Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 1, Issue 8, October 2013, Pages 201-207
Original researchOpen Access
Impact of initial time to prostate-specific antigen nadir on survival in prostate cancer with bone metastasis initially treated with maximum androgen blockade therapy
-
Yamamoto Y1,
Nozawa M1,*
 ,
Itami Y1,
Kobayashi Y1,
Saito Y1,
Shimizu N1,
Minami T1,
Hayashi T1,
Tsuji H1
Yoshimura K1
Ishii T1 and
Uemura H1
,
Itami Y1,
Kobayashi Y1,
Saito Y1,
Shimizu N1,
Minami T1,
Hayashi T1,
Tsuji H1
Yoshimura K1
Ishii T1 and
Uemura H1
- 1 Department of Urology, Kinki University Faculty of Medicine, 377-2 Ohno-Higashi, Osaka-Sayama, 589-8511, Japan
*Corresponding author: Masahiro Nozawa, Department of Urology, Kinki University, Faculty of Medicine, 377-2 Ohno-Higashi, Osaka-Sayama, 589-8511, Japan, Tel.: +81-72-366-0221; Fax: +81-72-365-6273. E-mail: nozawa06@med.kindai.ac.jp
Received 10 June 2013 Revised 30 August 2013 Accepted 15 September 2013 Published 22 September 2013
DOI: http://dx.doi.org/10.14312/2052-4994.2013-30.
Copyright: © 2013 Yamamoto Y, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Background: The objective of this study is to provide certain data on clinical outcomes and their predictors of traditional maximum androgen blockade (MAB) in prostate cancer with bone metastasis. Methods: Subjects were patients with prostate adenocarcinoma with bone metastasis initiated to treat with MAB as a primary treatment without any local therapy at our hospital between January 2003 and December 2010. Time to prostate specific antigen (PSA) progression, overall survival (OS) time, and association of clinical factors and outcomes were retrospectively evaluated. Results: A total of 57 patients were evaluable. The median age was 70 years. The median primary PSA was 203 ng/ml. Luteinizing hormone-releasing hormone agonists had been administered in 96.5% of the patients. Bicalutamide had been chosen in 89.4 % of the patients as the initial antiandrogen. The median time to PSA progression with MAB was 11.3 months (95% confidence interval [CI], 10.4 to 13.0). The median OS was 47.3 months (95% CI, 30.7 to 81.0). Gleason score 9 or greater, decline of PSA level equal to or higher than 1.0 ng/ml with MAB, and time to PSA nadir equal to or shorter than six months after initiation of MAB were independent risk factors for time to PSA progression (P=0.010, P=0.005, and P=0.001; respectively). Time to PSA nadir longer than six months was the only independent predictor for longer OS (HR, 0.255 [95% CI, 0.109 to 0.597]; P=0.002). Conclusions: Initial time to PSA nadir should be emphasized for clinical outcome analyses in future studies on prostate cancer with bone metastasis.
Keywords: androgen-deprivation therapy; bone metastasis; maximum androgen blockade; prognostic factor; prostate cancer; prostate-specific antigen
IntroductionTop
Androgen deprivation therapy (ADT) consisting of a luteinizing hormone-releasing hormone (LH-RH) agonist or surgical castration with or without an antiandrogen is the gold standard as initial systemic treatment for patients with metastatic prostate cancer [1] . Recently, several new agents that act on the androgen pathway have shown clinical benefits for patients whose prostate cancer progressed after initial ADT. For example, abiraterone acetate, which blocks androgen biosynthesis by inhibiting CYP17, has demonstrated improvement on overall survival (OS) in patients with metastatic castration-resistant prostate cancer (CRPC) that progressed after docetaxel-based chemotherapy in combination with low dose prednisone [2] . Abiraterone acetate has also shown a significant improvement in radiological progression-free survival (PFS) and OS in patients with chemotherapy-naïve metastatic CRPC [3] . There is another androgen biosynthesis blocker, called TAK-700 [4, 5]. Two large global phase III trials on this agent are now ongoing for patients with metastatic CRPC, one of which targets the stage after docetaxel failure and the other in a chemotherapy-naïve setting. Enzalutamide is the so-called second-generation antiandrogen which blocks not only binding of testosterone with androgen receptors (AR) but also nuclear translocation and DNA binding of AR [6] . Enzalutamide has prolonged overall survival in patients with post-docetaxel CRPC based on a placebo-controlled randomized study [7] .
These new agents mentioned above may be proven to be beneficial in the earlier stages of prostate cancer in the near future. Moreover, degarelix, an LH-RH antagonist, has achieved statistically significant prostate specific antigen (PSA) progression-free survival benefit over leuprolide, a LH-RH agonist [8-10]. In this situation, it is important to precisely evaluate the clinical efficacy of ADT consisting of traditional maximum androgen blockade (MAB) at this time. There have been several reports of outcomes from patients with prostate cancer treated with ADT [11-27]; however, the number of studies, that reported outcomes in patients with bone metastatic prostate cancer treated primarily with MAB without any local treatment, is limited. Recently, outcomes of MAB as an initial treatment for patients with bone metastatic prostate cancer have been reported, in which the significant impact of PSA kinetics not on OS but on disease progression was demonstrated [16] .
