Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 4, Issue 2, April 2014, Pages 68–73
Original researchOpen Access
Decitabine is more cost effective than cytarabine and daunorubicin in elderly acute myeloid leukemia patients
- 1 Department of Medicine, Roswell Park Cancer Institute, Buffalo, NY
- 2 State University of New York at Buffalo, Buffalo, NY
*Corresponding author: Meir Wetzler, MD FACP, Professor of Medicine, Chief, Leukemia Section, Roswell Park Cancer Institute, Elm and Carlton Streets, Buffalo, NY 14263. Tel.: 716-845-8447; Email: meir.wetzler@roswellpark.org
Received 10 February 2014 Revised 14 March 2014 Accepted 23 March 2014 Published 30 March 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-9
Copyright: © 2014 Batty N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Introduction: Decitabine is not approved in the United States (US) for acute myeloid leukemia (AML) because it did not improve overall survival compared with standard conventional induction treatment with cytarabine and daunorubicin (AD). We asked what would be the cost effectiveness of decitabine versus AD in AML patients older than 60 years of age. Methods: A semi-Markov model compiling survival and cost data was used based on survival probabilities from the literature. Data accounted for re-induction therapy with idarubicin, fludarabine, cytarabine and granulocyte colony-stimulating factor and consolidation therapy with high-dose cytarabine (HiDAC) but not for stem cell transplantation. The assumption-based model considered a maximum of four cycles of HiDAC and continuing decitabine until loss of benefit. Results: Assuming 1,000 patients for each treatment arm in a semi-Markov model over one year time horizon, the quality-adjusted life year (QALY) for AD vs. decitabine were 0.47 and 0.61. The percentage survival for AD and decitabine were 45.2% and 50.5%. Their costs were $168,863 and $108,084. The incremental cost-effectiveness ratio was -$60,779/0.14 = -$433,756 per QALY. By sensitivity analysis, decitabine was superior to AD in all parameters. Conclusion: Decitabine is a more cost-effective therapy for patients older than 60 years of age than AD. While cost effectiveness is certainly important, decitabine may be arguably considered for elderly newly diagnosed AML patients given the economic pressures in the US health system; however, this is not a criterion for drug approval.
Keywords: acute myeloid leukemia; elderly; cytarabine; daunorubicin and decitabine; cost effectiveness analysis
BackgroundTop
Acute myeloid leukemia (AML) is the most lethal form of leukemia in patients older than 60 years [1-3]. There are currently only 2 methods for treating this population: the standard of care known as (7+3) [4, 5], which consists of cytarabine and daunorubicin (AD), and hypomethylating agents such as decitabine [1, 6-8]. Complete remission rates (CR) are approximately 50% using both approaches [1, 4]. However, the CR rate following decitabine was achieved after a median of three courses and the median overall and disease-free survival were 55 and 46 weeks with 2% 30-day mortality. Similar results were achieved by AD after only one induction course but at the expense of 11-12% 30-day mortality. To date, there has not been a study to examine the cost-effectiveness of these two modalities. Therefore, we aimed to compare the direct and indirect costs of these two regimens in patients older than 60 years undergoing induction therapy for AML by constructing a theoretical model based on a diagnosis-related group (DRG) exempt health care system such as Roswell Park Cancer Institute (RPCI). As per the national comprehensive cancer network guidelines, the approved therapies for patients with AML older than 60 years of age with performance status less than 2 are clinical trials, hypomethylating agents or (7+3). At our cancer center, the most common used hypomethylating agent is decitabine. Therefore, the economic model is adjusted to compare this hypomethylating agent i.e. decitabine with (7+3). Clinical trials are inclusively considered in our model in both arms of our analysis. The patient age remains an important initial consideration when considering this induction therapy as per the national guidelines; however of course further stratification by performance status is nonetheless essential for final determination of this treatment. Of note, other regimen such as CAG (cytarabine, Aclacinomycin, Granulocyte colony-stimulating factor G-CSF) regimen is often used for the treatment of elderly patients AML, especially in some Asian countries; however, our economic analysis is modeled according to the US health system.
