Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 7, July 2014, Pages 96–99
Case reportOpen Access
Apocrine carcinoma of the scalp – A case report and highlights for diagnosis
- 1 Department of Surgery (RLA), Division of Otolaryngology – Head and Neck Surgery, William Beaumont Hospital, Troy, Michigan
- 2 Department of Anatomic and Clinical Pathology (LW), William Beaumont Health System, Royal Oak, Michigan
- 3 Department of Otolaryngology – Facial Plastic Surgery (DAG), Henry Ford Macomb Hospital, Clinton Township, Michigan, USA
*Corresponding author: Richard L. Arden, MD, FACS, Lakeshore Ear, Nose, Throat, PC, 11080 Hall Road, Suite A, Sterling Heights, MI 48314, Tel.: (586) 254-7200; Fax: (586) 254-7201; Email: richardlarden@aol.com
Received 21 May 2014 Revised 13 June 2014 Accepted 20 June 2014 Published 27 June 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-14
Copyright: ©2014 Arden RL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Apocrine sweat gland carcinoma is a rare cutaneous adnexal neoplasm that may occur in the scalp, eyelid, and ear canal. More often, it presents as an asymptomatic, slow-growing, solid or cystic mass with variable color. The clinical appearance may be confused with basal cell carcinoma and the pathologic appearance with metastatic breast carcinoma. Supportive morphologic and immunohistochemical findings, together with careful clinicopathologic correlation, aid in diagnosis. The mainstay of treatment is wide local excision and neck dissection for node positive disease. The role of elective neck dissection and adjuvant radiotherapy remain controversial. Chemotherapy has been used for palliation in disseminated disease. This case report offers an algorithm that can aid the clinician in differential diagnosis of this often elusive neoplasm.
Keywords: apocrine carcinoma; adnexal neoplasm; scalp carcinoma; immunohistochemistry
IntroductionTop
Sweat gland carcinomas (SGC) account for <0.01% of primary skin tumors and, of these, approximately 110 cases of apocrine differentiation (AC) have been reported to date. There is no racial or gender predilection [1]. The predominant age range at presentation is 50-70 years. While >50% of AC’s occur in the axilla, the ceruminous glands of the ear, glands of Moll in the eyelid, scalp, anogenital area, and even the chest, lip, foot, and wrist have been reported sites of occurrence.
Clinically, most SGC’s present as slow-growing, painless, red-violaceous papules that progress to form single or multiple solid nodules that infiltrate subcutaneous tissue, and are typically ignored for months to years. The predominant route of metastatic spread is to lymph nodes, but dissemination most commonly to lung and even to liver, bone, or brain may rarely occur [2].
Considerable variations in morphology, and often complex and admixed architectural patterns, complicate interpretation of SGC’s. According to the 2006 WHO classification, adnexal neoplasms can be divided into three categories based on their line of differentiation; eccrine-apocrine, follicular, and sebaceous [3]. AC’s are typically poorly circumscribed dermal neoplasms that may extend into the subcutis or overlying epidermis with pagetoid spread. Numerous architectural patterns including tubular, cribriform, papillary, and/or solid may be seen. Cells are usually large with abundant, granular eosinophilic cytoplasm. Characteristic decapitation secretion, the sine qua non of apocrine differentiation, is often seen throughout the lesion, but may only be focally present in poorly differentiated cases.
Immunohistochemistry is indispensable in the diagnosis of AC’s, particularly in excluding other cutaneous tumors which may mimic adenocarcinoma. This includes melanoma (S-100+) or squamous cell carcinoma (cytokeratin 5/6+ [CK5/6]), and metastatic adenocarcinomas from non-mammary primary sites, such as lung (thyroid transcription factor 1+ [TTF-1]), colon (cytokeratin 20+ [CK20]) or female genital tract (paired box 8+ [PAX-8]). Markers that have reportedly been expressed in AC’s include CAM5.2, cytokeratin 7 (CK7), epithelial membrane antigen (EMA), carcinoembryonic antigen (CEA), gross cystic disease fluid protein-15 (GCDFP-15), androgen receptor (AR), estrogen receptor (ER) and progesterone receptor (PR). However, there is ambiguity within the medical literature regarding the relative usefulness and proportion of cases that stain for these markers. Special stains, such as periodic acid-Schiff with diastase digestion (PASD), may highlight granules and intracytoplasmic iron. Neither morphology nor immunohistochemistry can reliably distinguish primary cutaneous apocrine carcinomas from apocrine carcinomas of the breast metastatic to the skin; clinicopathologic correlation to exclude a history of breast cancer is needed to confirm the diagnosis.
