Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 3, March 2014, Pages 48–53
Original researchOpen Access
Antitumor activity of Papua’s Myrmecodia pendans in human oral tongue squamous cell carcinoma cell line through induction of cyclin-dependent kinase inhibitor p27Kip1 and suppression of cyclin E
- 1 Departmen of Oral Medicine, Faculty of Dentistry, Universitas Gadjah Mada, Yogyakarta, Indonesia
*Corresponding author: Dr. Supriatno, DDS, MDSc, PhD, Department of Oral Medicine, Faculty of Dentistry, Universitas Gadjah Mada, Yogyakarta-55281, Indonesia. Tel. & Fax: +62 0274-515307; Email: pridentagama_oncolog@yahoo.com; Supriatno_fkg@ugm.ac.id
Received 16 November 2013 Revised 10 February 2014 Accepted 17 February 2014 Published 24 February 2014
Copyright: ©2014 Supriatno, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Oral tongue squamous cell carcinoma (OTSCC) is one of the most common cancers encountered in Indonesia, due to the prevalent habits of tobacco chewing, alcohol drinking and smoking. Oral tongue cancer is characterized by a high degree of local invasion and a high rate of metastasis to the cervical lymph nodes. Interestingly, treatment options for this cancer are limited. The aim of this study was to examine the antitumor activity of Papua’s Myrmecodia pendans (ant nest plant) in a human oral tongue squamous cell carcinoma cell line (B88) and to explore the possible mechanism in it. In the present study, B88 cells were treated with various concentration of ethanol extract of Papua’s M. pendans. The results revealed that B88 cells treated with Papua’s M. pendans were remarkable suppressed in cell growth and cell invasion, and had a significant induction of apoptosis characterized by an increase in activation of caspase-3 and -9. Furthermore, up-regulation of p27Kip1 and down-regulation of cyclin E protein was detected in B88 cells treated with Papua’s M. pendans. These results indicated that Papua’s M. pendans exhibited a high potential antitumor activity in human oral tongue squamous cell carcinoma through induction of p27Kip1 and suppression of cycline E.
Keywords: oral tongue cancer; Papua’s Myrmecodia pendans; p27Kip1; cyclin E; ethanol extract
IntroductionTop
Cell proliferation is controlled via an intricate network of extracellular and intracellular signaling pathways which process both negative and positive growth signals. The ultimate recipients of many of these signals are cyclin-dependent kinases (CDKs), a family of enzymes which catalyze events required for individual cell cycle transitions. CDKs require association with cyclins for activation, and the timing of CDK activation is dependent largely upon the timing of cyclin expression [1]. The activity of cyclins-CDKs complexes is affected by two groups of CDK inhibitors (CKIs) which have an inhibitory effect on cell cycle progression. The INK4 group including p15INK4B, p16INK4A, p18INK4C and p19INK4D forms complexes with CDK4 and/or CDK6 and cyclin D types, whereas the CIP/KIP group including p21WAF1, p27Kip1, and p57Kip2 has a more unselective inhibitory effect on several cyclin-CDK complexes. Both families have a conserved amino terminal domain that insufficient for both binding to cyclin/CDK complexes and inhibiting CDK catalytic activity [2]. p27Kip1 is an universal cyclin-dependent kinase inhibitor that directly inhibits the enzymatic activity of cyclin-CDK complexes, resulting in cell cycle arrest at G1 [3]. The p27Kip1 is an important prognostic factor in various types of malignancies. Recently, decreased expression of p27Kip1 has been frequently detected in human cancers [4, 5]. In addition, loss of p27Kip1 has been associated with disease progression and an unfavorable outcome in several types of malignancies [5]. It was reported that transfection of the p27Kip1 gene to human oral cancer cells leads to inhibition of cells proliferation, invasion and metastasis, and also induction of apoptosis [6, 7]. A low level of p27Kip1 expression was reported to be associated with poor prognosis and high malignancy in human colon [5], breast [8], gastric [9], lung [10], prostate [11], ovarian [12], thyroid [13] and lymphatic [14] cancers. Disruption of the cell cycle regulatory activity of p27Kip1 is related to the aggressiveness of cancer cells [1].
Oral tongue cancer is characterized by a high degree of local invasion and a high rate of metastasis to the cervical lymph nodes. This cancer frequently shows local recurrence after initial treatment, probably due to microinvasion and/or micrometastasis of tumor cells at the primary site [15]. Despite advances in surgery, radiotherapy and chemotherapy, the survival rate of patients with oral tongue cancer has not been significantly improved over the past several decades. Treatment options for recurrent or refractory oral cancers are limited. Also, the prognosis has not changed during the past 10 years [16]. However, as a new strategy for refractory cancer, herbal medicine has attracted keen interest.
