carbohydrate antigen 15.3 (CA 15.3) which is the most commonly used in clinical practice. CA15.3 is well characterized assay in peripheral blood that allows the detection of circulating MUC-1 antigen. MUC-1, which is one of cell surface associated mucin, is aberrantly over expressed in many adenocarcinomas especially breast cancer [12] .
The study was designed to investigate the serum big ET-1 levels in newly diagnosed primary breast cancer patients; before surgery and after adjuvant therapy. In addition this study was also aimed at examining the correlation between big ET-1 and CA15.3, clinical and pathological criteria of breast cancer, as well as their status during the follow up period after adjuvant therapy.
Patients and methodsTop
The present study was carried out on 40 consecutive female patients with newly diagnosed primary breast cancer at different clinical stages, admitted to the Surgical Department and followed up in the Department of Cancer Management and Research at Medical Research Institute, University of Alexandria within the period from 2010 to 2012. Fifteen healthy females were selected as a control group with matched ages.
Selected patients met the following criteria: all have primary invasive breast carcinoma with no clinical manifestation of infection and did not receive any immunomodulatory agent in the previous 3 weeks or a blood transfusion before surgery.
All patients were subjected preoperatively to the following: full medical history taking, detailed clinical examination, mammography and ultrasonography of the breast, plain X-ray of the chest, abdominal ultrasound, routine laboratory investigations, fine needle aspiration and/or core cutting needle biopsy from the breast to detect malignancy. Other radiological investigations were done during the follow up period including: CT of abdomen, chest, brain, together with isotopic bone scan for detection of metastasis if indicated. Furthermore, all patients were treated by different surgical techniques followed by proper adjuvant therapy protocols based on tumor stage and risk factors.
During the follow up period (1 year after finishing the adjuvant protocol therapy), 9 patients developed metastasis (22.5%). Therefore, breast cancer patients were re-divided into non-metastatic group (n=31) and metastatic group (n=9).
Blood samples
Two blood samples (5ml) were withdrawn from all breast cancer patients; the first sample was taken one day before surgery while the second sample was taken immediately after the completion of the adjuvant therapy protocol. In the follow up period, an additional blood sample was withdrawn for patients who developed metastasis before the initiation of a new adjuvant therapy protocol, while one blood sample was taken from the control group.
All blood samples were collected into sterile tubes, left to clot then centrifuged at 25°c for 10 minutes. The serum supernatant was stored at -80c until used for determination of big ET-1 and CA15.3.
Assay of biochemical parameters
Big ET-1 levels were measured with an enzyme-linked immunosorbent assay, according to manufacturer instructions (Biomedica Group Gmbh, Vienna, Austria) [13] . CA15.3 levels were measured with immunoradiometric assay (DIAsource Immuno Assay S.A. Belgium) [14] .
Quality control
All individuals agreed to participate after explaining to them the objectives of the study and signed an informed consent. Moreover, the study was approved by the local Institutional Ethical Committee.
Statistical analysis
Data management and analysis were performed using the Predictive Analytics Software (PASW Statistics version 18) and represented as mean ± SE.
For abnormally distributed data, Mann-Whitney test was used to analyze two independent populations and Wilcoxon signed ranks test was used to compare the different periods. Correlations between the biochemical parameters and the studied quantitative variables were assessed using Spearman coefficient.
Agreement of the different predictives with the outcome was used and expressed in sensitivity (which is defined as the proportion of people with the disease who have a positive result {a/ (a + c)}) and specificity (which is defined as a proportion of the people without the disease who have a negative result {d/ (b + d)}), where: a = true positive cases, b = false positive cases, c = false negative cases, and d = true negative cases [15] . The ROC curve analysis was used to compare the diagnostic values of big ET-1 and CA15.3 depending on the area under the ROC curve (AUC). The higher AUC corresponds to a better diagnostic test. A P value of less than 0.05 was considered statistically significant.