The objective of the current study is to provide certain data on the clinical efficacy of ADT by traditional MAB as initial systemic therapy for bone metastatic prostate cancer and to evaluate the impact of biomarkers on their clinical outcomes, which will be useful in development of future clinical study protocols.
Patients and methodsTop
Patients with bone metastatic prostate cancer, who had been initially treated with ADT, consisting of MAB, without any local therapy at Kinki University Hospital between January 2003 and December 2010 were retrospectively investigated. Patients with an Eastern Cooperative Oncology Group performance status of 0 or 1 at diagnosis were eligible for this study. Patients who had received any bisphosphonate or denosumab before disease progression with initial ADT were excluded.
The primary study endpoint was time to PSA progression with initial ADT, defined as the day of the first time of a consecutive three-time rise of serum PSA level after confirming the nadir. The secondary endpoint was OS. Baseline patient characteristics and PSA response and kinetics after initiation of ADT were evaluated for their impacts on time to PSA progression and OS. Time to PSA progression and OS were calculated by Kaplan-Meier method. Univariate and multivariate analyses were performed using the Cox’s proportional hazards regression model for evaluation of biomarkers to predict time to PSA progression and OS. P-value below 0.05 was considered to be significant. SPSS version 18.0 (SPSS, Chicago, IL, USA) was used for all statistical analyses.
ResultsTop
Patient characteristics
A total of 57 patients met the eligibility criteria and were evaluable for this analysis. Bone metastasis had been diagnosed with a bone scan and, in case of doubt, confirmed with magnetic resonance imaging or computed tomography (CT) for all patients included in this analysis. CT scan from chest to pelvic cavity had been also performed for each patient to detect other metastasis. No patients evaluated underwent biopsy of metastatic sites. The median follow-up period was 44.6 months (range, 6.0–99.6) at the evaluation. Table 1 shows the patient characteristics. The median age of the cohort at diagnosis was 70 (range, 41–90). The median serum primary PSA concentration was 203 ng/ml (range, 6.7–8,546). Seventy-nine percent of the patients had a Gleason score 8 or greater at baseline prostate biopsy. The extent of bone metastasis was evaluated according to the extent of disease (EOD) score. Thirty-four (59.6%) patients had EOD score of 2 or higher. Regional lymph node metastasis was observed in 61.5 % of the patients, three out of whom had also metastasis to the para-aortic lymph node. Pulmonary metastasis was detected in two patients.
| Characteristics | Values in number (%) |
| No. of patients | 57 |
| Median age at diagnosis (range) | 70 years (41–90 years) |
| Primary PSA (range) | 203 ng/ml (6.7–8546 ng/ml) |
| Baseline Gleason score | |
| 6 or less | 5 (8.7) |
| 7 | 5 (8.7) |
| 8 | 10 (17.5) |
| 9 or 10 | 35 (61.4) |
| Unknown | 2 (3.5) |
| Baseline EOD score | |
| 1 | 23 (40.3) |
| 2 | 19 (33.3) |
| 3 | 8 (14.0) |
| 4 | 7 (12.3) |
| Lymph node metastasis | |
| Regional | 35 (61.5) |
| Para-aorta | 3 (5.3) |
| Pulmonary metastasis | 2 (3.5) |
| Initial systemic therapy | |
| Surgical castration | 2 (3.5) |
| LH-RH agonist | |
| Leuprolide | 39 (68.4) |
| Goserelin Acetate | 16 (28.1) |
| Antiandrogen | |
| Bicalutamide | 51 (89.4) |
| Chlormadinone Acetate | 4 (7) |
| Flutamide | 2 (3.5) |
| Salvage systemic therapy after disease progression | |
| Alternative antiandrogen therapy | 31 (54.4) |
| Dexamethasone | 35 (61.4) |
| Estramustine phosphate | 33 (57.8) |
| Docetaxel | 28 (49.1) |
| Cisplatin | 4 (7) |
Abbreviations: PSA = prostate specific antigen; LH-RH = Luteinizing hormone-releasing hormone; EOD = extent of disease
LH-RH agonists had been administered in 96.5% of the patients, two thirds or more of whom had received leuprolide, in initial ADT. Bicalutamide had been chosen in 89.4 % of the patients as the initial antiandrogen. Anti-androgens had been administered from one to two weeks before LH-RH agonist administration. Patients who had undergone surgical castration had received anti-androgens simultaneously. Eighty-eight percent of the patients had experienced disease progression with initial ADT at the time of analysis. Alternative antiandrogen therapy had been adopted in 54.4% of the patients as the second-line treatment. Dexamethasone or estramustine phosphate had been used in 61.4% or 57.8% of the patients, respectively, as the second- or third-line hormone therapy with or without docetaxel. Overall 49.1% of the patients had been treated with a docetaxel-containing regimen as the first-line chemotherapy. Four patients (7.0%) had received cisplatine-based chemotherapy after failure to treatment with docetaxel-containing regimens. Palliative radiation therapy against bone metastases had been performed in 14 patients (24.6%, data not shown).