M. pendans (ant nest plant) is a plant that is attached to other plants (epiphyte) and thrived in the highlands of Wamena, Papua, Indonesia. Originally, M. pendans was introduced in the inland of Papua, and commonly used as a medicine by local residents. M. pendans was used by the people of Papua since 1950s for a mixture porridge and boiled drink to enhance the body immunity [17]. Interestingly, M. pendans was used as medicine for various diseases. Efficacy of M. pendans was showed by the content of active substances include polyphenols, flavonoids, tannins and glycosides [18]. M. pendans has been used to treat various diseases traditionally and safely without any side effects by some tribes in Wamena inland for a long time. It was reported that M. pendans can inhibit various types of human cancers including brain, breast, lung, colon, liver, uterus, prostate, skins, cervical and blood cancers [17, 18]. The fifty-inhibitory concentration (IC50) of ethanol extract of M. pendans was reported at 33.28 µg/ml in cervix cancer cell line (HeLa cell), and 353.18 µg/ml in breast cancer cell line (MCF-7 cell) [18, 19]. In addition, M. pendans can treat a variety of systemic diseases such as leukemia, heart diseases, tuberculosis, kidney and prostate, various allergies, migraine, rheumatism, hemorrhoid, and infection diseases [20]. Although M. pendans can inhibit various types of human cancers, its effect on oral cancers cells and the mechanism pathways are still unclear.
In the present study, the antitumor activity of M. pendans in a human oral tongue squamous cell carcinoma (B88) cell line and the possible mechanisms were examined.
Materials and methodsTop
Cell and cell culture
B88 cells [21] were maintained in Dulbecco's modified Eagle's medium (DMEM; Sigma, St. Louis, MO, USA) supplemented with 10% fetal calf serum (FCS; Moregate BioTech, Bulimba, Australia), 100 µg/ml streptomycin, and 100 U/ml penicillin (Invitrogen Corp., Carlsbad, CA, USA).
Preparation of crude ethanol extract
Fresh M. pendans plants were collected from Wamena inland, Papua, Indonesia. Plants were cut into small pieces and dried inside the incubator at 55oC. Five hundred gram of pieces were soaked in 500 ml of 70% ethanol, and put on shaker at 150 rpm for 3 days at ambient temperature. The mixtures were then filtered. The filtrates were evaporated using vacuum rotary evaporator (BÜCHI Rotavapor R-200/205, Model R205V800). Stock solutions of crude ethanol extracts were prepared by diluting the extracts with suspending agent (200 µl of dimethyl sulfoxide) followed with aquabidest solution to obtain a final concentration of 100 mg/ml.
Suppression of cell growth (MTT assay)
B88 cells were seeded on 96-well plates (Falcon, NJ, USA) at 2 x 104 cells per well in DMEM containing 10% FCS. After 24 h, the culture medium was replaced with new medium containing various concentrations (0, 50, 150, 300, 450 and 600 µg/ml) of ethanol extract of M. pendans. After 24 and 48 h, 25 µl fresh 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide [MTT, 5 mg/ml phosphate-buffered saline (PBS)] (Sigma-Aldrich, USA) was added to each well. After 4 h of incubation, the culture media were discarded, 100 µl of dimethyl sulfoxide (DMSO: Nacalai tesque, Inc., Kyoto, Japan) was added to each well and vibrated to dissolve the MTT formazan. The optical density was measured at 540 nm with a BioRad microplate reader (BioRad Laboratories, Hercules, CA, U.S.A.). Each determination was performed in quadruplicate.
Analysis of chemotactic ability of M. pendans
Chemotaxis (directed migration) was evaluated in the Boyden chamber apparatus (Neuro Probe, Inc., Cabin John, MD, USA). Briefly, subconfluent cells were starved for 24 h and harvested with 0.05% (w/v) trypsin (Invitrogen Corporation, USA) containing 0.02% (w/v) ethylenediamine tetra-acetic acid (EDTA, Invitrogen Corporation), washed twice with PBS, and resuspended to a final concentration of 5 x 105 per ml in serum-free medium with 0.1% (w/v) fraction V bovine serum albumin (BSA, Wako Pure Chemical Industries, Ltd). Polyvinylpyrrolidone (PVP) filters (Nuclepore Corp, Palo Alto, CA, USA) of 8-µm pore size were precoated with gelatin (Merck KGaA, Frankfurt, Darmstadt, Germany) (0.1 mg/ml) and rinsed in sterile water. Bottom wells of the chamber were filled with 30 µl of 10% FCS in DMEM containing various concentration of ethanol extract of M. pendans and covered with a gelatin-coated membrane [membrane was incubated in gelatin (0.1 mg/ml) solution for 30 min at 37oC. Then, 50 µl of cell suspension, yielding 500 cells/ml, was added to the top wells. After 24 h of incubation, the membranes were stained with Giemsa solution (Ted Pella Inc., Redding, CA, USA). Cells on the upper surface of the filter were carefully removed with a cotton swab, and the number of cells that had penetrated through the filter was counted under light microscope at 400x magnification. The counting was performed for 12 fields in each concentration. Each determination was carried-out in triplicate.