ResultsTop
The median age of the 40 female patients with breast cancer was 49 years (range: 35-80 years). Table 1 presents the clinicopathological parameters and characteristics of breast cancer patients’ group. Two patients (5%) had T1 tumor (<2 cm), 33 patients (82.5%) had T2 tumor (2-5 cm) and 5 patients (12.5%) had T3 tumor (>5 cm). In respect to pathological stage, 2.5% of patients were in stage I, 72.5% in stage II and 25% in stage III. Positive lymph nodes were observed in 70% of patients. Most of patients were estrogen receptor (ER) and progestrone receptor (PR) positive, while 22.5% of patients were human epidermal growth factor-2 (HER-2) positive.
| Clinicopathological parameters and patients characteristics | Patient group (n=40) | % |
| Menopausal status | ||
| Premenopause | 22 | 55 |
| Postmenopause | 18 | 45 |
| Tumour size (cm) | ||
| <2 (T1) | 2 | 5 |
| 2-5 (T2) | 33 | 82.5 |
| >5 (T3) | 5 | 12.5 |
| Lymph Node status(LN) | ||
| N0 | 12 | 30 |
| N1 (1-3) | 15 | 37.5 |
| (4-9) | 11 | 27.5 |
| ≥10 | 2 | 5 |
| Pathological stage | ||
| I | 1 | 2.5 |
| II | 29 | 72.5 |
| III | 10 | 25 |
| Histological grade | ||
| II | 31 | 77.5 |
| III | 9 | 22.5 |
| Pathology type | ||
| Invasive ductal carcinoma | 37 | 3 |
| Invasive lobular carcinoma | 92.5 | 7.5 |
| ER status | ||
| negative | 2 | 5 |
| positive | 34 | 85 |
| unknown | 4 | 10 |
| PR status | ||
| negative | 3 | 7.5 |
| positive | 33 | 82.5 |
| unknown | 4 | 10 |
| HER-2 status | ||
| negative | 10 | 25 |
| positive | 9 | 22.5 |
| unknown | 21 | 52.5 |
| Vascular invasion | ||
| negative | 21 | 52.5 |
| positive | 19 | 47.5 |
| Treatment→surgical | ||
| Mastectomy | 38 | 95 |
| Conservative surgery | 2 | 5 |
| →adjuvant | ||
| Hormonal therapy | 7 | 17 |
| Chemo + radiotherapy | 7 | 17.5 |
| Chemo + hormonal therapy | 5 | 12.5 |
| Chemo + radio+ hormonal therapy | 21 | 52.5 |
| Metastasis after treatment | ||
| No | 31 | 77.5 |
| Yes | 9 | 22.5 |
Abbreviations: N0: node negative; N1: node positive
Modified radical mastectomy was performed on 95% of the patients. During the follow up period, 9 patients developed metastasis as follows; 3 with bone metastasis, 4 with ovarian metastasis, 1 with brain matastasis and 1 with liver metastasis.
The mean serum big ET-1 levels of all breast cancer patients before surgery were statistically significantly higher than those of the control group (2.96 ± 1.17 and 0.62 ± 0.17 respectively, P<0.001). Following the adjuvant therapy protocol, no statistical significance in big ET-1 levels were observed as compared to the control group (P>0.05), whereas it was statistically significantly decreased as compared with their corresponding values before surgery (0.65 ± 0.05 and 2.96 ± 1.17 respectively, P ≤ 0.05). As regard CA15-3 levels, no significant difference in its mean value was observed between breast cancer patients before surgery, after the adjuvant therapy and the control group (18.72 ± 10.20, 16.3 ± 4.1 and 13.14 ± 4.21 respectively) (Table 2).
| Parameters | Control group (n=15) | Breast cancer patients (n=40) | |
| Before surgery | After the adjuvant therapy | ||
| Big ET-1(fmol/ml) | 0.62 ± 0.17 | 2.96 ± 1.17* | 0.65 ± 0.05♦ |
| CA15.3(U/ml) | 13.14 ± 4.21 | 18.72 ± 10.20 | 16.3 ± 4.1 |
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: *= values as compared to control group using Mann Whitney test; ♦= values as compared to patients group before surgery using Wilcoxon signed ranks test.
