Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 8, August 2014, Pages 111-115
Original researchOpen Access
Small satellite pulmonary nodules in operable lung cancer: Diagnosis and therapeutic strategy
- 1 Department of thoracic surgery, Chinese People's Liberation Army General Hospital, Beijing, China
*Corresponding author: Dr. Juntang Guo, Department of Thoracic Surgery, Chinese People's Liberation Army General Hospital, 28# Fuxing Street, Beijing-100853, China. Email: moc.anis@103gnatnujoug
Received 19 May 2014 Revised 10 July 2014 Accepted 17 July 2014 Published 25 July 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-17
Copyright: © 2014 Guo J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Objective: The use of high-resolution computed tomography (HRCT) has improved the management of non-small cell lung cancer (NSCLC), but has also increased the detection of indeterminate satellite nodules. Obtaining differential diagnosis of nodules less than 10 mm is difficult but essential for choosing optimal therapeutic strategies. Here, we evaluated the characteristics of small satellite nodules in patients with operable NSCLC and examined the optimal diagnostic and therapeutic approach in patients with small satellite nodules. Methods: Using data from a prospective database, all surgically treated patients diagnosed with NSCLC from 2008 to 2011 were retrospectively reviewed. Patients presenting with small pulmonary non-GGO nodule(s) were identified. Results: A total of 1206 patients underwent complete resection for NSCLC, out of which 45 patients presented with 60 nodules less than 10 mm on thoracic CT. Twenty-six nodules (43%) were malignant, 31 (52%) were benign, and 3 (5%) were of undetermined nature. Tumor histology (adenocarcinoma vs. nonadenocarcinoma), TNM stage (advanced stage vs. early stage), and nodule size (6–10 vs. ≤ 5 mm) were associated with nodule malignancy. The incidence of metastatic nodules in patients with stage III NSCLC was significantly higher than that in patients with stage I NSCLC. In contrast, the incidence of multiple primary lung cancers in patients with stage I NSCLC was significantly higher than that in patients with stage III NSCLC. Conclusion: Differential diagnosis of indeterminate satellite lesions in patients with operable NSCLC is critical. Surgery should be performed cautiously in patients with stage III NSCLC having nodules larger than 5 mm.
Keywords: lung cancer; satellite nodule; diagnosis; surgery; computed tomography
IntroductionTop
Advances in computed tomography (CT) have increased the detection of small pulmonary nodules [1]. Small coexisting lesions are often encountered on CT before surgery for non-small cell lung cancer (NSCLC) [2]. We defined these coexisting nodules in bilateral lung parenchyma with lung cancer as satellite pulmonary nodules. Unfortunately, most of these nodules cannot be characterized as benign or malignant with CT or other imaging modalities. Indeed, studies have shown that fiberoptic bronchoscopy is not often helpful in the diagnosis of peripheral pulmonary nodules [3]. Moreover, pulmonary nodules that are less than 10 mm in size cannot be evaluated accurately by 18F-fluorodeoxyglucose positron emission tomography (PET-FDG) [4], and CT-guided percutaneous needle aspiration is not very accurate for analysis of small pulmonary nodules [5]. In patients with operable lung cancer, the coexistence of satellite lesions may change the TNM status and influence therapeutic decisions. If a nodule is considered malignant, it may be an intrapulmonary metastasis or a second primary neoplasm. NSCLC may be classified as stage T3 (nodule in the same lobe), T4 (ipsilateral nodules in different lobes), or M1a (contralateral pulmonary nodules), or as synchronous multiple lung cancers, as detailed in the seventh edition of the TNM classification [6]. As prognosis is significantly better in patients with stage I (T1-2N0M0) or synchronous primary lung cancer than those with stage IIIB (T4N0M0) or stage IV (T1-2N0M1), the existence of malignant pulmonary nodules could be considered a contraindication to surgery [7]. A number of recent studies have supported the role of SPLC resection, which can yield comparable results as those in patients with single lung cancers of similar stages [8, 9].