Western blot analysis
Cell lysates were prepared from the B88-treated cells in Falcon tissue culture for 48 h. Briefly, samples containing equal amounts of protein (50 µg) were electrophoresis on a SDS-polyacryl-amide gel and transferred to a polyvynilidene fluoride (PVDF) membrane (BioRad, Hercules, CA, USA). The filters were blocked in TBS containing 5% non-fat milk powder at 37oC for 1 h, and then incubated with a 1:500 dilution of primary antibodies against the p27 protein (clone 1B4, mouse monoclonal antibody; Novocastra Laboratories, New Castle, UK) and cyclin E protein (clone HE12, mouse monoclonal antibody, Santa Cruz Biotech, USA). For detection of HRP-conjugated antibodies were used the enhanced chemiluminescent (ECL) plus kit (Amersham Pharmacia Biotech, UK). Anti α-tubulin monoclonal antibody (Zymed laboratories, San Francisco, USA) was used for normalization of the western blot analysis.
Induction of apoptosis using colorimetric assay
Induction of apoptosis was measured using the colorimetric assay kit (caspase-3 and -9; BioVision Research Product, CA, USA) according to the manufacturer’s directions. Briefly, equal amounts of cell extracts prepared from B88-treated cells for 48 h were incubated with the substrate (DVED-pNA or LEHD-pNA) in the assay buffer for 2 h at 37oC. Absorbance was measured at 405 nm using a microplate reader (BioRad, USA). Each determination was performed in triplicate.
Statistical analysis
Statistical differences between the means in the different groups were evaluated with Stat View 4.5 (version 5.0J, SAS Institute Inc, Cary, NC, USA) using one-way ANOVA and a post hoc t-test. The significance level was set at 5% for each analysis.
ResultsTop
Growth of cells in vitro
B88 cells treated with various concentrations of ethanol extract of M. pendans were examined by the MTT assay. Relative cell number was evaluated by comparing the absorbance in each group. No significant differences in cell number were observed in concentration of 50 and 150 µg/ml groups in 24 h, but in 48 h only in concentration of 50 µg/ml group compared with non-treated group. However, the cell growth of B88 cells treated with concentration 300 µg/ml to 600 µg/ml of M. pendans were significantly suppressed in 24 and 48 h as compared to non-treated B88 cells (P<0.05) (Figure1). IC50 of ethanol extract of M. pendans in tongue cancer cell line (B88 cell) was detected at 471.79 µg/ml.
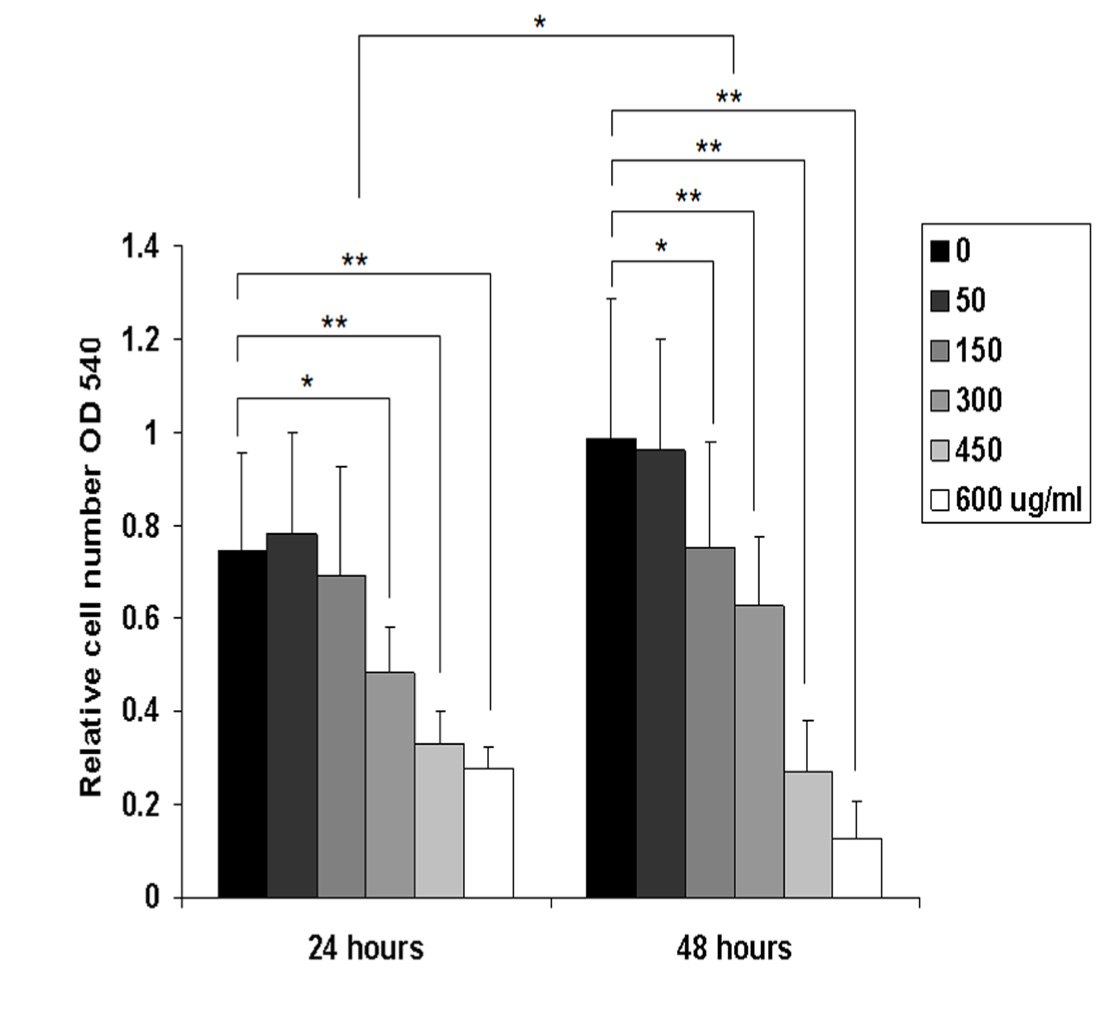
Figure 1 Relative cell number was evaluated by comparing the absorbance in different groups in 24 and 48 h using MTT assay. The cell growth of B88 cells treated with M. pendans was significantly suppressed in 24 and 48 h as compared to non-treated B88 cells. Values shown are the mean of six determinations; error bars indicate standard deviations. *, P<0.05 and **, P<0.01 when compared with that of control cells by one-way analysis of variance.