The mean serum big ET-1 levels of the metastatic group of breast cancer patients were significantly increased as compared to their corresponding values before surgery, after therapy and to the control group (4.05 ± 0.7, 2.97 ± 0.92, 0.56 ± 0.2 and 0.62 ± 0.17 respectively) (Table 3). Also, the mean serum CA15.3 levels in the same group were significantly increased as compared to their corresponding levels before surgery, after therapy and to the control group (89.32 ± 19.81, 19.07 ± 9.23, 13.81 ± 5.02 and 13.14 ± 4.21 respectively ) (Table 4).
| Big ET-1 (fmol/ml) | Control group (n=15) | Non metastatic patients (n=31) | Metastatic patients (n=9) | |||
| Before surgery | After therapy | Before surgery | After therapy | After metastasis | ||
| Mean ± S.D | 0.62 ± 0.17 | 2.94 ± 1.42 | 0.63 ± 0.14 | 2.97 ±0.92 | 0.56±0.2 | 4.05 ± 0.70 |
| P | <0.001* | 0.677 | <0.001* | 0.008♦ | <0.001* | |
| P | <0.001♦ | 0.02♦ | ||||
| P | <0.001¤ | |||||
| P | 0.008** | |||||
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: p(*)= values as compared to the control group using Mann Whitney test; p (♦)=values as compared to breast cancer patients group before surgery using Wilcoxon signed ranks test; p (¤)= values as compared to the non metastatic patients group after the adjuvant therapy using Mann Whitney test; p (**)= values as compared to metastatic group after the adjuvant therapy using Wilcoxon signed ranks test.
| CA15.3 (U/ml) | Control group (n=15) | Non metastatic patients (n=31) | Metastatic patients (n=9) | |||
| Before surgery | After therapy | Before surgery | After therapy | After metastasis | ||
| Mean ± S.D | 13.14±4.21 | 18.38± 11.16 | 15.95 ± 4.91 | 19.07± 9.23 | 13.81±5.02 | 89.32 ± 19.81 |
| P | 0.094 | 0.061 | 0.128 | 0.208 | <0.001* | |
| P | 0.563 | 0.008♦ | ||||
| P | <0.001¤ | |||||
| P | 0.008** | |||||
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: P (*)= refers to comparison between CA15.3 measured after metastasis to control group using Mann Whitney test; P (♦)= refers to comparison between CA15.3 measured after metastasis and to breast cancer patients group before surgery using Wilcoxon signed ranks test; P (¤)= refers to comparison between CA15.3 measured after metastasis and to the non metastatic patients group after the adjuvant therapy using Mann Whitney test; P(**)= refers to comparison between CA15.3 measured after metastasis and after the adjuvant therapy using Wilcoxon signed ranks test.
There was no statistically significant difference in the serum big ET-1 and CA15.3 levels of all cancer breast patients before surgery with any of the clinicopathological parameters and patient characteristics (Table 5).
| Clinicopathological parameters and patient characteristics |
Biochemical parameters | |
| Big ET-1 (fmol/ml) | CA15.3 (U/ml) | |
| Age | ||
| R | 0.113 | -0.068 |
| P | 0.487 | 0.675 |
| Tumor size | ||
| R | -0.231 | 0.06 |
| P | 0.151 | 0.713 |
| Lymph node | ||
| R | -0.035 | 0.005 |
| P | 0.83 | 0.994 |
| Pathological stage | ||
| R | 0.065 | 0.157 |
| P | 0.691 | 0.335 |
| Histological grade | ||
| R | -0.202 | -0.161 |
| P | 0.211 | 0.322 |
| Pathology type | ||
| R | 0.062 | 0.078 |
| P | 0.705 | 0.632 |
| ER | ||
| R | -0.088 | -0.107 |
| P | 0.609 | 0.534 |
| PR | ||
| R | -0.014 | -0.125 |
| P | 0.935 | 0.466 |
| HER-2 | R | 0.109 | -0.218 |
| P | 0.656 | 0.369 |
| Vascular invasion | ||
| R | -0.076 | 0.178 |
| P | 0.642 | 0.272 |
| Menopausal status | R | -0.142 | -0.041 |
| P | 0.348 | 0.8 |
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: r= Spearman coefficient
Before surgery, serum big ET-1 showed statistically significant AUC (100%, P<0.001) with sensitivity (80%) and specificity (100%) at a cut-off (2.0 fmol/ml), while serum CA15.3 showed insignificant AUC (0.663, P=0.065) (Figure 1 and Table 6).