In the current economic situation, ensuring cost-effective therapy becomes critical because an effective therapy may or may not be cost-effective in different health care systems. Aside from applying the best available published data for these two regimens, standardized economical parameters were examined to evaluate the ultimate preferred therapy in this setting. Different aspects of monetary expenditure were explored mainly in terms of direct and not indirect costs such as the impact on caregiver resources or a societal perspective.
MethodsTop
Overview and study design
A semi-Markov model was constructed for both approaches compiling survival and cost data [4, 6, 9, 10]. The model accounted for different health states of AML as a disease (Figure 1). We then produced a decision-tree diagram using survival probabilities based on the literature (Figure 2). Data accounted for induction, re-induction and consolidation therapies but not for stem cell transplantation. The assumption-based model considered a maximum of four cycles of HiDAC and continuing decitabine until loss of benefit.
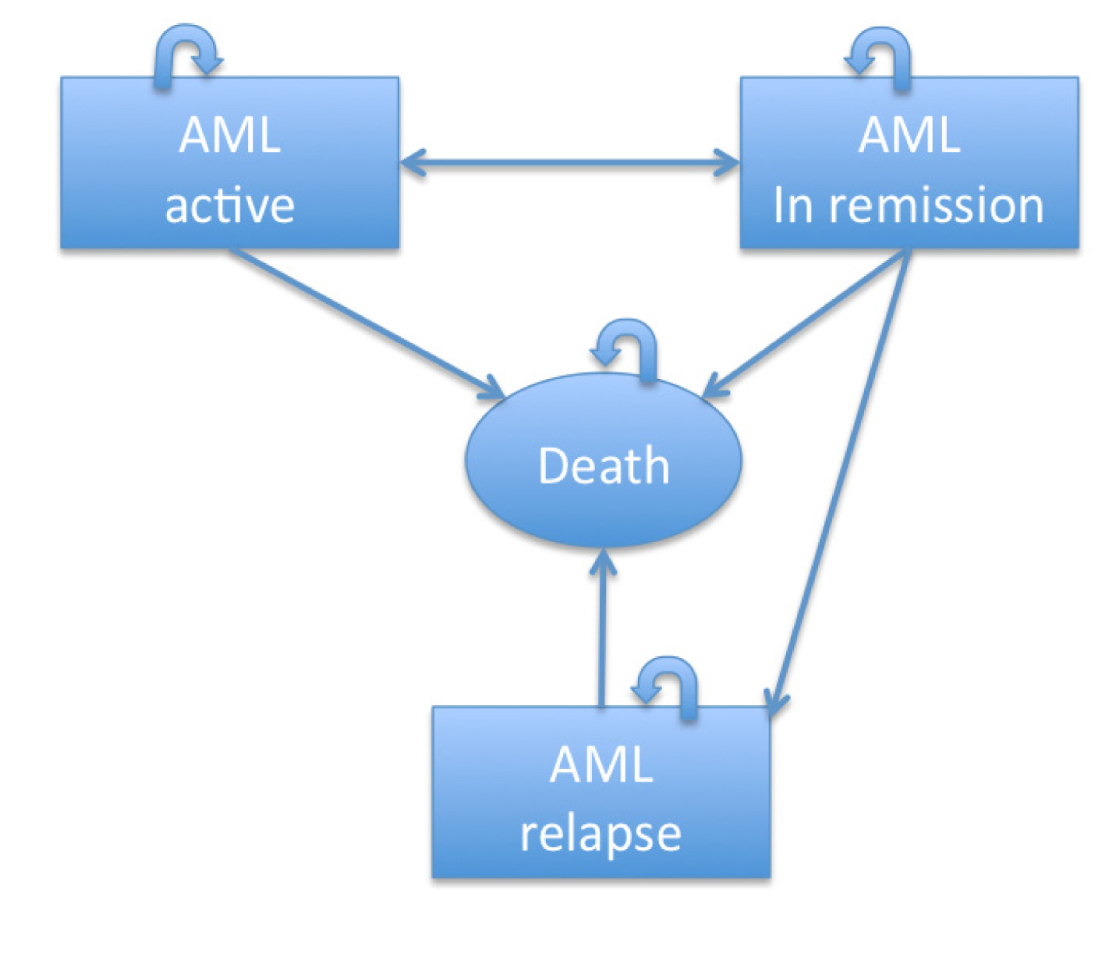
Figure 1 Markov health states in the model: This figure displays the health states among which patients transition in the model. Patients begin the model in a state of AML active disease. They can either stay in that state (indicated by the curved arrow, which would mean having refractory disease) or transition to another health state that is, achieving remission or dying without achieving remission. A double-sided arrow signifies that patients can move back and forth between health states. A one-sided arrow signifies that patients can move only from one health state to the next.