Several previous studies have focused on pulmonary nodules including focal ground-glass opacity (GGO) in patients with NSCLC [10, 11]. Mixed nodular GGO have a relative high proportion of lung carcinoma even though when lesions were small in size [12, 13]. There are few investigations about non-GGO nodules coexisted with NSCLC. The aim of this study was to analyse the clinical data and histological characteristics of indeterminate non-GGO nodules in patients who underwent surgical treatment for NSCLC and to discuss diagnostic and therapeutic approaches to pulmonary nodules coexisting in operable lung cancer.
Patients and methodsTop
Patients
Data of all NSCLC patients who underwent surgical treatment between 1 July 2008 and 30 June 2011 were retrospectively reviewed in order to identify patients presenting with at least one pulmonary nodule coexisting with lung cancer on initial thoracic CT. Patients who met the following inclusion criteria were included in the study: diagnosed with operable NSCLC received surgical therapy; had not undergone peri-operative radiotherapy or chemotherapy; had pulmonary nodules less than 10 mm in diameter on ipsilateral or contralateral lung neoplasms on CT; the nature of the nodules was not determined by CT, but was identified by pathology, or nodules were followed-up for more than 24 months. Patients who met the following exclusion criteria were not included in the study: tumors had a typical GGO, including pure GGO and mixed GGO; had nodules associated with calcification or typical inflammatory nodules; had nodules with SUVmax(standard uptake volume)≥4 on PET-CT; or had nodules that were highly suspected to be metastases.
Methods
All chest examinations were performed using a Somaton Plus 4 scanner (Siemens, Erlagen, Germany). Helical scanning was carried out with a slice thickness of 1.5 mm using the following parameters: 120 kVp, 120–240 mA, and 0.75- or 1-s scan time. CT images were evaluated by 2 thoracic radiologists. In addition to chest CT, all patients underwent ultrasound of the upper abdomen, magnetic resonance imaging of the brain, and whole-body bone scanning to rule out systemic metastasis. PET using 18F-FDG was performed in 28 patients. Four subpleural nodules with uniform density and >5mm in diameter were chosen for biopsy. Twenty nodules located in parenchyma or less than 5mm in diameter were chosen for observation. Postoperative follow-up CT for indeterminate lesions was performed every 3–6 months for over a period of 2 years. A nodule was considered benign when its benignity was histologically proven or when a nodule spontaneously disappeared or was stable in size over a period of at least 24 months. Nodules with a doubling time from 30–360 days were suspected to be malignant [7]. PET-CT, fine-needle biopsy, or surgery was subsequently performed. Patients presenting with several nodules were classified as having malignant nodules if at least one nodule was malignant. Pathologic stage was determined according to the American Joint Committee on Cancer guidelines, seventh edition. However, the nature of the nodules was not taken into account in TNM staging. Multiple primary lung cancer (MPLC) was defined according to the description by Martini and Melamed [8].
Statistical analyses
Statistical analyses for comparisons between groups were realized using Χ² or Mann-Whitney U tests for continuous variables and Fisher’s exact test for proportions. A P<0.05 was considered statistically significant. Statistical analysis was performed using Stat-View 5.0 software (SAS, Inc., Cary, NC, USA).
ResultsTop
Patient characteristics
Between 1 July 2008 and 30 June 2011, 1206 patients with NSCLC underwent surgical treatment in our department. Two hundred fifty-eight (21.4%) patients presented at least one pulmonary nodule coexisting with NSCLC on initial thoracic CT. Forty-five patients (18 men and 27 women, mean age: 59.6 9.2 years) with 60 nodules less than 10 mm in diameter were included in this study. Lobectomy was performed in 21 patients, lobectomy plus wedge resection was performed in 20 patients, and wedge resection was performed in 4 patients. Five patients underwent second operations.