Time to prostate specific antigen progression with initial androgen deprivation therapy
The median time to PSA progression with initial ADT was 11.3 months (95% confidence interval [CI], 10.4 to 13.0, Figure 1).
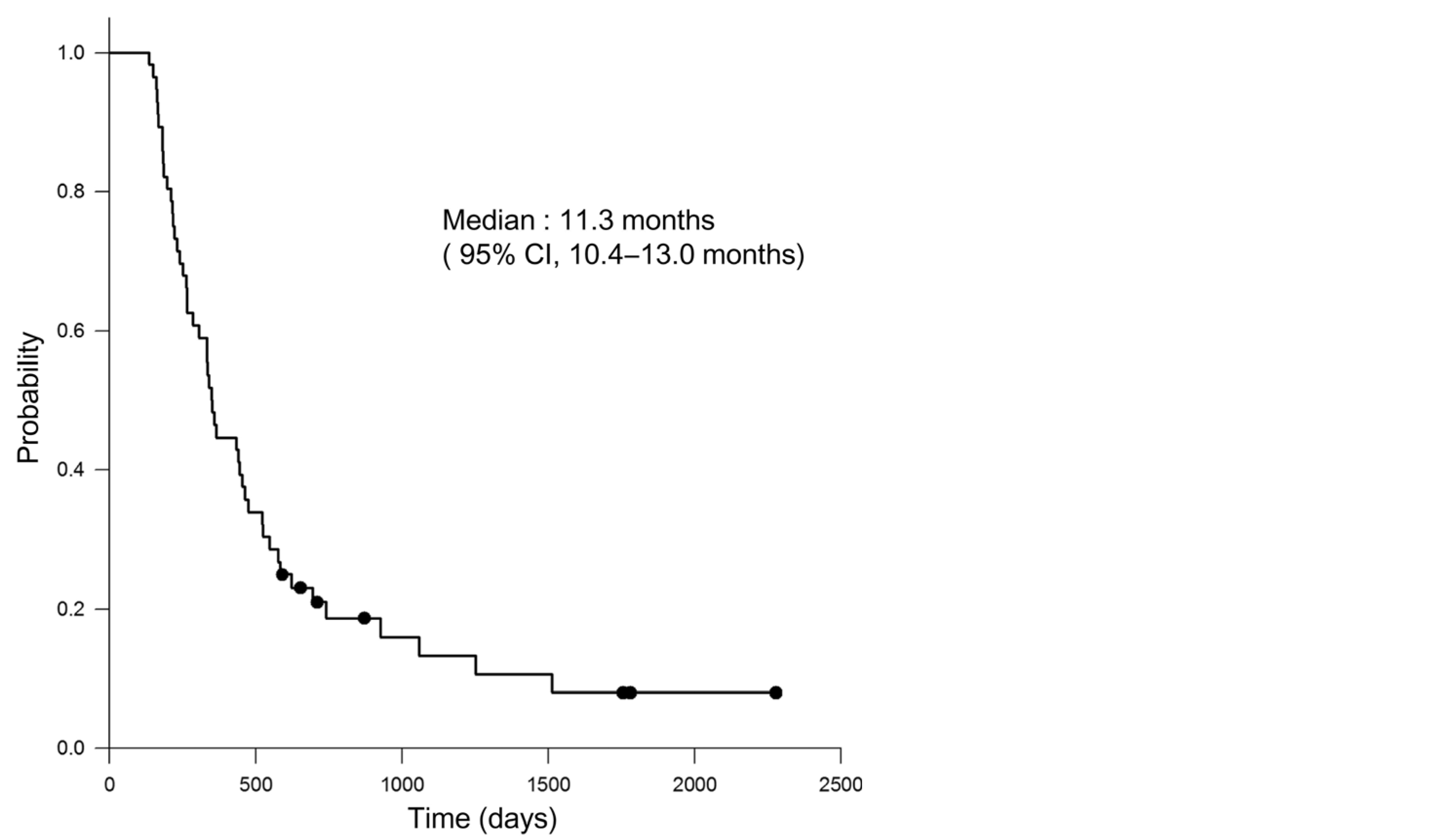
Figure 1 Kaplan-Meier analysis of the time to prostate-specific antigen progression after initiation of androgen-deprivation therapy.