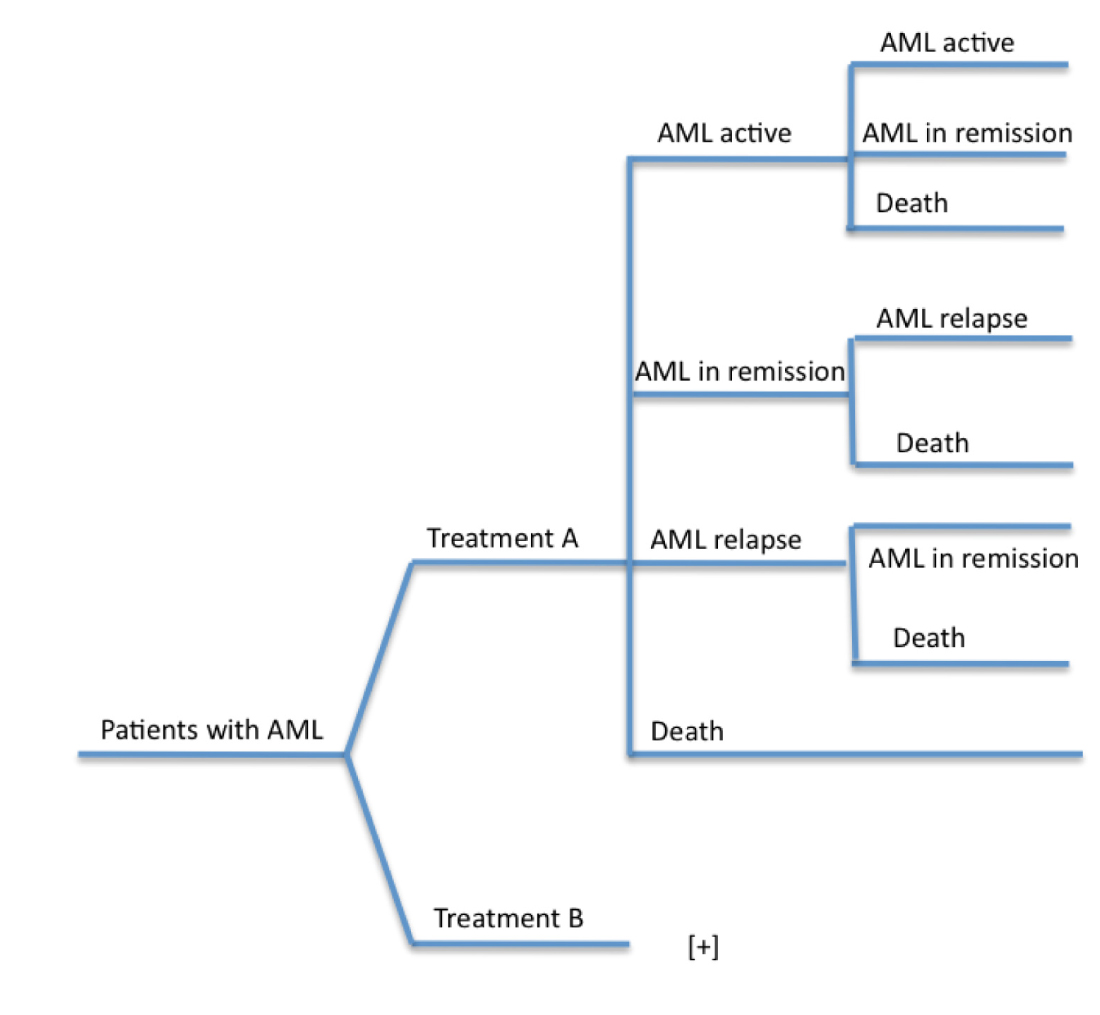
Figure 2 Decision-treed diagram of model structure: The figure displays the possible health states that patients may transition through the model. Treatment A: standard of care (AD). Treatment B: Decitabine.