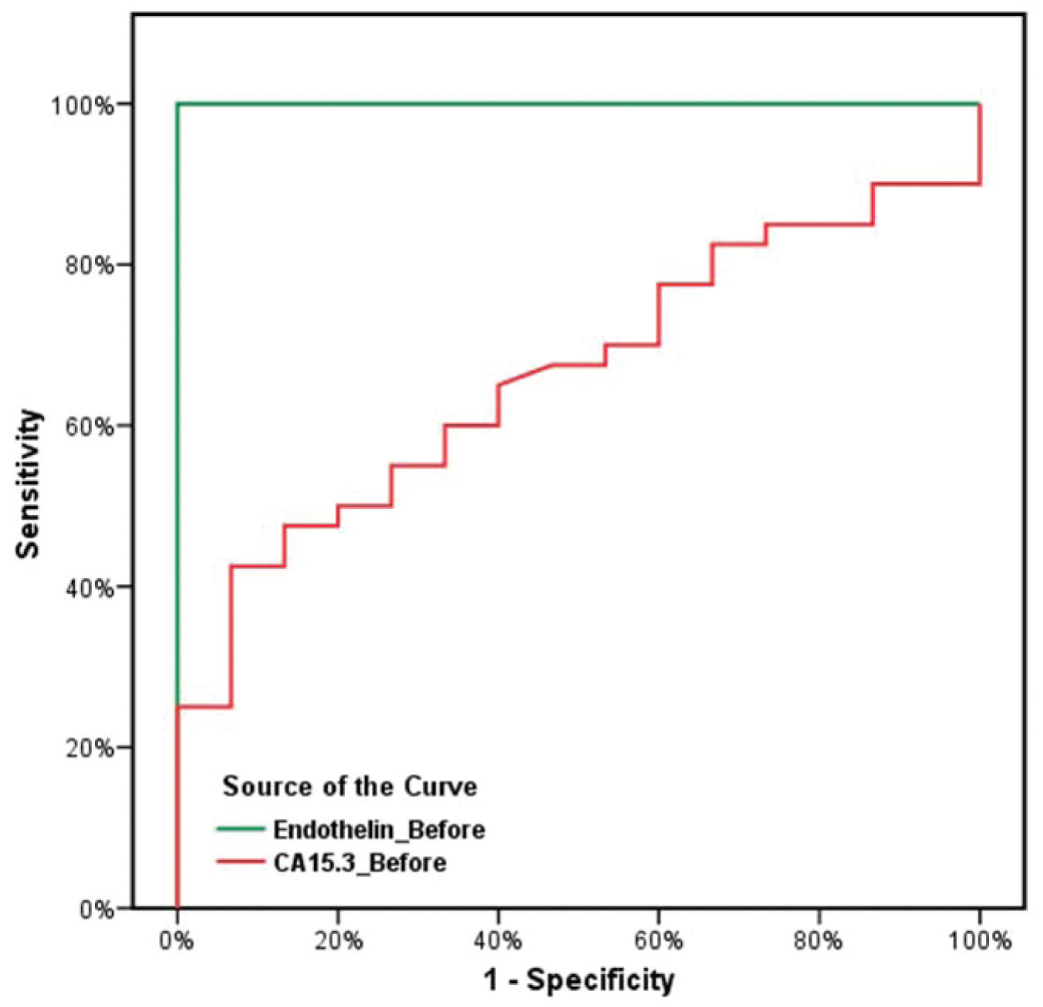
Figure 1 ROC curves for determination diagnostic accuracy of serum big ET-1 and CA 15.3 to identify breast cancer patients before surgery as compared to the control group.
| Parameters | Cut-off value | Control | Breast cancer patients | Sn ( %) | Sp ( %) | AUC (% | (p-value) |
| Big ET-1 (fmol/ml) | < 2.0 | 15 | 8 | 80 | 100 | 100 | <0.001* |
| ≥ 2.0 | 0 | 32 | |||||
| CA15.3 (U/ml) | < 30 | 15 | 35 | 12.5 | 100 | 66.3 | 0.065 |
| ≥30 | 0 | 5 |
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: Sn= Sensitivity; Sp= Specificity; AUC= Area under the curve.