Locations of the nodules
Thirty-two patients presented with 1 nodule, and 13 patients presented with several nodules (8 patients had 2 nodules and 5 patients had 3–5 nodules). There were 27 lesions that measured 5 mm or less in diameter, while 33 lesions measured 6–10 mm. Twenty-one patients had 30 ipsilateral pulmonary nodules to the main tumor (7 nodules in same lobe and another 17 nodules in different lobes), 20 patients had 22 contralateral pulmonary nodules, and 4 patients had 8 bilateral nodules (Tables 1 and 2).
| Characteristics | Group | P-value | ||
| Patients with benign nodules | Patients with malignant nodules | |||
| Number of patients(n) | 20 | 22 | ||
| Number of nodules(n) | 31 | 26 | ||
| Age (years) | 57 ± 7.5 | 61.9 ± 11.1 | 0.11 | |
| Gender (n) | 0.4 | |||
| Male | 9 | 8 | ||
| Female | 11 | 14 | ||
| Tobacco | 0.14 | |||
| Smoker or former-smoker | 7 | 7 | ||
| Nonsmoker | 13 | 15 | ||
| History of other cancer | 0.93 | |||
| Yes | 1 | 2 | ||
| No | 19 | 20 | ||
| Histology | 0.0004* | |||
| Adenocarcinoma | 17 | 21 | ||
| Squamous carcinoma | 3 | 0 | ||
| Other | 0 | 1 | ||
| cTNM | 0.044* | |||
| I | 19 | 14 | ||
| II | 0 | 2 | ||
| III | 1 | 6 | ||
Abbreviations: * = P < 0.05.
| Characteristics | Benign nodules | Malignant nodules | P-value |
| Localization/NSCLC | 0.68 | ||
| Same lobe | 6 | 2 | |
| Other ipsilateral lobe | 15 | 11 | |
| Contralateral lung | 14 | 9 | |
| Size of nodules | 0.04* | ||
| 6–10 mm | 18 | 15 | |
| ≤ 5 mm | 17 | 7 | |
| Shape | 0.003* | ||
| Well limited(n=40) | 30(86%) | 10(45%) | |
| Spiculated(n=17) | 5(14%) | 12(55%) |
Abbreviations: * = P < 0.05
Characteristics of primary cancers
In pathological examination, adenocarcinoma was identified in 36 (80%) cases, adenocarcinoma in situ was identified in 4 cases, squamous carcinoma was identified in 4 patients, and adenosquamous carcinoma was identified in 1 patient. NSCLC staging is shown in Table 3. Twelve patients with advanced NSCLC and 3 patients with stage Ib NSCLC underwent 4 cycles chemotherapy. One patient with stage IIb NSCLC did not receive chemotherapy because of a low PS score. Two patients with stage Ib and one with stage Ia were given treatment with epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor (TKI) treatment for 6 months.
| Stage | Number of patients | Number of nodules | Metastasis | Multiple primary lung cancer |
| Ia | 21 | 27 | 0 | 7 |
| Ib | 11 | 16 | 0 | 2 |
| IIa | 3 | 3 | 1 | 1 |
| IIb | 2 | 3 | 1 | 0 |
| IIIa | 8 | 10 | 5 | 0 |
Nodules characteristics
Thirty-two nodules were identified by pathology. Seventeen nodules were malignant, and 15 nodules were benign. The other 28 were followed-up for a median 30 months. Among the malignant nodules, 7 (41%) presented with the same histology as the primary tumor, and 10 (59%) were of a different histological type (Table 4). Among 28 nodules of undetermined nature, 5 were enlarged during follow-up, including 2 secondary lung cancers identified by second surgeries, 1 metastatic nodule, and 2 diagnosed as malignant nodules by PET-CT. Three nodules were smaller in size or had disappeared. The other 20 nodules showed stability during the 24-month follow-up (Figure 1).