As shown in Table 2, the age at diagnosis, the serum primary PSA concentration, the baseline EOD score and the status of lymph node metastasis were not associated with time to PSA progression. Baseline Gleason score 9 or greater had a significant impact on time to PSA progression (hazard ratio [HR], 3.614 [95% CI, 1.356 to 9.624]; P=0.010) based on the multivariate analysis. Existence of component of Gleason grade 5 had no significance on time to PSA progression (data not shown).
| Variable | Univariate | Multivariate | ||
| HR (95% CI) | p value | HR (95% CI) | p value | |
| Age at diagnosis | ||||
| ≤70 | referent | referent | ||
| >70 | 1.350 (0.716–2.547) | 0.354 | 1.381 (0.738–2.691) | 0.312 |
| Primary PSA | ||||
| ≤203ng/ml | referent | referent | ||
| >203ng/ml | 1.840 ( 0.992–3.414) | 0.053 | 1.854 ( 1.004–3.419) | 0.051 |
| Baseline Gleason score | ||||
| ≤8 | referent | referent | ||
| ≥9 | 3.282 (1.217–8.853) | 0.019 | 3.614 (1.356–9.624) | 0.01 |
| Baseline EOD score | ||||
| 1 | referent | referent | ||
| ≥2 | 1.963 (1.102–3.498) | 0.022 | 1.290( 0.879–3.475) | 0.062 |
| Lymph-node metastasis | ||||
| Yes | referent | referent | ||
| No | 0.756 (0.426–1.340) | 0.339 | 1.094( 0.539–2.220) | 0.803 |
| PSA decline less than 1.0ng/ml with initial ADT | ||||
| Yes | referent | referent | ||
| No | 2.394 (1.243–4.611) | 0.009 | 2.527 (1.320–4.826) | 0.005 |
| Time to nadir PSA with initial ADT | ||||
| ≤6months | referent | referent | ||
| >6months | 0.102 ( 0.046–0.227) | < 0.001 | 0.105 ( 0.047–0.234 ) | < 0.001 |
Abbreviations: CI = confidence interval; HR = hazard ratio; PSA = prostate specific antigen; ADT = androgen-deprivation therapy; EOD = extent of disease
Decline in serum PSA level in any degree had been detected in all of the patients with the initial ADT, whereas 38 (66.7%) patients had achieved PSA decline less than 1.0 ng/ml. Patients who did not achieve PSA decline less than 1.0 ng/ml with initial ADT had a significant risk of early time to PSA progression (HR, 2.527 [95% CI, 1.320 to 4.826]; P=0.005).
The median time to reaching nadir levels of serum PSA with initial ADT was 5.9 months (95% CI, 5.6 to 6.2) for all patients (data not shown). Time to PSA nadir more than six months was the strongest independent predictor for longer time to PSA progression based on the multivariate analysis (HR, 0.105 [95% CI, 0.047 to 0.234]; P<0.001).
Overall survival
Thirty patients (52.6%) had died at the time of analysis. Out of 30 patients died, 26 (87%) had died of the prostate cancer, three other cancers, and one pneumonitis. The median OS was 47.3 months (95% CI, 30.7 to 81.0, Figure 2). Baseline patient characteristics including the age at diagnosis, the primary PSA concentration, Gleason score, EOD score, and the status of lymph node metastasis were not associated with OS based on multivariate analysis (Table 3). Docetaxel-based chemotherapy also did not have a significant impact on OS.