Furthermore, the ROC curve analysis was used to evaluate the prognostic performance of both markers before surgery to predict metastasis. Unfortunately, it revealed that both of them performed poorly in identifying patients who developed metastasis from those who did not (AUC=0.588, p=0.427) and (AUC=0.538, p=0.734) respectively (Figure 2 and Table 7).
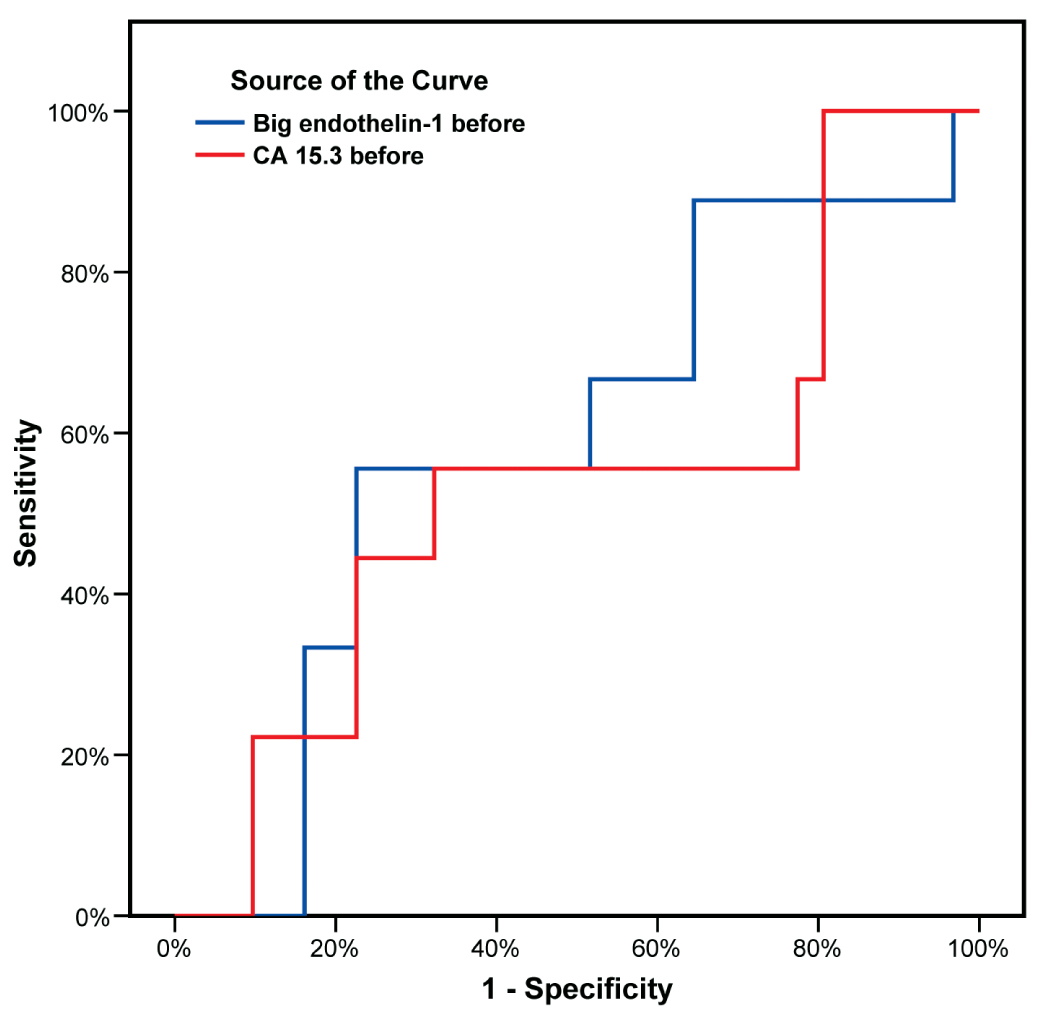
Figure 2 ROC curves for determination of prognostic accuracy of big ET-1 and CA15.3 to identify patients with metastasis from patients without before surgery.
| Parameters | Cut-off value | Sn (%) | Sp (%) | AUC | (p-value) |
| Big ET-1b (fmol/ml) | 2 | 88.89 | 22.58 | 0.588 | 0.427 |
| CA15.3 (U/ml) | 30 | 22.22 | 90.32 | 0.538 | 0.734 |
Differences were considered statistically significant at P ≤ 0.05.