Case reportTop
A 67 year-old Caucasian female presented with a three-year history of a slowly growing asymptomatic occipital scalp mass. Past medical history included insulin-dependent diabetes mellitus, coronary artery disease, meningioma, and carotid body paraganglioma, but was negative for radiation treatment in the head and neck, preexistent skin lesion in that area, or cutaneous malignancy. Examination showed a well-circumscribed, dark red, raised, firm nodular subcutaneous mass with granular appearance in its superficial aspect, clinically resembling basal cell carcinoma. No pathological adenopathy was evident.
Excisional biopsy to the subgaleal plane with direct primary closure was performed, revealing a 2.4 cm tumor extending to a depth of 9 mm, located 0.8 mm from the closest deep margin. Histologic sections showed a well-circumscribed multinodular dermal tumor with predominantly tubulopapillary architecture and focal necrosis (Figure 1). The neoplastic cells had granular eosinophilic cytoplasm and distinct decapitation secretion. Nuclei were bland, with smooth nuclear borders, fine chromatin and occasional small nucleoli (Figure 2). Angiolymphatic and perineural invasion were absent. Immunohistochemistry showed the lesion to be diffusely positive for AE1/3, CK7, CEA, 34βE12, and GCDFP-15 (Figure 3); focal staining was seen with ER (strong staining in 10% of nuclei), PR (strong staining in 10% of nuclei), and mammaglobin. The neoplastic epithelial cells were negative for S-100, cytokeratin 14 (CK14), CK5/6, PAX-8, TTF-1, CK20, and vimentin (Table 1). The combined histologic and immunohistochemical findings were diagnostic of a well-differentiated apocrine carcinoma.