Suppression of chemotactic ability of M. pendans
Cell migration is an essential process involved in tumor invasion and metastasis. The chemotactic ability of each various extract concentration of M. pendans to B88 cells with Boyden chamber kit was evaluated for 24 h incubation. As seen in Figure 2, B88 cells treated with concentration of 300 µg/ml to 600 µg/ml of M. pendans showed significantly lower ability of chemotactic compared with that of the non-treated B88 cells (P<0.01).
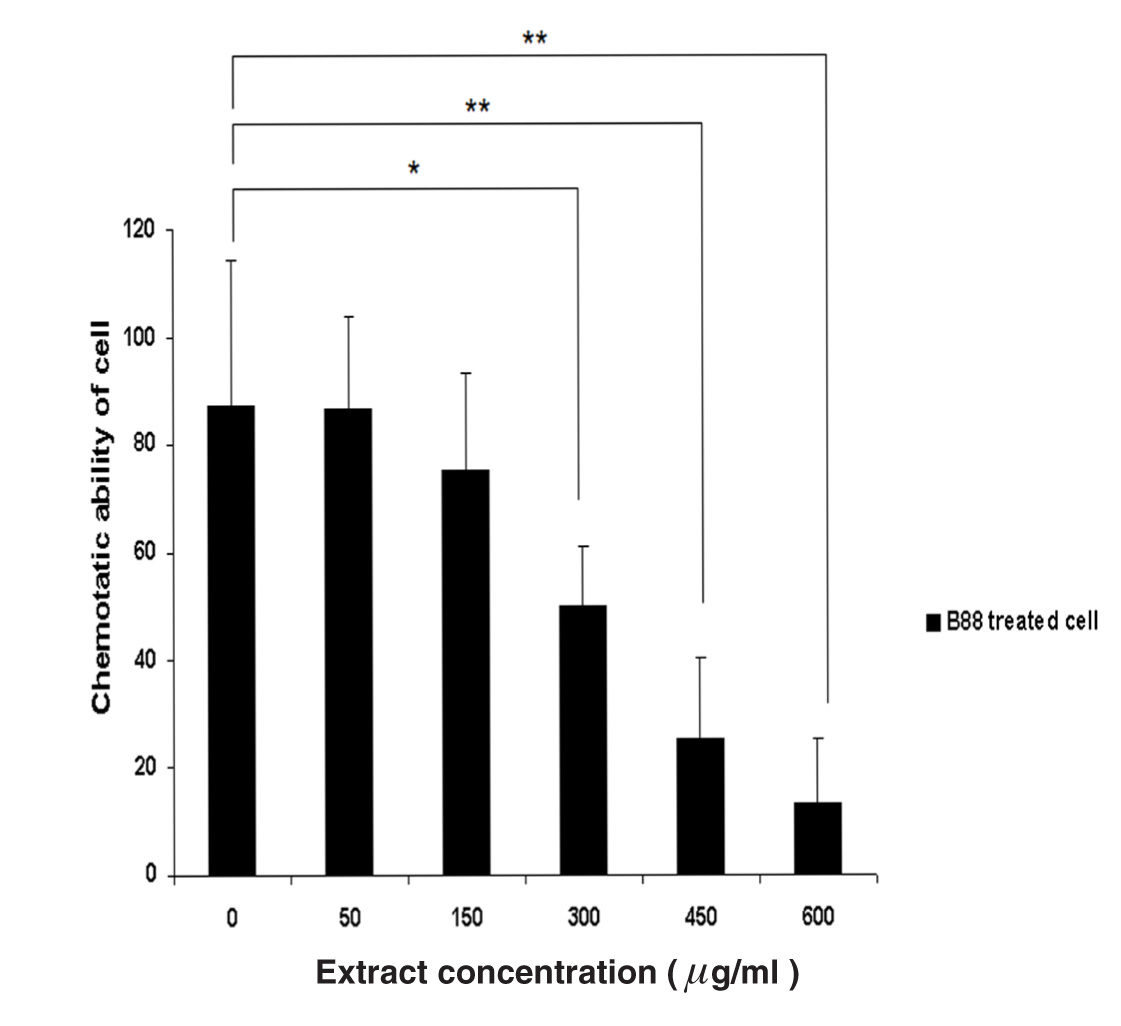
Figure 2 Analysis of chemotactic ability of M. pendans to B88 cells was examined by Boyden chamber assay. B88 cells treated with M. pendans showed significantly lower ability of chemotactic compared with that of the non-treated B88 cells. Values are shown as mean; error bars indicate standard deviation.