| Histopathology | No. of pulmonary nodules |
| Malignant | |
| Metastases | 7 |
| Second primary cancer | 10 |
| Benign | |
| Fibrosis | 5 |
| Inflammation | >4 |
| Benign tumor | 4 |
| Intrapulmonary lymph node | 2 |
| Total | 32 |
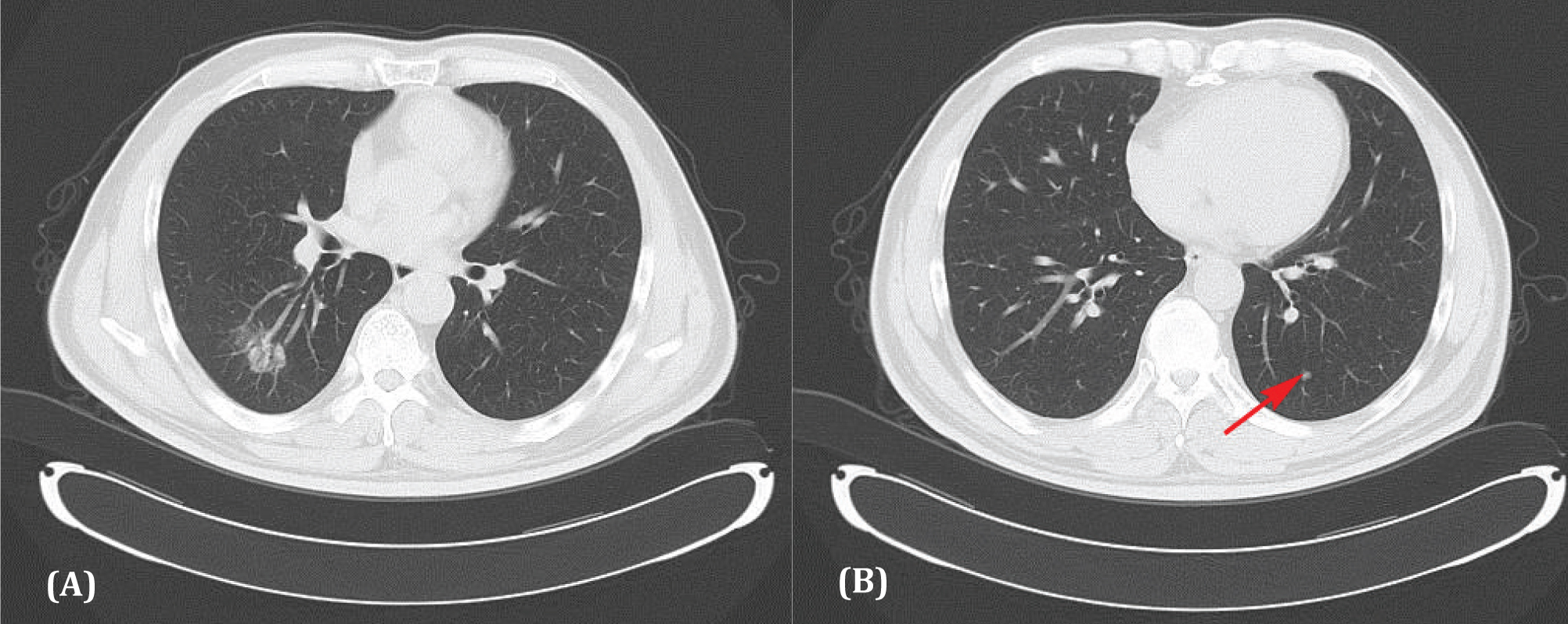
Figure 1 Computer tomogram showing a right lower lobe adenocarcinoma (A) with a small nodule in the left lower lobe (B). No change was observed in the nodule 25 months later.