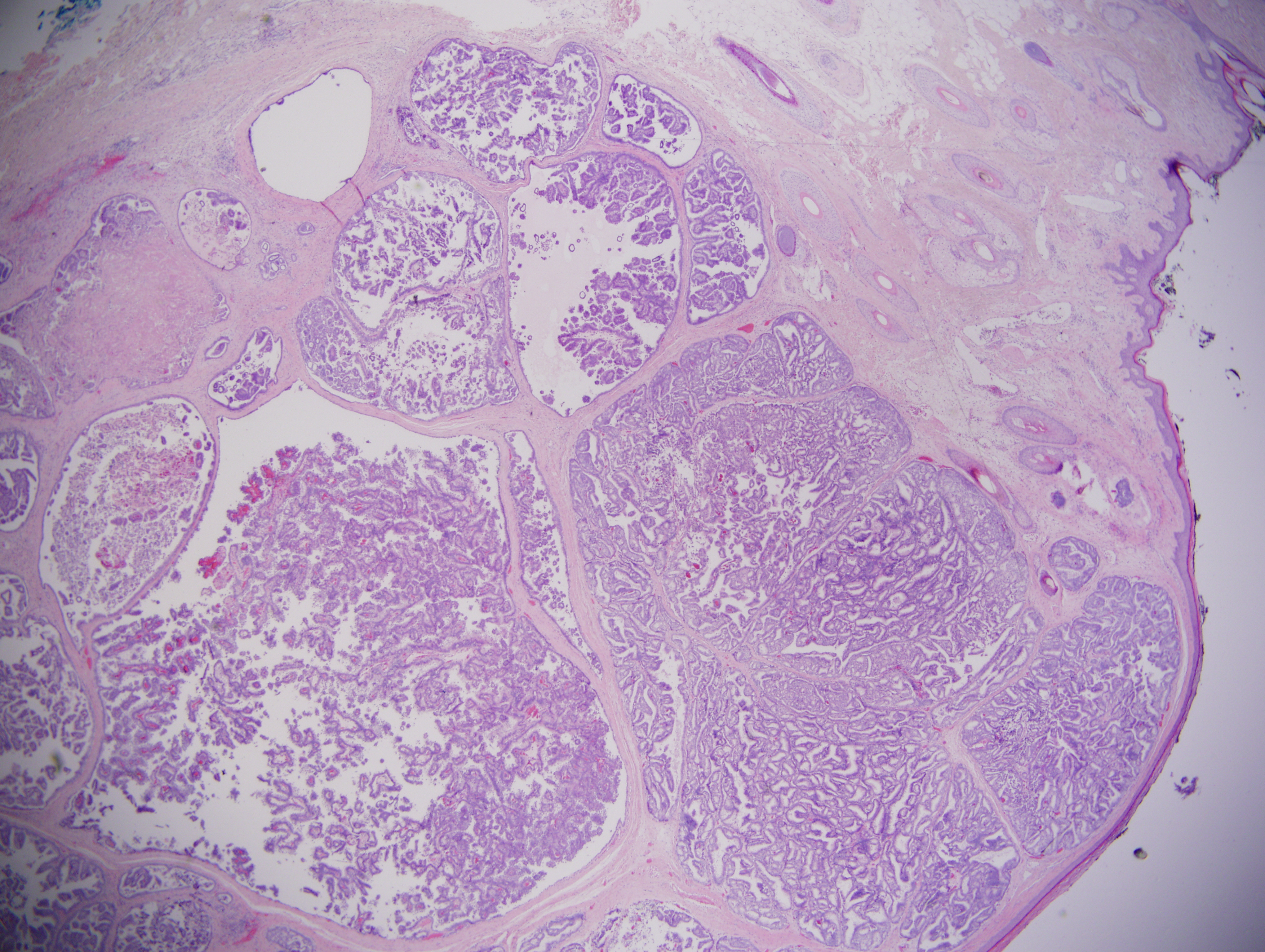
Figure 1 Hematoxylin and eosin stain, 20x magnification. This low power image demonstrates a well-circumscribed lesion centered in the dermis. The tumor is multinodular with predominantly tubulopapillary architecture. Focal necrosis can be appreciated in the upper left.
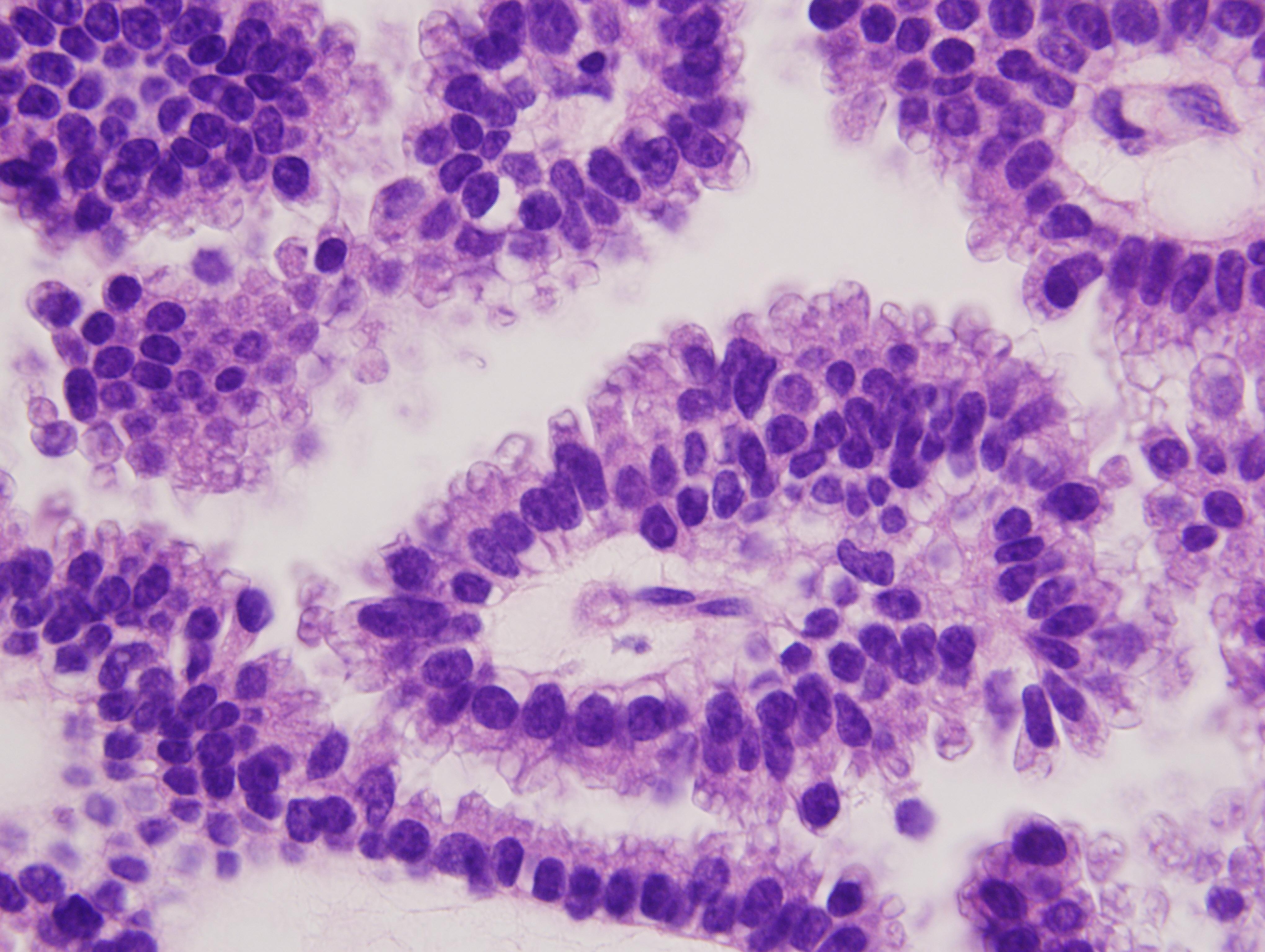
Figure 2 Hematoxylin and eosin stain, 1000x magnification. This high power image demonstrates cytologic features of the neoplastic apocrine cells, including abundant, granular eosinophilic cytoplasm with prominent apical decapitation secretion. The oval-shaped nuclei have smooth nuclear borders and fine chromatin with occasional small nucleoli.
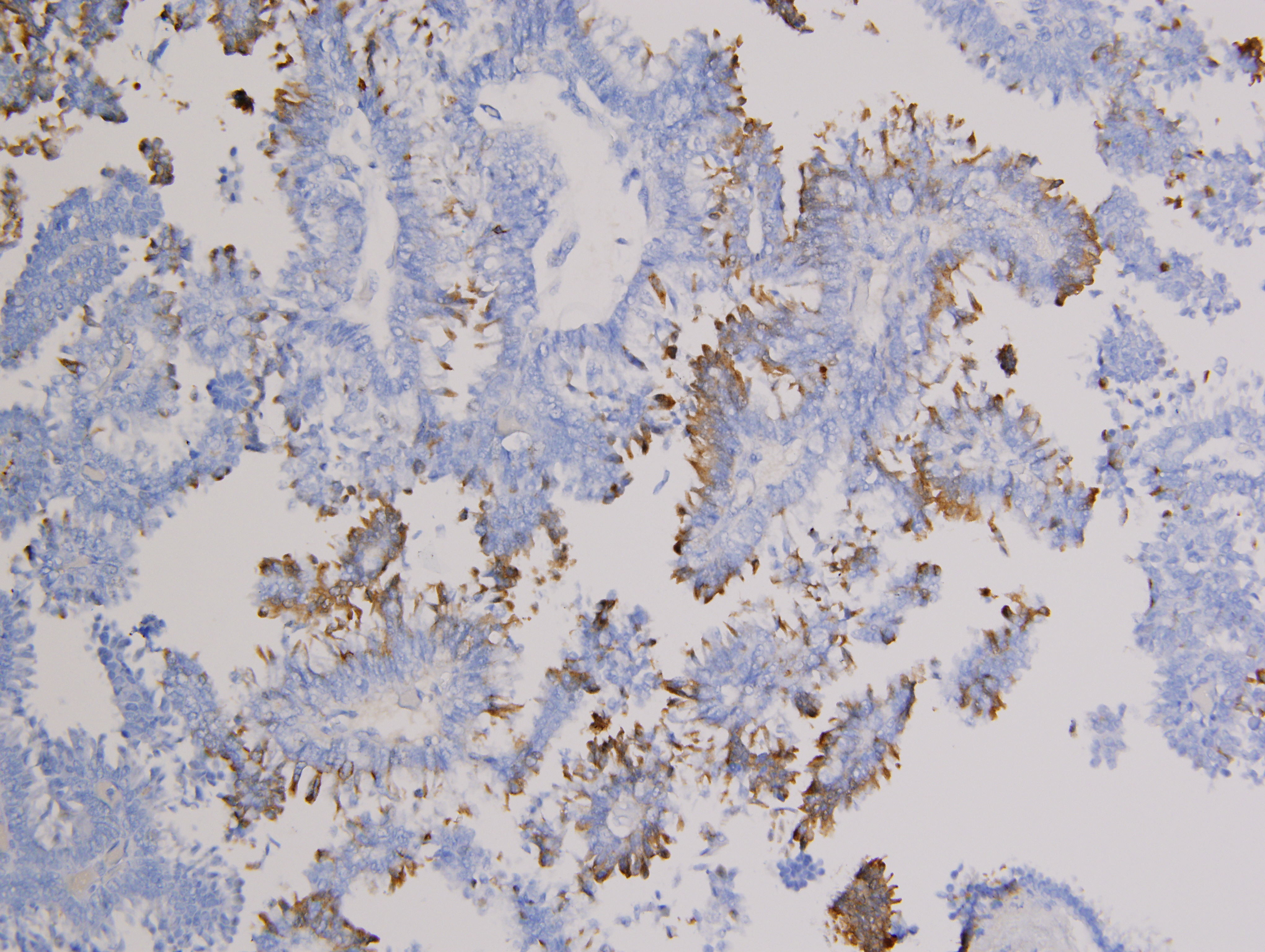
Figure 3 Immunohistochemical stain for gross cystic disease fluid protein (GCDFP-15), 200x magnification. Neoplastic apocrine epithelial cells demonstrate patchy, strong cytoplasmic staining with antibodies to GCDFP-15.
| Positive | Focally positive | Negative |
| AE 1/3 | Estrogen receptor | S-100 |
| CK7 | Progesterone Receptor | CK14 |
| CEA | Mammaglobin | CK5/6 |
| GCDFP-15 | PAX-8 | |
| 34?E12 | TTF-1 | |
| CK20 | ||
| Vimentin |