Expression of p27Kip1, cyclin E and α-tubulin protein
Western blot analysis was used to examine the protein expression of p27Kip1, cyclin E and α-tubulin in different B88-treated group. As shown in Figure 3, gradual up-regulation of p27Kip1 protein was detected from concentration of 300 µg/ml to 600 µg/ml. However, down-regulation of cyclin E protein was observed in the same concentration. The expression of α-tubulin as an internal control was approximately the same in each B88-treated group.
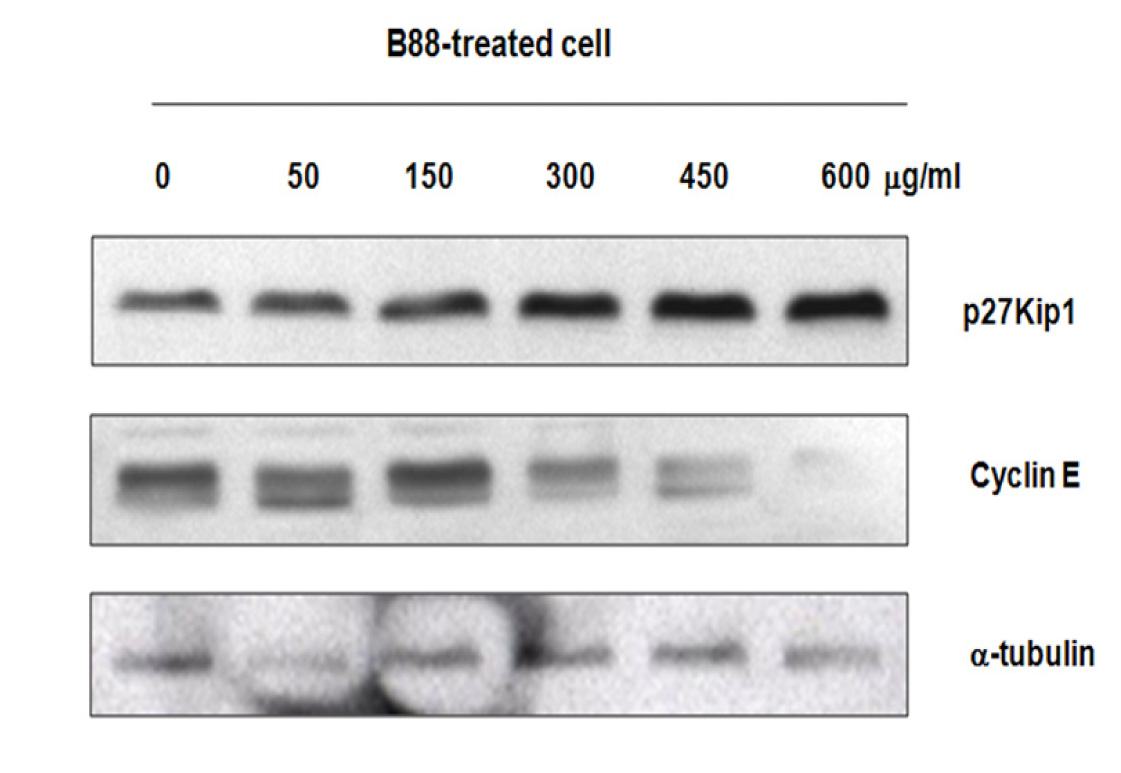
Figure 3 Expression of p27Kip1, cyclin E and α-tubulin protein was determined by western blot. The B88 cell line was treated with various concentration of ethanol extract of M pendans for 48 h.
Activation of caspases-3 and -9
To investigate whether up-regulation of p27Kip1 protein can induce apoptosis, a colorimetric assay was conducted. The activity of caspase-3 and -9 in B88 cells treated with ethanol extract of M. pendans for 48 h was investigated using the colorimetric assay kit according to the manufacturer’s directions. B88-treated cells (at 600 µg/ml group) increased the proteolytic activity following increase of both caspase-3 and -9 (3.70 fold and 5.85 fold, respectively) (Figure 4). These results suggested that M. pendans induced apoptosis through extrinsic and intrinsic pathways.