According to the diagnostic criterion, twenty-six nodules (43%) were malignant, 31 nodules (52%) were benign, and 3 nodules (5%) were of undetermined nature. According to the histological nature of the nodule(s), we classified patients as having malignant, benign, or indeterminate nodule(s). Twenty-two patients (49%) had malignant nodules, and a total of 26 nodules (37%) were identified in these patients. Fourteen patients were diagnosed as having synchronous MPLCs (multiple primary lung cancers), and 8 were diagnosed as having intrapulmonary metastases. Twenty patients (44%) had benign nodule(s), with a total of 35 nodules identified in these patients. Additionally, 3 patients (with 3 nodules) were classified as having nodules of undetermined nature (Table 1). The incidence of metastatic nodules in patients with stage III NSCLC was significantly higher than that in patients with stage I NSCLC (P < 0.01), whereas the incidence of MPLCs in patients with stage I NSCLC was significantly higher than in patients with stage III NSCLC (P < 0.01).
Comparison of the characteristics of patients with benign and malignant nodules
No significant differences in age, gender, smoking status or history of other cancers were noted between patients with malignant and benign nodules. Tumor histology (adenocarcinoma vs. nonadenocarcinoma, p = 0.0004) and TNM stage (advanced stage vs. early stage, p = 0.044) differed significantly between the 2 groups of patients.
Of 33 nodules greater than 5 mm in diameter, 15 nodules were malignant. However, of the 27 nodules less than 5 mm in diameter, only 7 nodules were malignant. The difference between these groups was statistically significant. However, no significant difference was observed in the anatomical localization of the primary NSCLC. Malignant nodules were frequently spiculated [12 (55%) vs. 5 (14%), p = 0.003] (Table 2).
DiscussionTop
In patients with potentially operable lung cancer, the diagnosis of coexisting small nodules of 10 mm or less is critical to avoid insufficient or excessive surgical procedures. In this study, 45 patients with NSCLC coexisting with 60 small non-GGO nodules were enrolled. We identified 3 factors associated with nodule malignancy in NSCLC: tumor histology, tumor stage, and nodule size. Patient age, gender, and tobacco exposure were not associated with malignancy. As expected, tumor histology was a predominant characteristic. A high prevalence of malignant nodules was mainly observed in ADC (adenocarcinoma). These tumors are often multifocal [14]. The associated factors of nodule malignancy vary widely in different studies. Ruppert et al. [15] concluded that tumor histology, localization of the nodules in an upper lobe, colocalization in the same lobe as the primary NSCLC, nodule size, and nodule shape were associated with nodule malignancy. They developed a predictive score to estimate the probability of malignancy of pulmonary nodules in NSCLC. However, in a study by Caretta et al. [10], no significant correlations between nodule malignancy and nodule size, localization with regard to the tumor, tumor histology, or cTNM were detected. Ginsberg and colleagues [16] reported that in patients with known malignancy, nodules smaller than 5 mm were more likely to be benign, whereas those over 5 mm but smaller than 10 mm were more likely to be malignant. In the current study, among the 60 nodules observed, the rate of benign nodules less than 5 mm in diameter was higher than the benign nodules larger than 5 mm in diameter.
The nature of lung cancer-associated nodules less than 10 mm in diameter has been shown to vary greatly. Kim et al. [2] published a study in which 62 patients (44%) had a total of 138 small (≤ 10 mm) nodules in the nonprimary lobes. Of these nodules, 132 (93%) were benign, and only 6 were malignant. Caretta et al. [10] identified 32 nodules in 29 patients who underwent surgery for NSCLC. Thirty-two percent of these nodules were malignant. Similar results were found by Yuan et al. [11], wherein of the 26% of operable NSCLC patients with pulmonary nodules, 25% of the nodules were malignant. In the present study, 43% of the pulmonary nodules associated with lung cancer were malignant, which was higher than that observed in previous studies. However, in line with our study Zwirewich et al. [17] reviewed the pathology of accessory nodules accompanying a dominant nodule in 142 lung malignancies. Thirty-two of 142 (23%) lung malignancies had accompanying accessory nodules. Surprisingly, 25 of 32 (78%) patients had nodules that proved to be malignant. The differences observed may have resulted from variations in inclusion criteria for patients with nodules, as well as the presence of selective bias. In the present study, all of the coexisting nodules evaluated were solid nodules, while previous studies have included both solid and GGO nodules. CT scanners, acquisition parameters, stage of primary tumor, and patient race may have also contributed to the observed differences.