Economical analysis
Cost effectiveness of the two approaches was assessed using an incremental cost-effectiveness ratio (ICER), where the difference of costs between the two strategies constituted the numerator and the incremental quality adjusted life-year (QALY) constituted the denominator. The detailed description of the concept of quality of life (QoL) is presented under ‘Utilities’ .
Study population
Patients entering the model reflected the demographics of clinical trials for AD and decitabine [1, 4, 6, 7, 11]. Each cohort incorporated 1,000 AML patients 60 and older years of age assigned to either treatment approach. Our assumption was that all patients would meet criteria to be treated with AD. Applying published survival data to our model, survival rates of AD versus decitabine were reproduced from reported clinical trials [4, 6].
Treatments
In our model, the patients were randomly assigned to one of the on two different approaches. The standard conventional induction therapy included standard-dose cytarabine (100-200 mg/m2 continuous infusion x 7 days) and daunorubicin (60 mg/m2 x 3 days). Consolidation therapy following AD consisted of HiDAC (3 gm/m2 every 12 h on days 1, 3 and 5) for four courses. The comparative treatment arm was decitabine 20 mg/m2/day as a daily one-hour infusion for 5 days every 4 weeks until progression. . On both arms, IDA-FLAG (idarubicin, fludarabine, cytarabine, G-CSF) was considered as a salvage therapy for patients having refractory disease or relapsing following CR [12]. Clinical trials and stem cell transplantation were not accounted for in our model.
Costs
The model considered the direct medical costs of treatment, including drug cost and administration. Cost of associated supportive care such as red blood cell and platelet transfusions were not individually considered for each treatment. However, they were indirectly evaluated since the cost of these supportive measures was embedded in the inpatient or outpatient clinic visit (since RPCI is a DRG exempt health care system). Additionally, for practical purposes assumption was conducted that such costs would be comparable between the two treatments.
Drug costs
Table 1 shows the costs for each drug based on a body surface area (BSA) of 1.85 m2 [13, 14]. A DRG exempt health care system such as RPCI does not charge for the cost of drugs separately; however, this is included in the day of hospital stay or clinic visits.
| Drug | Cost $ | Unit |
| Decitabine | 1,637.94 | 50 mg |
| Cytarabine | 12.08 | 2 gm |
| Daunorubicin | 58.62 | 20mg/4ml |
Administration costs
The costs of drug administration were based on the length of hospital stay cost or the cost of the total number of infusion clinic visits according to the 2012 billing policies (Table 2).
| Type | Unit | Cost $ |
| Direct hospital stay | One day | 2,104.75 |
| Infusion clinic visit | One day | 524.07 |
Time horizon and perspective
Time horizon is the time period in which costs and treatment benefits are accounted for [15]. Due to the old age of our population and the nature of AML with a median survival of approximately one year in elderly AML [1], a lifetime horizon of one year was chosen for this analysis [2]. In addition, QALY, that is, the benefit gained, accounting for both the quantity and quality gained by each treatment [15], was calculated.