Abbreviations: Sn= Sensitivity; Sp= Specificity; AUC= Area under the curve.
DiscussionTop
Current management of breast cancer focused on early diagnosis and individual care. This was mainly due to improvements in imaging methods, the discovery of novel biomarkers, and the development of tailored therapies. The discovery of specific biomarkers of predictive, prognostic, and diagnostic value over the past few decades had significantly contributed to this task [16] . With the objective of improving breast cancer control, the researchers investigated serum big ET-1 levels in newly diagnosed patients with breast cancer, in addition to big ET-1 correlation with CA15.3, clinical and pathological criteria of breast cancer patients, as well as the outcome of the disease.
The progression of breast cancer depended on the development of vascularization. In addition to the vascular endothelial growth factor pathway, the ET pathway played a critical role in such development [17] . The biological precursor big ET-1 had a much longer half-life than ET-1 and was mainly cleared through the kidney and liver [9, 18]. It had been demonstrated that human breast cancer cells express the ECE, which converted big ET-1 to ET-1 [19] . Therefore, big ET-1 could be considered a candidate marker for the investigation of the ET-1 secretory activity [9] .
This study revealed that serum levels of big ET-1 were significantly higher in all breast cancer patients before surgery than in the control group. This could be due to the deregulation of ECE expression or activity affecting the balance between the precursor molecule and its active form [20] or simply due to the presence of the tumor [21] . Serum levels of big ET-1 in all patients significantly decreased after finishing the adjuvant therapy protocol recording normal levels. This could be attributed to the removal of the tumor as well as the effect of the adjuvant therapy itself. These findings are consistent with other researches [21–24].
The serum level of CA15.3 showed no significant difference between breast cancer patients and control group, neither before surgery nor after the adjuvant therapy. This emphasizes the idea that CA15.3 is not suitable for the diagnosis of early or localized form of breast cancer [25, 26].
The main utility for CA15.3 was for monitoring therapy in patients with metastatic breast cancer [27] . In this research, patients who developed metastasis during the follow up period their CA15.3 levels were significantly increased as compared to their corresponding levels; before surgery, after the adjuvant therapy and also to the control group. These findings were consistence with other researcher results who found that circulating levels of CA15.3 were higher in breast cancer patients with progression or metastatic disease [25] . In another study, CA15.3 levels were increased in only 3% of patients with localized cancer while it was increased up to 70% of patients with metastatic disease [28] .This might be due to the over expression of the MUC-1 gene which encodes CA15.3 [29] or simply related to tumor burden [12] .
No third sample was taken at the end of follow up period for the non-metastatic group of patients for measuring serum levels of big ET-1 and CA15.3. Therefore, further research is needed to explore the change of these biochemical markers among that group of patients after therapy.
The results of this study did not show any significant correlation between the two markers and age, tumor size, pathological grade, lymph node status, hormone receptor status, and menopausal status. This observation was in accordance with other studies [16, 21] that stated that there was no correlation between circulating big ET-1 and tumor size, thus the increase in big ET-1 was due to a difference in production and secretion rate of tumors. Besides, the lack of a correlation between CA15.3 and the clinicopathological characteristics was due to cytokines which depend on the neoplasm activity and not on the stage of the disease or the histological type of the tumor [25, 30–32].
The use of AUC confirmed the validity of ET-1 in the diagnosis of breast cancer. The sensitivity and specificity of big ET-1 at the estimated cut-off value of 2 fmol /ml was 80 and 100% respectively. On the contrary CA15.3 failed to discriminate between the breast cancer and the control groups (AUC=66.3%, p>.05). This finding was in accordance with previous studies which emphasized that CA15.3 lack in sensitivity for the early diagnosis of breast cancer [32, 33]. In the study population both biochemical markers were not able to predict early breast cancer metastasis. However, this might be due to small sample size (9 cases in the metastatic group).