In the present study, the incidence of synchronous MPLC in early stage NSCLC was higher than that in advanced NSCLC. All 10 patients with MPLC were diagnosed with adenocarcinoma. MPLC mostly arose in adenocarcinoma that may coexist with atypical adenomatous hyperplasia (AAH). Most patients in this study had histologically diagnosed adenocarcinoma or adenocarcinoma in situ. Several recent studies have shown that patients with node-negative MPLC have a relative good prognosis, with 5-year survival rates ranging from 69.9% to 83% [18-20]. Therefore, patients with stage I primary lung cancer coexisting with intrapulmonary nodules are recommended to undergo operation, even if histological findings in nodules are not available [21]. On the other hand, the incidence of metastatic nodules in stage Ⅲ NSCLC are significantly higher than that in stage I tumors. When satellite nodules were observed in patients with clinical stage Ⅲ NSCLC, CT-guided biopsy or thoracoscopic surgery is recommended in order to exclude the possibility of metastasis. Otherwise, close follow-up is necessary.
The major limitation of the present study is that it is a retrospective single-center study. In addition, some very small nodules might have been missed because of the partial volume effect. Our subjects were lung cancer patients who had undergone an operation. We did not review CT images of lung cancer patients who did not undergo an operation. Different radiologists have different levels of experience, which may lead to bias. For these reasons, the referral of patients with potentially operable lung cancer may not accurately reflect the general population, and there may be other factors that influence the likelihood of malignancy for coexisting small nodules.
ConclusionTop
Differential diagnosis of indeterminate satellite nodules in patients with operable NSCLC is critical. A relevant number of indeterminate satellite nodules in patients with early stage NSCLC are benign or represent a second primary lung cancer and should not therefore be considered a contraindication to surgical treatment when differential diagnosis cannot be obtained. If nodules larger than 5mm are detected in patients with stage Ⅲ NSCLC, surgery should be performed cautiously until the nodules are found to not be metastatic.
Acknowledgments
We thank all the staff at the Department of Thoracic Surgery at Chinese PLA General Hospital. The clinical portion of this work was supported by Prof. Yu’e Sun, Prof. Naikang Zhou, Dr. Weimin Dai, and Dr. Yunxi Wang.
Conflict of interest
The authors express no conflict of interest.
ReferencesTop
[1] Remy-Jardin M, Remy J, Giraud F, Marquette CH (1993) Pulmonary nodules: detection with thick-section spiral CT versus conventional CT. Radiology 187:513–520. Article Pubmed
[2] Kim YH, Lee KS, Primack SL, Kim H, Kwon OJ, et al. (2002) Small pulmonary nodules on CT accompanying surgically resectable lung cancer: likelihood of malignancy. J Thorac Imaging 17:40–46. Article Pubmed
[3] Torrington KG, Kern JD (1993) The utility of fiberoptic bronchoscopy in the evaluation of the solitary pulmonary nodule. Chest 104:1021–1024. Article Pubmed
[4] Nomori H, Watanabe K, Ohtsuka T, Naruke T, Suemasu K, et al. (2004) Evaluation of F-18 fluorodeoxyglucose (FDG) PET scanning for pulmonary nodules less than 3 cm in diameter, with special reference to the CT images. Lung Cancer 45:19–27. Article Pubmed