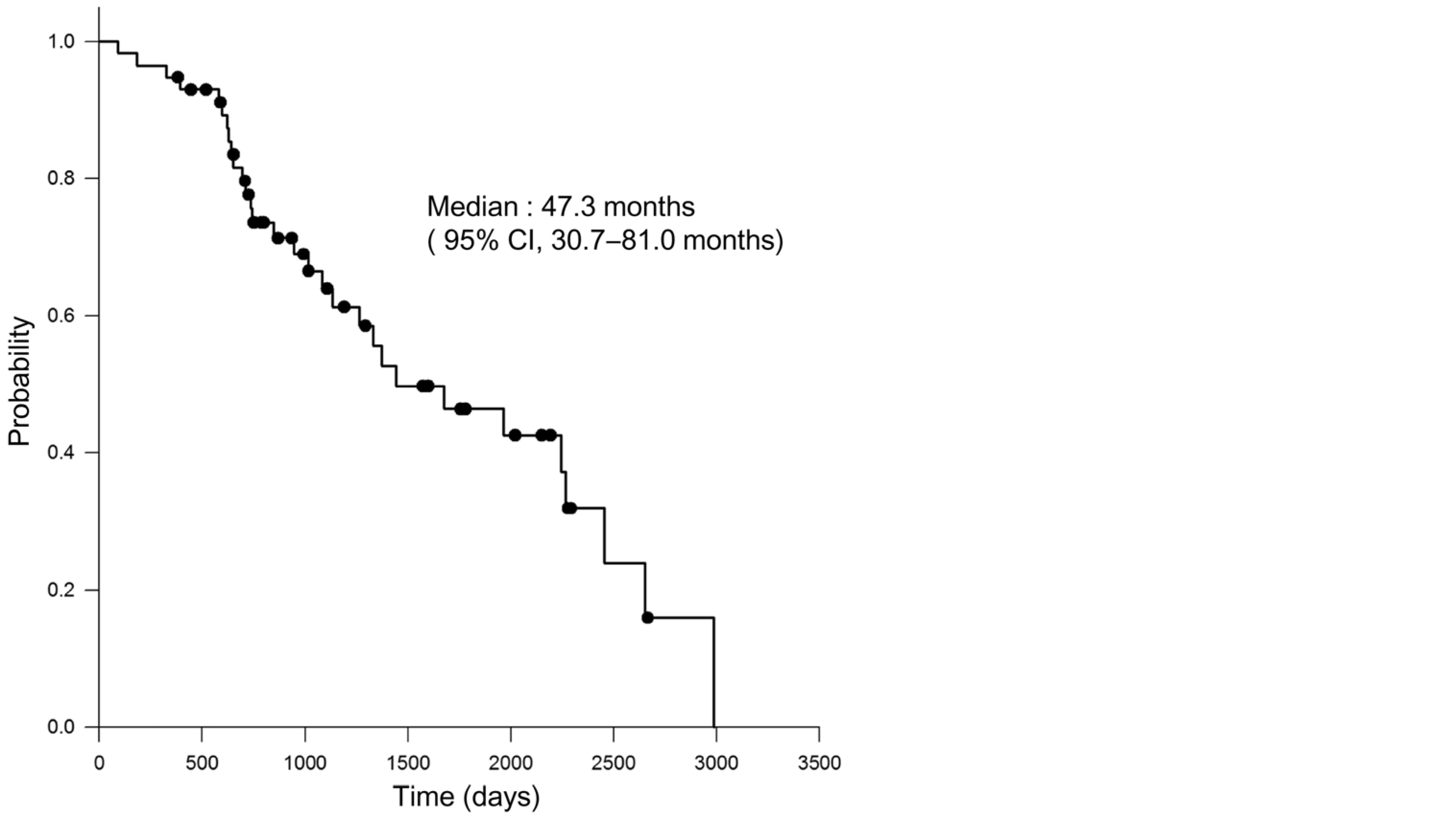
Figure 2 Kaplan-Meier analysis of overall survival.
| Variable | Univariate | Multivariate | ||
| HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Age at diagnosis | ||||
| ≤70 | referent | referent | ||
| >70 | 1.681 (0.699–4.043) | 0.246 | 1.701 (0.704–4.113) | 0.225 |
| Primary PSA | ||||
| ≤203ng/ml | referent | referent | ||
| >203ng/ml | 1.886 (0.823 – 4.321) | 0.134 | 1.935 (0.872–4.483) | 0.112 |
| Baseline Gleason score | ||||
| ≤8 | referent | referent | ||
| ≥9 | 0.531 (0.205–1.375) | 0.193 | 0.556 (0.231–1.403) | 0.131 |
| Baseline EOD score | ||||
| 1 | referent | referent | ||
| ≥2 | 2.502 (1.194–5.244) | 0.015 | 2.290(0.768–6.825) | 0.087 |
| Lymph-node metastasis | ||||
| Yes | referent | referent | ||
| No | 0.794 (0.399–1.580 | 0.518 | 1.094(0.539–2.220) | 0.803 |
| Docetaxel-based chemotherapy | ||||
| Yes | referent | referent | ||
| No | 1.528 (0.752–3.103) | 0.242 | 1.494(0.771–2.096 ) | 0.776 |
| PSA decline less than 1.0ng/ml with initial ADT | ||||
| Yes | referent | referent | ||
| No | 2.082 (0.870–4.984) | 0.1 | 2.217 (0.996–4.931) | 0.051 |
| Time to nadir PSA with initial ADT | ||||
| ≤6months | referent | referent | ||
| >6months | 0.252 (0.104–0.615) | 0.002 | 0.255 (0.109–0.597 | 0.002 |
Abbreviations: CI = confidence interval; HR = hazard ratio; PSA = prostate specific antigen; ADT = androgen-deprivation therapy; EOD = extent of disease
PSA decline less than 1.0 ng/ml with initial ADT did not significantly affect OS (HR, 2.27 [95% CI, 0.996 to 4.931]; P=0.051). Time to PSA nadir more than six months with initial ADT independently predicted longer OS with HR of 0.255 (95% CI, 0.109 to 0.597; P=0.002) based on multivariate analysis.