ConclusionTop
This study found that the testing for serum big ET-1 is more useful than CA15.3 for the diagnosis of breast cancer and future trials will be necessary to establish the importance of big ET-1 as a prognostic marker and to formulate a time-line for its measurement in patients with high risk for developing metastasis.
Acknowledgment
Thanks are due to Dr. Shaymaa Essam El-Feky, assistant lecturer of Radiation Science, Department of Radiation Science, Medical Research Institute, Alexandria University, for her sincere help.
Conflict of interest
The authors wish to express that they have no conflict of interest.
ReferencesTop
[1] Jemal A, Bray F, Center MM, Ferlay J, Ward E, et al. (2011) Global cancer statistics. CA Cancer J Clin 61:69–90. Article Pubmed
[2] Panieri E (2012) Breast cancer screening in developing countries. Best Pract Res Clin Obstet Gynaecol 26:283-290. Article Pubmed
[3] Prado A, Andrades P, Parada F (2010) Recent developments in the ability to predict and modify breast cancer risk. J Plast Reconstr Aesthet Surg 63:1581-1587. Article Pubmed
[4] Mavaddat N, Antoniou AC, Easton DF, Garcia-Closas M (2010) Genetic susceptibility to breast cancer. Mol Oncol 4:174-191. Article Pubmed
[5] Levin ER (1995) Endothelins. N Engl J Med 333:356-363. Pubmed
[6] Grimshaw MJ (2007) Endothelins and hypoxia- inducible factor in cancer. Endocr Relat Cancer 14:233-244. Article Pubmed
[7] Nunez DJ, Brown MJ, Davenport AP, Neylon CB, Schofield J, et al. (1990) Endothelin-1 mRNA is widely expressed in porcine and human tissues. J Clin Invest 85:1537-1541. Article Pubmed
[8] Opgenorth TJ, Wu-Wong JR, Shiosaki K (1992) Endothelin-converting enzymes. FASEB J 6:2653-2659. Article Pubmed
[9] Hemsén A, Ahlborg G, Ottosson-Seeberger A, Lundberg JM (1995) Metabolism of Big endothelin-1 (1-38) and (22-38) in the human circulation in relation to production of endothelin-1 (1-21). Regul Pept 55:287-297. Article Pubmed
[10] Hossain E, Islam K, Yeasmin F, Karim MD, Rahman M, et al. (2012) Elevated levels of plasma big endothelin-1 and its relation to hypertension and skin lesions in individuals exposed to arsenic. Toxicol Appl Pharmacol 259:187-194. Article Pubmed
[11] BagnatoA, Spinella F, Rosano L (2008) The endothelin axis in cancer:the promise and the challenges of molecularly targeted therapy. Can J Physiol Pharmacol 86:473-484. Article Pubmed
[12] Park BW, Oh JW, Kim JH, Park SH, Kim KS, et al. (2008) Preoperative CA15.3 and CEA serum levels as predictor for breast cancer outcome. Ann Oncol 19:675-681. Article Pubmed
[13] Arun C, Swift B, Porter KE, West KP, London NJ, et al. (2002) The role of big endothelin-1 in colorectal cancer. Int J Biol Markers 17:268-274. Article Pubmed
[14] Seregni E, Coli A, Mazzucca N, Italian Group RIA-IRMA Test, Italian Association of Nuclear Medicine (2004) Circulating tumour markers in breast cancer. Eur J Nucl Med Mol Imaging 31:15-22. Article Pubmed
[15] Akobeng AK (2007) Understanding diagnostic tests 1: sensitivity, specificity and predictive values. Acta Paediatr 96:338-341. Article Pubmed
[16] Kalles V, Zografos GC, Provatopoulou X, Kalogera E, Liakou P, et al. (2012) Circulating levels of endothelin-1 (ET-1) and its precursor (Big ET-1) in breast cancer early diagnosis. Tumour Biol 33:1231-1236. Article Pubmed