Abbreviations: GCDFP-15 = secretory cytoplasmic glycoprotein produced by apocrine acinar cells of breast, salivary gland, sweat gland, and seminal vesicles; CK20 = low molecular weight keratin found in gastrointestinal tract, urothelium and Merkel cells; PAX-8 = transcription factor expressed in thyroid, nephric ducts, Mullerian ducts; TTF-1 = transcription factor expressed in thyroid, parathyroid, lung, and anterior pituitary; S-100 = calcium binding protein expressed in many cell types including neural crest; in this case, excludes melanoma/favors apocrine over eccrine.
Workup included a chest x-ray and mammogram with normal findings. CT scan of the neck demonstrated scattered nonenlarged lymph nodes in the anterior and posterior cervical chains which were non-FDG avid on PET scan.
Ultimately, the patient underwent wide local re-excision of the scalp scar with 2 cm peripheral margins to a 2 mm depth into the outer calvarial cortex. The defect was repaired by a rotational scalp flap and final pathologic margins remained clear. Prolonged three-month interval follow-ups are planned with annual neck CT scans to monitor for potential locoregional recurrence.
DiscussionTop
SGC’s are rare adnexal neoplasms whose clinical behavior is typically characterized by a protracted course, potential for rapid local growth, recurrence, and a tendency for both regional and rarely (14 cases reported to date) [4] distant metastases. As a subset of these, apocrine sweat gland carcinomas of the scalp are exceedingly rare, with the first well-documented report without metastasis published in 2012 [5]. Due to its low prevalence, prognostic factors are difficult to establish. Management of the primary site should entail a wide local excision (WLE) of 1-2 cm, guided by histologic findings. At critical subsites for tissue preservation, Mohs technique could be considered.
While the role of therapeutic regional nodal dissection is well-established for demonstrable lymphatic metastases, the indication and value of elective nodal dissection (END) remains controversial and unresolved in the literature. Despite the fact that local recurrence more often tends to occur before lymphatic spread, either one may occur many years after the primary excision. This supports the belief by some authors that END does not appear to improve survival or decrease recurrence rates in the absence of documented metastases [6]. Other authors have argued END should be considered with recurrent lesions after WLE, or with highly undifferentiated tumors, because of the strong tendency for regional nodal spread. This approach is supported by historical data in which >50% of patients with moderate to poorly differentiated AC’s had lymph node metastases at the time of diagnosis [4].
Sentinel lymph node biopsy (SLNB), while capable of detecting occult lymphatic spread and providing useful staging information with therapeutic implications for high-risk cutaneous malignancies (e.g., Merkel cell carcinoma, melanoma), has yet to show prognostic value for well-differentiated SGC’s. In two retrospective studies involving SLNB of three scalp SGC’s, that included two eccrine and one hydradenocarcinoma [7, 8], two harbored lymphatic spread and underwent regional lymphadenectomy, only one of which was found to have additional lymph nodes positive for tumor.