Calculations
Third party payer perspective was used, specifically, the cost per number of courses of therapy to achieve CR was calculated by multiplying the cost of direct hospitalization days by the number of days of hospital stay and the number of cycles administered. The induction therapy, that is, the first cycle of either AD or decitabine, were calculated based on an inpatient hospitalization and the re-induction therapy with IDA-FLAG was also calculated on an inpatient hospitalization basis. The consolidation therapies with HIDAC and the following decitabine therapies were calculated as outpatient therapies based on as the total number of clinic visits.
Utilities
A utility value was assigned to each health state, which represented a QoL measure for that specific health state. Utility values typically ranged from 0 to 1, where 0 represented death, 1 represented perfect health and intermediate values represented degrees between these extremes. The QALY was calculated as the sum of products for the time spent in each state and the utility for that state over all the health states. There were no patients in our population with a perfect health i.e. a utility value of 1 by convention. QoL for patients with AML in remission on AD was considered equal to 0.91. This utility value was adopted, as equivalent to those patients with myelodysplastic syndrome who are transfusion independent [16, 17]. QoL for patients with active AML and treated with AD was 0.524 based on published reports [17, 18]. QoL for patients with active AML and treated with decitabine was estimated to be 0.71; this number was derived as the mean of QoL for AML with active disease and QoL of AML in remission [16-18]. QoL for death was 0. QoL for patients with AML in remission and continued on decitabine treatment was derived as the mean of QoL for patients with AML in remission on AD and QoL for patients with active AML treated with decitabine and was 0.81 [16, 17]. Additionally, we adopted a similar value of 0.81 for the QoL of patients receiving consolidation therapy with HiDAC [16, 17].
Statistical analysis
The semi-Markov model does not involve statistical analysis. All of the numerical calculations were carried out by R project, a general computing environment written by the R project team [19].
Results Top
Assuming 1,000 patients for each treatment arm in a semi-Markov model over one year time horizon, the QALYs for AD versus decitabine were calculated to be 0.47 and 0.61, respectively. In regards to clinical outcome, both treatment approaches conferred comparable overall survival rates (Table 3). In fact, the overall survival rates for AD versus decitabine were 45.2% and 50.5%. Decitabine-treated patients had a survival benefit of 0.14 of life expectancy in years when compared with AD-treated patients (0.69 vs. 0.55). Moreover, decitabine-treated patients had an improved QALY of 0.14 when compared with AD-treated patients (0.61 vs. 0.47). Over one year, decitabine-treated patients incurred fewer costs than AD-treated patients (Table 4).
| Month | AD | Decitabine | ||
| Survival rate | Survival rate (disease free) | Survival rate | Survival rate (disease free) | |
| 1 | 0.98 | 0.62 | ||
| 2 | 0.81 | 0.51 | ||
| 3 | 0.55 | 0.29 | 0.98 | 0.5 |
| 4 | 0.52 | 0.39 | ||
| 5 | 0.51 | 0.39 | ||
| 6 | 0.49 | 0.39 | 0.72 | 0.52 |
| 7 | 0.48 | 0.38 | /td> | |
| 8 | 0.47 | 0.38 | ||
| 9 | 0.47 | 0.38 | 0.57 | 0.44 |
| 10 | 0.46 | 0.38 | ||
| 11 | 0.46 | 0.38 | ||
| 12 | 0.45 | 0.38 | 0.51 | 0.44 |
Abbreviations: AD= cytarabine and daunorubicin
| AD | Decitabine | |
| Percentage survived at the end of period | 45.26% | 50.50% |
| Life expectancy in years | 0.55 | 0.69 |
| QALY in years | 0.47 | 0.61 |
| Expected cost in $ | 168,863 | 108,084 |
Abbreviations: ICER= Incremental cost-effectiveness ratio; QALY= quality-adjusted life year
Our results indicated that decitabine was cost saving compared with AD across all key parameters. For example, treatment over one year with decitabine cost $60,779 less than AD (Table 4). The ICER was estimated to be -$60,779/0.14 = -$433,756/year. This demonstrated that decitabine was less expensive and provided at least an equivalent clinical benefit to AD. Model ICER as represented in Table 5 was most sensitive to hazard ratios, cost of drugs and time horizon. By sensitivity analysis, decitabine was superior to AD in all cases of alternative parameters we considered (Table 5). Therefore, the conclusion that decitabine was cost saving when compared with AD was robust for all variations of ICERs (Table 5).