DiscussionTop
The median time to PSA progression and OS were 11.3 months (95% CI, 10.4 to 13.0) and 47.3 months (95% CI, 30.7 to 81.0), respectively for patients with prostate cancer with bone metastasis initially treated with MAB based on the present study. According to large-scale prospective trials, the median time to disease progression (TTP) and OS was reported as 16.5 to 22.3 months and 33.5 to 41.4 months, respectively for patients with advanced prostate cancer treated with ADT consisted of combination of LH-RH agonist or bilateral orchiectomy and an antiandrogen [17, 18]. However, subjects in these trials included patients with prostate cancer locally-advanced but non-metastatic, metastatic but not-bony, or metastatic after radical local treatments. The current study definitely provides the outcomes of MAB without any local treatment as the initial therapy for patients with bone metastatic prostate cancer.
Our result on time to PSA progression compares with findings on TTP previously reported, because it is estimated to take 6 to 12 months from PSA progression to the occurrence of radiologically detectable new lesions [28, 29]. Our result on OS may be rather superior to previous reports [17, 18] and the reason for that may be that 49% of the patients received docetaxel after failure to ADT in our study whereas few patients were treated with docetaxel in the past studies, though docetaxel treatment did not have a significant impact on OS in the present study. It has been reported that progressive bone disease was one of poor prognostic factors for disease control in patients with metastatic CRPC treated with docetaxel [30] . These findings suggest that the efficacy of docetaxel may be limited in terms of controlling bone disease in metastatic CRPC patients.
Based on the current study, the baseline Gleason score had a significant impact not on OS but on time to PSA progression for patients with bone metastatic prostate cancer treated with MAB. Association between Gleason score and TTP was also reported in a previous study [19] . On the other hand, the impact of Gleason score on OS is controversial; some reported a positive relationship between them [11] and others negative [12, 15, 20, 21].
The primary PSA level did not affect time to PSA progression nor OS in the present study. A correlation between the primary PSA level and TTP was reported in patients with hormone-naïve non-metastatic prostate cancer treated with ADT [22] . In patients with bone metastatic hormone-sensitive prostate cancer, the association between primary PSA level and TTP was also disputable. Some reported a positive association between them [12, 14] and others negative [13, 15]. However, no correlation has been reported between primary PSA level and OS [11-16]. Furthermore, in studies including patients with locally-advanced and metastatic prostate cancer, the primary PSA level did not independently predict TTP nor OS [11, 21, 23]. These findings suggest the primary PSA level may not affect OS in patients with prostate cancer treated with ADT.
Our result suggested PSA decline under 1.0 ng/ml after MAB can predict long-term PSA control by MAB. The nadir PSA level less than 0.2 ng/ml has been reported as an independent predictor for the longer disease control period in patients with bone metastatic hormone-sensitive prostate cancer treated with [16] . PSA decline to 1.0 ng/ml or less was reported to be associated with longer OS in prostate cancer patients treated with flutamide monotherapy [24] . In a phase III study in which patients were treated with LH-RH agonist with or without bicalutamide, whose PSA declined under 4 ng/ml within 12 weeks after initiation of treatment predicted favorable OS [25] . These findings suggest the importance of PSA response to the treatment in disease control for prostate cancer treated with ADT. However, the impact of PSA kinetics after treatment on patient outcome is debatable. Some studies reported that rapid decrease of PSA level was associated with longer progression-free survival [26, 27], and others suggested the opposite [11, 14, 16, 21]. Matzkin et al. reported that patients reaching their nadir PSA values up to six months after treatment began had a trend toward a shorter PFS compared with those reaching their lowest values at nine and 12 months [14] . Sasaki et al. reported that longer time to PSA nadir (>8 months) was an independent good prognostic factor for disease progression [16] . Based on the current study, the time to nadir PSA longer than six months had the most significant impact on longer time to PSA progression and was the only independent predictor investigated for longer OS. These findings suggest that the time to nadir PSA may influence on TTP and OS in patients with prostate cancer treated with ADT. The mechanism underlying this clinical observation is still unclear. It is speculated that a rapid decrease of PSA level from the initiation of ADT reflects rapid removal of hormone-sensitive prostate cancer cells, which may induce an adequate environment for the growth of hormone-resistant prostate cancer cells. Rapid decrease of PSA level after ADT may be due to ablation of androgen-receptor (AR) function, and the quick suppression of AR during ADT may have a negative effect on disease progression, because AR can act as a tumor suppressor for prostate cancer cells [31, 32]. Androgen deprivation in androgen dependent prostate cancer cells results in early G1 arrest. It is possible that two types of prostate cancer cells may exist; one has the character of differentiating to CRPC during cell cycle arrest with rapid reduction of PSA induced by ablation of AR function, and the other gradually goes to apoptosis after cell cycle arrest with slow reduction of PSA. The former would be rich in the shorter time to nadir PSA group, and the latter in the longer time to nadir PSA group [33] .