Attributable to the rarity of this tumor and lack of controlled studies, recommendations on the efficacy, dose, and/or schedule of adjuvant radiation treatment are lacking. One group of authors has recommended that adjuvant radiotherapy should be considered in cases in which tumor size exceeds 5 cm, resection margins are positive or close (<1cm), the tumor is moderately to poorly differentiated, angiolymphatic invasion is present, and/or extranodal extension or extensive nodal involvement (>4 lymph nodes) is identified [9]. Similarly, existing literature provides only scattered case reports of disseminated AC treated with variable chemotherapeutic drugs and schedules. The longest complete response, which was two years, was reported with a four-drug regimen using doxorubicin, cyclophosphamide, vincristine, and bleomycin in a case of nodal and visceral metastasis [10]. The low incidence of AC would likely preclude randomized controlled clinical trials.
Overall, five-year local recurrence rates are 28%, with survival rates of 99% for localized disease and 43% for distant disease [1]. Ten-year disease-free survival is 9% for lymph node positive and 56% for lymph node negative status. Given that AC’s are 62% ER positive, 60% PR positive, and 36% AR negative [11], future directions may include hormonal therapy to improve locoregional and distant control rates, recognizing that higher grade AC’s are less likely to express steroid receptors.
The case presented herein highlights the complex and often elusive nature of SGC characterization, often attributable to the considerable variation in pattern and ambiguity of immunohistochemical diagnostic criteria. Using the latter with characteristic morphologic findings, and the lack of clinical or pathologic features to support metastatic adenocarcinoma originating from sites such as breast, lung, prostate or GI tract, a diagnosis of primary cutaneous AC can usually be made. Of particular note, breast carcinoma with apocrine differentiation is histologically indistinguishable from AC’s and has been misdiagnosed and mistreated, owing to the prevalence of the former and rarity of the latter. Morphologic features supporting a primary cutaneous tumor include occurrence of the tumor in close relationship to normal apocrine glands and, if a prior breast malignancy is available for pathologic review and comparison, demonstration of marked differences in the morphologic and immunohistochemical findings between the breast and skin tumors. The immunohistochemical profile of this tumor (Figure 4) ruled out other primary cutaneous malignancies and non-mammary metastatic carcinomas, but a metastatic apocrine carcinoma from the breast could not be entirely excluded without the clinical findings of a negative mammogram and lack of history of breast cancer to further support the diagnosis.
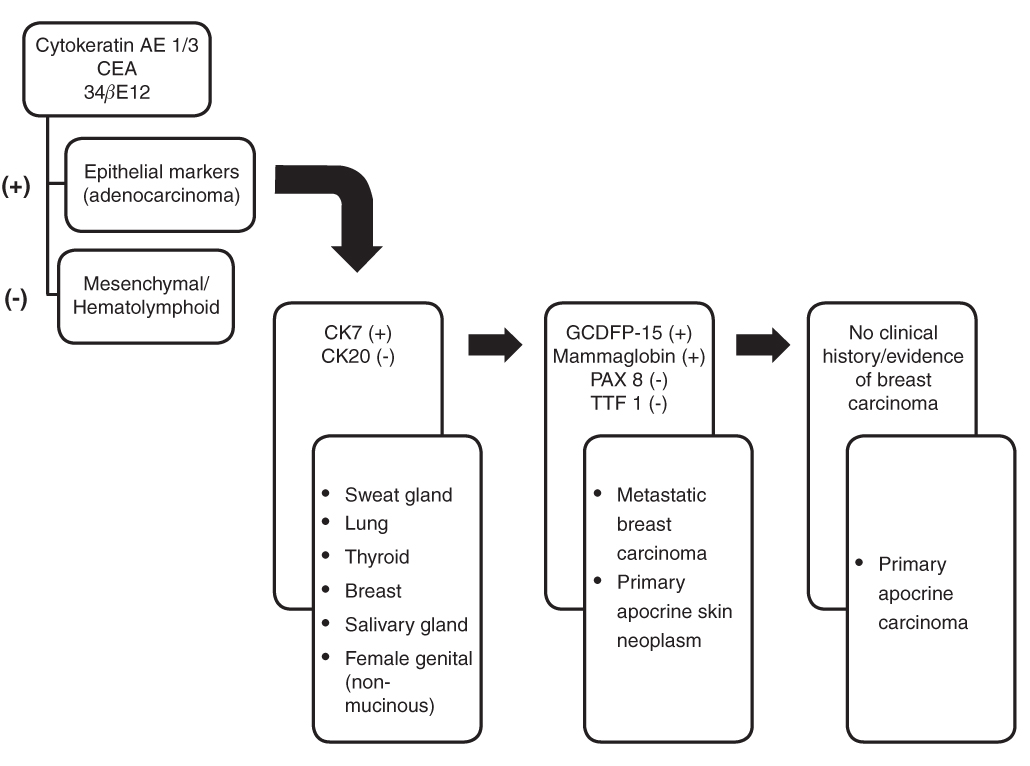
Figure 4 Implication of immunohistochemical results.