| Parameter[1] | ICER (per QALY gained) | |
| Upper | Lower | |
| Total cost of AD (± 10%) | -909,752 | -541,378 |
| Total cost of Dec (± 10%) | -613,934 | -837,196 |
| Discount rate (10% and 0%) | -749,138 | -701,802 |
| QoL for AD (± 10%) | -1,194,505 | -521,022 |
| QoL for Dec (± 10%) | -460,870 | -1,704,558 |
| Time Horizon (± 3 months) | -757,490 | -716,384 |
| Time Horizon (± 6 months) | -800,484 | -766,769 |
| HR/OS rate [2] | -676,830 | -752,498 |
| HR/OS adjustment done on AD only | -465,878 | -1,056,103 |
| HR/OS adjustment done on Dec only | -1,187,486 | -547,913 |
Abbreviations: 1= Variation of parameter by 10% range was based on prior published report; 2= Variation for HR/OS (hazard ratio for overall survival) was based on HR 0.5 +/- 0.05; ICER= Incremental cost-effectiveness ratio; QALY= quality-adjusted life year
DiscussionTop
Inducing AML patients 60 years of age and older with decitabine resulted in an ICER from a US third-party perspective of approximately $170,51 per QALY gained over a one-year time horizon. These results are based on published clinical trials examining AD versus decitabine for AML patients [4, 7, 11, 12]. The study also demonstrated comparable survival rates of AD and decitabine after compiling our model, which was consistent with published literature (Table 3). Consequently, the percentage of patients who survived at the end of period, the life expectancy in years, the QALY in years and the expected cost in dollars are presented in Table 4.
Base-case analysis
The calculated ICER of decitabine over AD suggests that decitabine dominates AD. Even though the initial cost of these two treatments were comparable, the total expected cost of AD is much higher than the total expected cost of decitabine due to higher cost of subsequent treatment at relapse and higher probability of relapse. While the total survival rates are also comparable at the end of the time horizon we considered, QALY for decitabine was much higher than that for AD, merely a reflection of a much higher QoL when treated for AML by decitabine (0.71) versus AD (0.524). The latter is an important reason why decitabine dominated AD. The QoL numbers were derived by estimation based on available literature; however, if CEA were based on a clinical trial then RAND's standards (are standards for assessing QoLs for various treatments of diseases established by the RAND corporation) to estimate QoL [20] would be feasible.
As decitabine dominated AD, the emphasis to discuss ICER in terms of dollar figures was not the major important factor in this setting. In other words, the original purpose of ICER, that is, additional dollar amount required to spend to increase QALY by a year was not pertinent in this clinical scenario. Since the decitabine was superior to AD primarily as it conveyed a higher QoL.
Sensitivity analysis
One-way sensitivity analysis for our basic parameters was conducted and revealed that decitabine was superior in all tested parameters. In all cases, most notably, variations in costs, discount rates, time horizon, and hazard ratios for overall survival, the conclusion of dominance of decitabine would not be overturned. This is a very strong conclusion, compared with all other studies, which found that their results were somewhat sensitive to discount rates and time horizon.
The series of sensitivity analyses demonstrated the strength of cost-effectiveness analysis results for decitabine over AD. The results were most sensitive to the hazard ratio, the unit cost of the drugs, the QoL and the discount rate. As important parameters in our evaluation, time horizon reflected the beneficial effect of each treatment approach per lifetime, while the discount rate was the rate that needed to discounted at future date at to estimate accurately net present value of cost and benefits. Both time horizon and discount rate are important parameters in economic assessments. Despite the fact that drug costs for decitabine comprised an important proportion in the incremental costs versus AD, the unit cost of decitabine did not have the most significant impact on the results. As the nature of the treatment approach with AD is inpatient based therapy versus predominantly an outpatient administration with decitabine, therefore the health system incurred much more expenses with an inpatient hospitalization as opposed to outpatient clinic visits.