Baseline EOD score has been reported as a prognostic factor for disease progression in patients with bone metastatic hormone-sensitive prostate cancer treated with ADT [13, 16]. The 2-year survival rates have been reported, according to the EOD score from 1 to 4, to be 94%, 74%, 68%, and 40%, respectively [34] . The EOD score did not independently affect time to PSA progression nor OS in the present study, though there were trends for better treatment outcomes in patients with EOD score of 1. This result may come from the relatively small number of patients in the present study.
There are some limitations in the present study including its retrospective nature and the relatively small number of patients analyzed. Serum testosterone levels had not been measured. It has been suggested that the serum testosterone level might determine the aggressiveness of prostate cancer and the lower serum testosterone level at baseline might predict the worse treatment outcomes in patients treated with MAB [35, 36]. The variety of treatments after PSA progression may influence OS time. However, treatments after failure to primary ADT often vary according to disease status including the extent of objective lesions and the condition of patients’ symptoms. Because of that, our findings rather reflect the treatment outcomes in the real-world clinic.
ConclusionTop
We reported the clinical outcomes of patients with prostate cancer with bone metastasis initially treated with MAB. Baseline Gleason score less than 9, PSA decline under 1.0 ng/ml after initiation of treatment, and time to PSA nadir longer than six months independently predicted longer time to PSA progression. Time to PSA nadir longer than six months had significant impact on OS time. Our findings suggest time to PSA nadir should be emphasized on for clinical outcome analyses in future studies.
Acknowledgments
We would like to thank Mr. Marco A. De Velasco for his assistance in proofreading the manuscript.
Conflict of interest
The authors have no conflicts of interest to disclose.
ReferencesTop
[1] Mohler JL, Armstrong AJ, Bahnson RR, Boston B, Busby JE, et al. (2012) Prostate cancer, Version 3.2012: featured updates to the NCCN guidelines. J Natl Compr Canc Netw 10:1081-1087. Article Pubmed
[2] de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, et al. (2011) Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med 364:1995-2005. Article Pubmed
[3] Ryan CR, Smith MR, De Bono JS, Molina A, Logothetis C, et al. (2012) Interim analysis (IA) results of COU-AA-302, a randomized, phase III study of abiraterone acetate (AA) in chemotherapy-naive patients (pts) with metastatic castration-resistant prostate cancer (mCRPC). ASCO Annual Meeting. J Clin Oncol 30. Article
[4] Kaku T, Hitaka T, Ojida A, Matsunaga N, Adachi M, et al. (2011) Discovery of orteronel (TAK-700), a naphthylmethylimidazole derivative, as a highly selective 17,20-lyase inhibitor with potential utility in the treatment of prostate cancer. Bioorg Med Chem 19:6383-6399. Article Pubmed
[5] Yamaoka M, Hara T, Hitaka T, Kaku T, Takeuchi T, et al. (2012) Orteronel (TAK-700), a novel non-steroidal 17,20-lyase inhibitor: effects on steroid synthesis in human and monkey adrenal cells and serum steroid levels in cynomolgus monkeys. J Steroid Biochem Mol Biol 129:115-128. Article Pubmed
[6] Tran C, Ouk S, Clegg NJ, Chen Y, Watson PA, et al. (2009) Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 324:787-790. Article Pubmed
[7] Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, et al. (2012) Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 367:1187-1197. Article Pubmed
[8] Klotz L, Boccon-Gibod L, Shore ND, Andreou C, Persson BE, et al. (2008) The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int 102:1531-1538. Article Pubmed
[9] Tombal B, Miller K, Boccon-Gibod L, Schröder F, Shore N, et al. (2010) Additional analysis of the secondary end point of biochemical recurrence rate in a phase 3 trial (CS21) comparing degarelix 80 mg versus leuprolide in prostate cancer patients segmented by baseline characteristics. Eur Urol 57:836-842. Article Pubmed
[10] Crawford ED, Tombal B, Miller K, Boccon-Gibod L, Schröder F, et al. (2011) A phase III extension trial with a 1-arm crossover from leuprolide to degarelix: comparison of gonadotropin-releasing hormone agonist and antagonist effect on prostate cancer. J Urol 186:889-897. Article Pubmed
[11] Choueiri TK, Xie W, D'Amico AV, Ross RW, Hu JC, et al. (2009) Time to prostate-specific antigen nadir independently predicts overall survival in patients who have metastatic hormone-sensitive prostate cancer treated with androgen-deprivation therapy. Cancer 115:981–987. Article Pubmed