Since there are no distinguishable characteristics to raise suspicion, SGC’s are often not clinically suspected and are typically mistaken for basal cell carcinoma, as in our case. A heightened awareness and diligent workup of this rare entity is necessary if optimal outcomes are to be achieved.
ConclusionTop
AC is a very rare tumor that may occur on the face and scalp. It is frequently not suspected clinically and requires a high index of suspicion, with supportive histopathologic and immunohistochemical findings. Differentiation between cutaneous metastases from the breast (a modified apocrine gland) and a primary cutaneous adnexal tumor can be difficult. The standard treatment is WLE with neck management dictated by nodal status and primary tumor characteristics. Chemotherapy has a limited and palliative role in management.
Conflict of interest
The authors wish to express that they have no conflict of interest.
ReferencesTop
[1] Hollowell KL, Agle SC, Zervos EE, Fitzgerald TL (2012) Cutaneous apocrine adenocarcinoma: defining epidemiology, outcomes, and optimal therapy for a rare neoplasm. J Surg Oncol 105:415–419. Article Pubmed
[2] Gallerani E, Ciriolo M, Rossini C, Cavalli F (2007) Axillary apocrine carcinoma with brain metastases. J Clin Oncol 25:5655–5656. Article Pubmed
[3] Le Boit PE, Burg G, Weedon D, Sarasin A (2006) World Health Organization Classification of Tumours. Pathology and Genetics of Skin Tumours. Oxford, UK: Lyon: IARC Press.
[4] Vucinić I, Stojadinović T, Mikez ZB, Danić D, Coha B (2012) Apocrine carcinoma of the scalp with aggressive clinical course--a case report and review of the literature. Coll Antropol 36:209–212. Article Pubmed
[5] Kim HK, Chung KI, Park BY, Bae TH, Kim WS, et al. (2012) Primary apocrine carcinoma of scalp: report of primary scalp cutaneous apocrine carcinoma indistinguishable from cutaneous metastasis of breast carcinoma. J Plast Reconstr Aesthet Surg 65:e67-e70. Article Pubmed
[6] Tlemcani K, Levine D, Smith RV, Brandwein-Gensler M, Staffenberg DA, et al. (2010) Metastatic apocrine carcinoma of the scalp: prolonged response to systemic chemotherapy. J Clin Oncol 28:412–414. Article Pubmed
[7] Delgado R, Kraus D, Coit DG, Busam KJ (2003) Sentinel lymph node analysis in patients with sweat gland carcinoma. Cancer 97:2279–2284. Article Pubmed
[8] Bogner PN, Fullen DR, Lowe L, Paulino A, Biermann JS, et al. (2003) Lymphatic mapping and sentinel lymph node biopsy in the detection of early metastasis from sweat gland carcinoma. Cancer 97:2285–2289. Article Pubmed
[9] Chamberlain RS, Huber K, White JC, Travaglino-Parda R (1999) Apocrine gland carcinoma of the axilla: review of the literature and recommendations for treatment. Am J Clin Oncol 22:131–135. Article Pubmed
[10] Mezger J, Remberger K, Schalhorn A, Wohlrab A, Wilmanns W (1986) Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Med Oncol Tumor Pharmacother 3:29–34. Article Pubmed
[11] Robson A, Lazar AJ, Ben Nagi J, Hanby A, Grayson W, et al. (2008) Primary cutaneous apocrine carcinoma: a clinico-pathologic analysis of 24 cases. Am J Surg Pathol 32:682–690. Article Pubmed