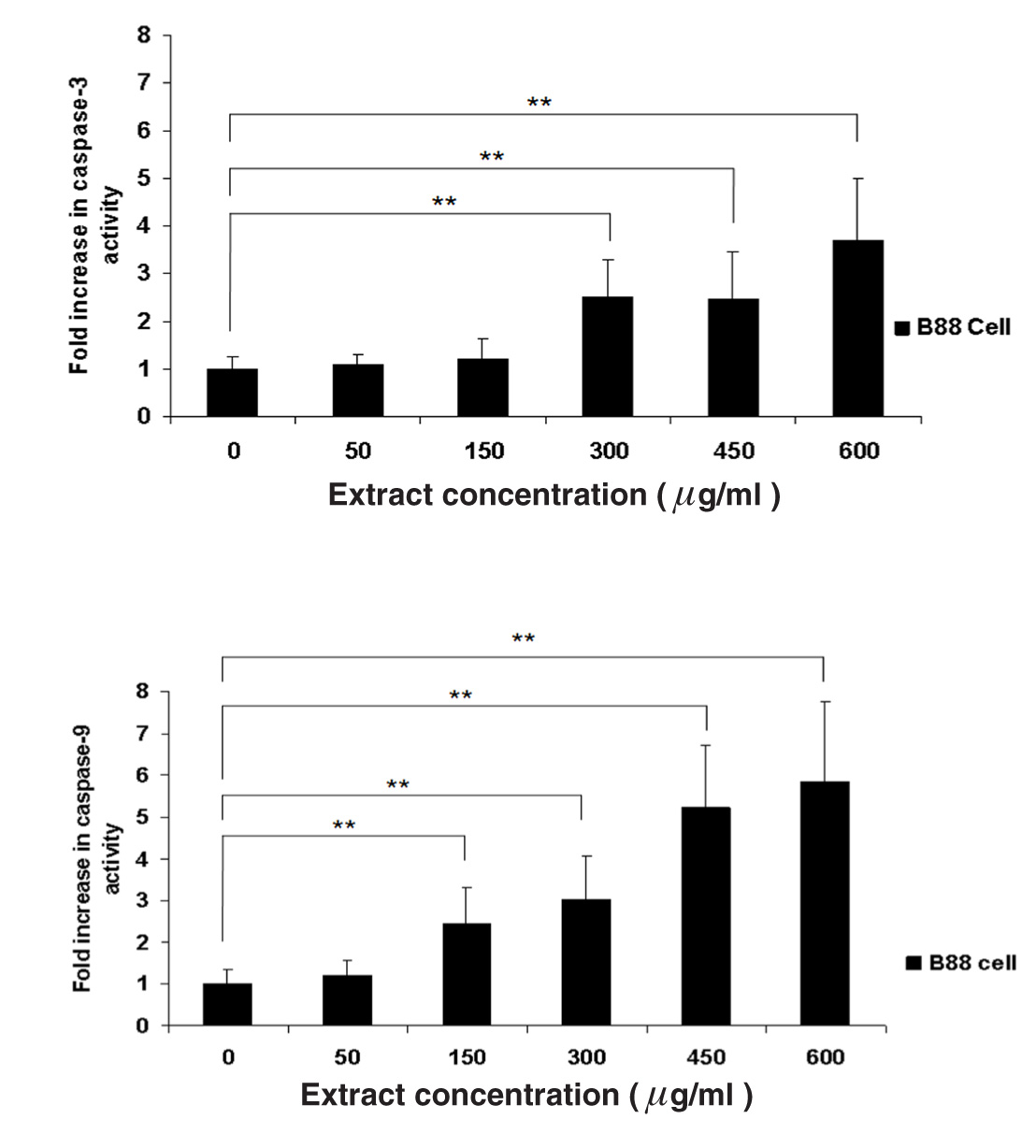
Figure 4 Activity of caspase-3 and -9 in B88-treated cell. Cells were treated with various concentration of ethanol extract of M. pendans. B88-treated cells was increased the proteolytic activity of both caspase-3 and -9. Values are shown as mean; error bars indicate standard deviation.
DiscussionTop
The normal operation of cell cycle requires a delicate balance between positive and negative regulatory factors. Any alteration in this balance can result in abnormal cell proliferation, which may lead to malignant transformation. Loss of normal cell cycle control plays a crucial role in the genesis of most types of cancer. The cell cycle is modulated by the interaction of multiple cell cycle molecules, including cyclins, cyclin-activating kinase (CAK), CDK and cyclin-dependent kinases inhibitors (CDIs). In addition to its role as a CDI, p27Kip1 is a putative tumor suppressor gene [22], a regulator of drug resistance in solid tumors [23], and a promoter of apoptosis [24]. Furthermore, p27Kip1 acts as a safeguard against inflammatory injury [25] and plays a role in cell differentiation [26]. In the present study, expression of p27Kip1 protein was gradually increased in concentrations of M. Pendans from 300 µg/ml to 600 µg/ml. Conversely, down-regulation of cyclin E protein was detected in the same concentration. In addition, the expression of α-tubulin as an internal control was approximately the same in each B88-treated group. Hayashi et al. reported that high cyclin E and low p27Kip1 expression were potential poor prognostic factor in lung adenocarcinoma [27].
Cyclin E overexpression is implicated in carcinogenesis and increase aggressivity in human cancer cell, but in current study, expression of cyclin E was markedly suppressed by M. pendans accompanied by up-regulation of p27Kip1 protein. It was reported that p27Kip1 may induce cell cycle arrest and apoptosis, and suppress the ability of cell growth, invasion and metastasis in an oral cancer cell line [1, 7]. We have already reported that overexpression of p27Kip1 played an important role in retarding the cancer invasion process as well as metastasis mediated by up-regulating E-cadherin and down- regulating tumor invasion and metastasis-1 (Tiam-1) either directly or indirectly [6].