[17] Akhtari M, Mansuri J, Newman KA, Guise TM, Seth P (2008) Biology of breast cancer bone metastasis. Cancer Biol The 7:3–9. Article Pubmed
[18] Nelson JB, Opgenorth TJ, Fleisher LA, Frank SM (1999) Perioperative plasma endothelin-1 and big endothelin-1 concentrations in elderly patients undergoing major surgical procedures. Anesth Analg 88:898-903. Article Pubmed
[19] Patel KV, Schrey MP (1995) Human breast cancer cells contain a phosphoramidon-sensitive metalloproteinase which can process exogenous big endothelin-1 to endothelin-1: a proposed mitogen for human breast fibroblasts. Br J Cancer 71:442– 447. Article Pubmed
[20] Smollich M, Götte M, Yip GW, Yong ES, Kersting C, et al. (2007) On the role of endothelin-converting enzyme-1 (ECE-1) and neprilysin in human breast cancer. Breast Cancer Res Treat 106:361–369. Article Pubmed
[21] Yildirim Y, Gunel N, Coskun U, Sancak B, Bukan N, et al. (2008) Serum big endothelin-1 levels in female patients with breast cancer. Int Immunopharmacol 8:1119–1123. Article Pubmed
[22] Ferrari-Bravo A, Franciosi C, Lissoni P, Fumagalli L, Uggeri F (2000) Effects of oncological surgery on endothelin-1 secretion in patients with operable gastric cancer. Int J Biol Markers 15:56-57. Pubmed
[23] Teng XJ, Shen ZX, Xiang JJ, Shen L, Yuan L, et al. (2006) Pre- and post-operative plasma big endothelin-1 levels in patients with gastric carcinoma undergoing radical gastrectomy. Anticancer Res 26:2503–2508. Article Pubmed
[24] Wülfing P, Diallo R, Kersting C, Wülfing C, Poremba C, et al. (2003) Expression of endothelin-1, endothelin-A, and endothelin-B receptor in human breast cancer and correlation with long-term follow-up. Clin Cancer Res 9:4125–4131. Article Pubmed
[25] Quaranta M, Daniele A, Coviello M, Venneri MT, Abbate I, et al. (2007) MMP-2, MMP-9, VEGF and CA 15.3 in breast cancer. Anticancer Res 27:3593-3600. Article Pubmed
[26] El Agouza IM, Eissa SS, El Houseini MM, El-Nashar DE, Abd El Hameed OM ( 2011) Taurine: a novel tumor marker for enhanced detection of breast cancer among female patients. Angiogenesis 14:321–330. Article Pubmed
[27] Duffy MJ, Evoy D, McDermott EW (2010) CA 15-3: uses and limitation as a biomarker for breast cancer. Clin Chim Acta 411:1869–1874. Article Pubmed
[28] Duffy MJ, Duggan C, Keane R, Hill AD, McDermott E, et al. (2004) High preoperative CA 15-3 concentrations predict adverse outcome in node-negative and node-positive breast cancer: study of 600 patients with histologically confirmed breast cancer. Clin Chem 50:559-563. Article Pubmed
[29] Jamall S, Ishaq M, Alam JM, Khadim M (2010) CA 15-3 and lipid profile in preoperative breast cancer patients. Pak J Biochem Mol Biol 43:114-118. Article
[30] Martín A, Corte MD, Alvarez AM, Rodriguez JC, Andicoechea A, et al. (2006) Prognostic value of pre-operative serum CA 15.3 levels in breast cancer. Anticancer Res 26:3965-3972. Article Pubmed
[31] Agyei Frempong MT, Darko E, Addai BW (2008) The use of carbohydrate antigen (CA) 15-3 as a tumor marker in detecting breast cancer. Pak J Biol Sci 11:1945-1948. Pubmed
[32] Zheng Y, Chen Y, Hu M, Lin Y, Chen Y (2012) Correlation of Preoperative Serum Tumor Markers with Clinicopathological Features and Prognosis in Breast Cancer. Cancer Clin Oncol 1:124-32. Article
[33] Keyhani M, Nasizadeh S, Dehghannejad A (2005) Serum CA15-3 measurement in breast cancer patients before and after mastectomy. Arch Iranian Med 8:263-6. Article