Nonetheless, our analysis had several limitations that need to be considered when interpreting the results. First and foremost, this analysis is based on a theoretical model and not on data of clinical trials. Additionally, the QoL data were extrapolated from other diseases and in certain health states were estimated for treatment and disease stage. Ideally, a QoL data must be based on RAND’s standard criteria [20, 21]. A secondary analysis to consider a societal perspective was not performed. However, this was inherent to the nature of our analysis. This would have included the effect of interventions on lost work productivity, caregiver costs, and effects of cancer on QoL of spouses/ partners. We did not want to perform an analysis similar to other models when they adopted numbers deducted for prostate cancer or chronic lymphocytic leukemia [13]. On the other hand, despite that grade 3 and 4 adverse events were reported to be significantly different between both treatments, these adverse events may have also been associated with prolongation of hospital inpatient stay or frequent clinic visits. The costs of these complications were not accounted. However, they were indirectly considered by calculating the costs of hospital inpatient stay or frequent clinic visits. At this point, these were out the scope of this analysis.
We recommend conducting prospective clinical trial to evaluate objectively the utility values for each health state and their corresponding treatment prior to applying an economic model to compare these approaches. As there is no available literature to report on patient-outcomes with AML undergoing this type of induction therapy. Therefore, we did extrapolate the utilities for AML in a similar fashion to other published report for example from a study of chronic myeloid leukemia in blast phase [17, 18]. We also have used utility data reported in patients with myelodysplasia to complete our model [16, 17]. On the other hand, it is essential to note that our results must be carefully applied when considering a diagnosis related group health care system. Finally, perhaps more reliable data must be derived from economic analyses of phase III clinical trials despite the questionable feasibility of such studies [22].
ConclusionTop
By applying an economic model to the best available clinical data, our results suggest that decitabine comparable clinical benefit was complimented with a superior cost-effectiveness when treating elderly patients with AML. Induction therapy with decitabine was associated with comparable survival, improved QALY and fewer expenses. While cost effectiveness is certainly important, decitabine may be arguably considered for elderly newly diagnosed AML patients given the economic pressures in the US health system; however, this is not a criterion for drug approval.
Conflict of interest
All the authors declare that they have no conflict of interest.
ReferencesTop
[1] Burnett A, Wetzler M, Löwenberg B (2011) Therapeutic advances in acute myeloid leukemia. J Clin Oncol 29:487–494. Article Pubmed
[2] Appelbaum FR, Gundacker H, Head DR, Slovak ML, Willman CL, et al. (2006) Age and acute myeloid leukemia. Blood 107:3481–3485. Article Pubmed
[3] Juliusson G, Antunovic P, Derolf A, Lehmann S, Möllgård L, et al. (2009) Age and acute myeloid leukemia: real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood 113:4179–4187. Article Pubmed
[4] Löwenberg B1, Ossenkoppele GJ, van Putten W, Schouten HC, Graux C, et al. (2009) High-dose daunorubicin in older patients with acute myeloid leukemia. N Engl J Med 361:1235–1248. Article Pubmed