MTT assay was examined and the result indicated that the relative cell number had no significant differences in concentration of 50 and 150 µg/ml groups in 24 h. However, concentration of 300 µg/ml to 600 µg/ml of M. pendans were significantly suppressed the B88 cell growth with inhibited cell growth at 35.2% to 63.1% in 24 h, and 36.7% to 87.3% in 48 h. These data suggest that the ant nest plant of M. pendans strongly suppressed the B88 cell growth (Figure 1). Moreover, the chemotactic ability of M. pendans to B88 cell was evaluated using Boyden chamber apparatus. We found that B88 cells treated with concentrations of 300 µg/ml and 600 µg/ml of M. pendans showed marked lower ability of chemotactic to cells compared with that of the non-treated B88 cell. The inhibition rate of chemotactic ability to cells at 450 and 600 µg/ml group was 71.1% and 84.9%, respectively (Figure 2).
An increase in apoptosis percentages and activation of caspase-3 and -9 in B88-treated cells strongly suggests that apoptosis occurred in those cultures. Similar results were reported by Supriatno et al., who demonstrated that induction of apoptosis can be caused by up-regulation of p27Kip1 in oral cancer [7]. Azuma et al. reported that activation of caspase-3 leads to the cleavage of PARP and DNA fragmentation, indicating that caspase-3 targets cellular proteins for proteolytic cleavage and results in cell death [28]. Caspase-3 can be activated by either an extrinsic apoptosis pathway, through the activation of caspase-8, or an intrinsic apoptosis pathway (via release of cytochrome c from mitochondria). Activated caspase-8 can directly cleave and activate the executioner caspases, such as caspase-3. Alternatively, it can cleave one of the Bcl-2 family members (such as Bid) to induce the release of mitochondrial cytochrome c, which also leads to activation of caspase-3 via formation of apoptosome (consisting of Apaf-1 and caspase-9). In the current study, increased activation of caspase-3 and -9 in B88 cells treated with M. pendans (600 µg/ml) revealed that apoptosis occurred through extrinsic (3.70 fold) and intrinsic pathways (5.85 fold). Interestingly, apoptosis activity through intrinsic pathways was significantly stronger than extrinsic pathway.
ConclusionTop
Ant nest plant of M. pendans exhibited a strong antitumor activity in human oral tongue squamous cell carcinoma (B88 cell) through the induction of p27Kip1 and suppression of cyclin E.
Acknowledgments
The author heartfully thanks and appreciates Prof. Mitsunobu Sato, DDS., Ph.D; Dr. Koji Harada, DDS., Ph.D and Dr. T. Bando, DDS., Ph.D (Second Department of Oral and Maxillofacial Surgery, School of Dentistry, Tokushima University, Japan) for all their encouragement and support. Appreciation and thanks is also due to LPPM UGM through Insentif Jurnal Karya Ilmiah Internasional 2013, and to all the staff members at the Department of Oral Medicine, Faculty of Dentistry, Universitas Gadjah Mada, Yogyakarta, Indonesia.
Conflict of interest
The author wish to express that he has no conflict of interest.
ReferencesTop
[1] Supriatno, Harada K, Yoshida H, Sato M (2005) Basic investigation on the development of molecular targeting therapy against cyclin-dependent kinase inhibitor p27Kip1 in head and neck cancer cells. Int J Oncol 27: 627-635. Article Pubmed
[2] Supriatno, Harada K, Kawaguchi S, Onoue T, Yoshida H, et al. (2004) Characteristics of antitumor activity of mutant type p27Kip1 gene in an oral cancer cell line. Oral Oncol 40: 679-687. Article Pubmed
[3] Harada K, Supriatno, Kawaguchi S, Onoue T, Kawashima Y, et al. (2005) High antitumor activity using intratumoral injection of plasmid DNA with mutant-type p27Kip1 gene following in vivo electroporation. Oncol Rep 13: 201-206. Article Pubmed
[4] Harada K, Supriatno, Yoshida H, Sato M (2002) Low p27Kip1 expression is associated with poor prognosis in oral squamous cell carcinoma. Anticancer Res 22: 2985-2989. Pubmed
[5] Loyer P, Trembley JH, Katona R, Kidd VJ, Lahti JM (2005) Role of CDK/cyclin complexes in transcription and RNA splicing. Cell Signal 17:1033-1051. Article Pubmed
[6] Supriatno, Yuletnawati SE, Widiasto A (2011) Effect of intratumoral injection of mutant type p27Kip1 followed by in vivo electroporation on radiotherapy-resistant human oral tongue cancer xenografts. Mol Med Report 4: 41-46. Article Pubmed