[5] Li H, Boiselle PM, Shepard JO, Trotman-Dickenson B, McLoud TC (1996) Diagnostic accuracy and safety of CT-guided percutaneous needle aspiration biopsy of the lung: comparison of small and large pulmonary nodules. AJR Am J Roentgenol 167:105–109. Article Pubmed
[6] Groome PA, Bolejack V, Crowley JJ, Kennedy C, Krasnik M, et al. (2007) The IASLC lung cancer staging project: validation of the proposals for revision of the T N, and M descriptors and consequent stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J Thorac Oncol 2:694–705. Article Pubmed
[7] Henschke CI, Yankelevitz DF, Yip R, Reeves AP, Farooqi A, et al. (2012) Lung Cancers Diagnosed at Annual CT Screening: Volume Doubling Times. Radiology 263:578–583. Article Pubmed
[8] Martini N, Melamed MR (1975) Multiple primary lung cancers. J Thorac Cardiovasc Surg 70:606–612. Article Pubmed
[9] Libby DM, Smith JP, Altorki NK, Pasmantier MW, Yankelevitz D, et al. (2004) Managing the small pulmonary nodule discovered by CT. Chest 125:1522–1529. Article Pubmed
[10] Carretta A, Ciriaco P, Canneto B, Nicoletti R, Del Maschio A, et al. (2002) Therapeutic strategy in patients with non-small cell lung cancer associated to satellite pulmonary nodules. Eur J Cardiothorac Surg 21:1100–1104. Article Pubmed
[11] Yuan Y, Matsumoto T, Hiyama A, Miura G, Tanaka N, et al. (2003) The probability of malignancy in small pulmonary nodules coexisting with potentially operable lung cancer detected by CT. Eur Radiol 13:2447–2453. Article Pubmed
[12] Lee SW, Leem CS, Kim TJ, Lee KW, Chung JH, et al. (2013) The long-term course of ground-glass opacities detected on thin-section computed tomography. Respir Med 107:904–910. Article Pubmed
[13] Lee HJ, Goo JM, Lee CH, Yoo CG, Kim YT, et al. (2007) Nodular ground-glass opacities on thin-section CT: size change during follow-up and pathological results. Korean J Radiol 8:22–31. Article Pubmed
[14] Chang YL, Wu CT, Lee YC (2007) Surgical treatment of synchronous multiple primary lung cancers: Experience of 92 patients. J Thorac Cardiovas Surg 134:630–637. Article Pubmed
[15] Ruppert AM, Lerolle U, Carette MF, Lavole A, Khalil A, et al. (2011) Coexisting pulmonary nodules in operable lung cancer: Prevalence and probability of malignancy. Lung Cancer 74:233–238. Article Pubmed
[16] Ginsberg MS, Akin O, Berger DM, Zakowski MF, Panicek DM (2004) Pulmonary tumorlets: CT findings. AJR Am J Roentgenol 183:293–296. Article Pubmed
[17] Zwirewich CV, Miller RR, Müller NL (1990) Multicentric adenocarcinoma of the lung: CT-pathologic correlation. Radiology 176:185–190. Article Pubmed
[18] Yu YC, Hsu PK, Yeh YC, Huang CS, Hsieh CC, et al. (2013) Surgical results of synchronous multiple primary lung cancers: similar to the stage-matched solitary primary lung cancers Ann Thorac Surg 96:1966–1974. Article Pubmed
[19] Kushibe K, Kawaguchi T, Nishimoto Y, Takahama M, Tojo T, et al. (2006) Operative indications for lung cancer with satellite lesions. Asian Cardiovasc Thorac Ann 14:316–320. Article Pubmed
[20] Okada M, Tsubota N, Yoshimura M, Miyamoto Y (1998) Operative approach for multiple primary lung carcinomas. J Thorac Cardiovasc Surg 115:836–840. Article Pubmed
[21] Yoon HI, Yim JJ, Lee CT, Kim YW, Han SK, et al. (2006) Clinical characteristics of a second small nodule(s) associated with cT1-2N0M0 non-small-cell lung cancer. Lung 184:273–278. Article Pubmed