[5] Wang J, Yang YG, Zhou M, Xu JY, Zhang QG, et al. (2013) Meta-analysis of randomised clinical trials comparing idarubicin cytarabine with daunorubicin cytarabine as the induction chemotherapy in patients with newly diagnosed acute myeloid leukaemia. PLoS One 8:e60699. Article Pubmed
[6] Lübbert M, Rüter BH, Claus R, Schmoor C, Schmid M, et al. (2012) A multicenter phase II trial of decitabine as first-line treatment for older patients with acute myeloid leukemia judged unfit for induction chemotherapy. Haematologica 97:393–401. Article Pubmed
[7] Blum W, Garzon R, Klisovic RB, Schwind S, Walker A, et al. (2010) Clinical response and miR-29b predictive significance in older AML patients treated with a 10-day schedule of decitabine. Proc Natl Acad Sci U S A 107:7473–7478. Article Pubmed
[8] NCCN, The National Comprehensive Cancer Network (NCCN). Article
[9] Dabrowska DM (2012) Estimation in a semi-Markov transformation model. Int J Biostat 8:Article 15. Article Pubmed
[10] Raberto M, Rapallo F, Scalas E (2011) Semi-Markov graph dynamics. PLoS One 6:e23370. Article Pubmed
[11] Kantarjian H, Oki Y, Garcia-Manero G, Huang X, O'Brien S, et al. (2007) Results of a randomized study of 3 schedules of low-dose decitabine in higher-risk myelodysplastic syndrome and chronic myelomonocytic leukemia. Blood 109:52–57. Article Pubmed
[12] Steinmetz HT, Schulz A, Staib P, Scheid C, Glasmacher A, et al. (1999) Phase-II trial of idarubicin, fludarabine, cytosine arabinoside, and filgrastim (Ida-FLAG) for treatment of refractory, relapsed, and secondary AML. Ann Hematol 78:418–425. Article Pubmed
[13] Hornberger J, Reyes C, Shewade A, Lerner S, Friedmann M, et al. (2012) Cost-effectiveness of adding rituximab to fludarabine and cyclophosphamide for the treatment of previously untreated chronic lymphocytic leukemia. Leuk Lymphoma 53:225–234. Article Pubmed
[14] Hornberger J, Reyes C, Lubeck D, Valente N (2008) Economic evaluation of rituximab plus cyclophosphamide, vincristine and prednisolone for advanced follicular lymphoma. Leuk Lymphoma 49:227–236. Article Pubmed
[15] Pan F, Peng S, Fleurence R, Linnehan JE, Knopf K, et al. (2010) Economic analysis of decitabine versus best supportive care in the treatment of intermediate- and high-risk myelodysplastic syndromes from a US payer perspective. Clin Ther 32:2444–2456. Article Pubmed
[16] Goss TF, Szende A, Schaefer C, Totten PJ, Knight R, et al. (2006) Cost effectiveness of lenalidomide in the treatment of transfusion-dependent myelodysplastic syndromes in the United States. Cancer Control 13 Suppl:17–25. Article Pubmed
[17] Gidwani R, Khan ZM, Fenaux P, Beach CL, Pashos CL (2012) A cost-effectiveness analysis of using azacitidine vs. decitabine in treating patients with myelodysplastic syndromes. J Med Econ 15:145–154. Article Pubmed
[18] Dalziel K, Round A, Garside R, Stein K (2005) Cost effectiveness of imatinib compared with interferon-alpha or hydroxycarbamide for first-line treatment of chronic myeloid leukaemia. Pharmacoeconomics 23:515–526. Article Pubmed
[19] DeLima M, Ghaddar H, Pierce S, Estey E (1996) Treatment of newly-diagnosed acute myelogenous leukaemia in patients aged 80 years and above. Br J Haematol 93:89–95. Article Pubmed
[20] Hays R (2001) The RAND-36 Measure of Health-Related Quality of Life. The Finnish Medical Society Duodecim. Web- only. Annals of Medicine, v. 33, pp. 350–357. Article
[21] Collet TH, Salamin S, Zimmerli L, Kerr EA, Clair C ,et. al. (2011) The quality of primary care in a country with universal health care coverage. J Gen Intern Med 26:724–730. Article Pubmed
[22] Bennett CL, Golub R, Waters TM, Tallman MS, Rowe JM (1997) Economic analyses of phase III cooperative cancer group clinical trials: are they feasible Cancer Invest 15:227–236. Article Pubmed