[7] Supriatno, Harada K, Hoque MO, Bando T, Yoshida H, et al. (2002) Overexpression of p27Kip1 induces growth arrest and apoptosis in an oral cancer cell line. Oral Oncol 38:730-736. Article Pubmed
[8] Zhang Q, Tian L, Mansouri A, Korapati AL, Johnson TJ, et al. (2005) Inducible expression of a degradation-resistant form of p27Kip1 causes growth arrest and apoptosis in breast cancer cells. FEBS Lett 579:3932–3940. Article Pubmed
[9] Jimmy BY, Samarasinge K, Raju GC, Moochhala SM, Ti TK (2000) Expression of Cell-Cycle Regulators p27 and Cyclin Correlates with Survival in Gastric Carcinoma Patients. J Surg Res 94: 56-60. Article Pubmed
[10] Zhuang Y, Yin HT, Yin XL, Wang J, Zhang DP (2011) High p27 expression is associated with a better prognosis in East Asian non-small cell lung cancer patients. Clin Chim Acta 412: 2228-2231. Article Pubmed
[11] Li R, Wheeler TM, Dai H, Sayeeduddin M, Scardino PT, et al. (2006) Biological correlate of p27compartmental expression in prostate cancer. J Urol 175:528-532. Article Pubmed
[12] Patah LEM, Camarotto KC, Gonçalves WJ (2004) P27 expression in epithelial ovarian tumors. Int J Gynaecol Obstet 85:179-180. Article Pubmed
[13] Karlidag T, Cobanoglu B, Keles E, Alpay HC, Ozercan I, et al. (2007). Expression of Bax, p53, and p27/kip in patients with papillary thyroid carcinoma with or without cervical nodal metastasis. Am J Otolaryngol 28:31-36. Article Pubmed
[14] Erlanson M, Portin C, Linderholm B, Lindh J, Roos G, et al. (1998) Expression of cyclin E and the cyclin-dependent kinase inhibitor p27 in malignant lymphomas – prognostic implication. Blood 92: 770-777. Article Pubmed
[15] Ammar A, Uchida D, Begum NM (2004) The clinicopatho¬logical significance of the expression of CXCR4 protein in oral squamous cell carcinoma. Int J Oncol 25: 65-71. Article Pubmed
[16] Parkin DM, Pisani P, Ferlay J (1999) Estimates of the worldwide incidence of 25 major cancers in 1990. Int J Cancer 80: 827-841. Article Pubmed
[17] Subroto, MA, Saputro H (2006) Gempur Penyakit dengan Sarang Semut, Penebar Swadaya Press, Depok, Indonesia, p. 15-31.
[18] Fatmawati D, Puspitasari PK, Yusuf I (2011) Cytotoxic Effect of Ethanolic Extract of Sarang Semut (Myrmecodia pendens) on HeLa Cervix Cancer Cell Line. In Vitro Experimental Study. Sains Medika 3: 112-120.
[19] Soeksmanto A, Subroto MA, Wijaya H, Simanjuntak (2010) Anticancer Activity test for Extract of Sarang Semut Plant (Myrmecodya Pendens) to HeLa and MCM-B2 Cell. Pak J Biol Sci 13: 148-151. Pubmed
[20] Hertiani T, Sasmito E, Sumardi, Ulfah M (2010) Preliminary Study on Immunomodulatory Effect of Sarang-Semut Tubers Myrmecodia tuberosa and Myrmecodia pendens. J Bio Sci 10: 136-141. Article
[21] Harada K, Bando T, Yoshida, Sato M (2001) Characteristics of antitumour activity of cepharanthin against a human adenosquamous cell carcinoma cell line. Oral Oncol 37:643-651. Article Pubmed
[22] Nakayama K, Ishida N, Shirane M, Inomata A, Inoue T, et al. (1996) Mice lacking p27Kip1 display increased body size, multiple organ hyperplasia, retinal dysplasia and pituitary tumors. Cell 85:707-720. Article Pubmed
[23] St. Croix B, Florenes VA, Rak JW, Flanagan M, Bhattacharya N, et al. (1996) Impact of the cyclin-dependent kinase inhibitor p27Kip1 on resistance of tumor cells to anticancer agents. Nat Med 2: 1204-1210. Article Pubmed
[24] Katayose Y, Kim M, Rakkar ANS, Li Z, Cowan KH, et al. (1997) Promoting apoptosis: a novel activity associated with the cyclin-dependent kinase inhibitor p27. Cancer Res 57: 5441-5445. Article Pubmed
[25] Ophascharoensuk V, Fero ML, Hughes J, Roberts JM, Shankland SJ (1998) The cyclin-dependent kinase inhibitor p27Kip1 safeguards against inflammatory injury. Nat Med 4: 575-580. Article Pubmed
[26] Durand B, Gao FB, Raff M (1997) Accumulation of the cyclin-dependent kinase inhibitor p27/Kip1 and the timing of oligodendrocyte differentiation. EMBO J 16: 306-317. Article Pubmed
[27] Hayashi H, Ogawa N, Ishiwa N, Yazawa T, Inayama T, et al. (2001) High cyclin E and low p27/Kip1 expressions are potentially poor prognostic factors in lung adenocarcinoma patients. Lung Cancer 34: 59 – 65. Article Pubmed
[28] Azuma M, Harada K, Supriatno, Tamatani T, Motegi K, et al. (2004) Potentiation of induction of apoptosis by sequential treatment with cisplatin followed by 5-fluorouracil in human oral cancer cells. Int J Oncol 24: 1449-1455. Article Pubmed