[12] Figg WD, Franks ME, Venzon D, Duray P, Cox MC, et al. (2004) Gleason score and pretreatment prostate-specific antigen in survival among patients with stage D2 prostate cancer. World J Urol 22:425-430. Article Pubmed
[13] Matzkin H, Soloway MS, Schellhammer PF, Chodak G, Smith JA, et al.(1993) Prognostic factors in stage D2 prostate cancer treated with a pure nonsteroidalantiandrogen. Cancer 72:1286-1290. Article Pubmed
[14] Matzkin H, Eber P, Todd B, van der Zwaag R, Soloway MS (1992) Prognostic significance of changes in prostate-specific markers after endocrine treatment of stage D2 prostatic cancer. Cancer 70:2302-2309. Article Pubmed
[15] Hussain M, Tangen CM, Higano C, Schelhammer PF, Faulkner J, et al. (2006) Absolute prostate-specific antigen value after androgen deprivation is a strong independent predictor of survival in new metastatic prostate cancer: data from Southwest Oncology Group Trial 9346 (INT-0162). J Clin Oncol 24:3984-3990. Article Pubmed
[16] Sasaki T, Onishi T, Hoshina A(2012) Cutoff value of time to prostate-specific antigen nadir is inversely correlated with disease progression in advanced prostate cancer. Endocr Rel at Cancer 19:725-730. Article Pubmed
[17] Crawford ED, Eisenberg M, McLeod DG, Spaulding JT, Benson R, et al. (1989) A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N Engl J Med. 321:419-424. Article
[18] Eisenberger MA, Blumenstein BA, Crawford ED, Miller G, McLeod DG, et al. (1998) Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N Engl J Med 339:1036-1042. Article Pubmed
[19] Benaim EA, Pace CM, Lam PM, Roehrborn CG (2002) Nadir prostate-specific antigen as a predictor of progression to androgen-independent prostate cancer. Urology 59:73–78. Article Pubmed
[20] Kwak C, Jeong SJ, Park MS, Lee E, Lee SE (2002) Prognostic significance of the nadir prostate specific antigen level after hormone therapy for prostate cancer. J Urol. 168:995–1000. Article Pubmed
[21] Morote J, Trilla E, Esquena S, Abascal JM, Reventos J (2004)Nadir prostate-specific antigen best predicts the progression to androgen-independent prostate cancer. Int J Cancer 108:877–881. Article Pubmed
[22] Ross RW, Xie W, Regan MM, Pomerantz M, Nakabayashi M, et al. (2008) Efficacy of androgen deprivation therapy (ADT) in patients with advanced prostate cancer: association between Gleason score, prostate-specific antigen level, and prior ADT exposure with duration of ADT effect. Cancer 112:1247-1253. Article Pubmed
[23] Huang SP, Bao BY, Wu MT, Choueiri TK, Goggins WB, et al. (2011) Impact of prostate-specific antigen (PSA) nadir and time to PSA nadir on disease progression in prostate cancer treated with androgen-deprivation therapy. Prostate 71:1189–1197. Article Pubmed
[24] Collette L, de Reijke TM, Schröder FH; EORTC Genito-Urinary Group (2003) Prostate specific antigen: a prognostic marker of survival in good prognosis metastatic prostate cancer? (EORTC 30892). Eur Urol 44:182-189. Article Pubmed
[25] Usami M, Akaza H, Arai Y, Hirano Y, Kagawa S, et al. (2007) Bicalutamide 80 mg combined with a luteinizing hormone-releasing hormone agonist (LHRH-A) versus LHRH-A monotherapy in advanced prostate cancer: findings from a phase III randomized, double-blind, multicenter trial in Japanese patients. Prostate Cancer Prostatic Dis 10:194-201. Article Pubmed
[26] Arai Y, Yoshiki T, Yoshida O (1990) Prognostic significance of prostate specific antigen in endocrine treatment for prostatic cancer. J Urol 144:1415–1419. Pubmed
[27] Furuya Y, Akimoto S, Akakura K, Igarashi T, Murakami S, et al. (1998) Response of prostate-specific antigen after androgen withdrawal and prognosis in men with metastatic prostate cancer. Urol Int 60:28–32. Article Pubmed
[28] Denis LJ, Carnelro de Moura JL, Bono A, Sylvester R, Whelan P, et al. (1993) Goserelin acetate and flutamide versus bilateral orchiectomy: a phase III EORTC trial (30853). EORTC GU Group and EORTC Data Center. Urology 42:119-129. Article Pubmed
[29] Mulders PF, Fernandez del Moral P, Theeuwes AG, Oosterhof GO, van Berkel HT, et al. (1992) Value of biochemical markers in the management of disseminated prostatic cancer. Eur Urol 21:2–5. Article Pubmed
[30] Armstrong AJ, Tannock IF, de Wit R, George DJ, Eisenberger M, et al. (2010) The development of risk groups in men with metastatic castration-resistant prostate cancer based on risk factors for PSA decline and survival. Eur J Cancer46:517-525. Article Pubmed
[31] Agus DB, Cordon-Cardo C, Fox W, Drobnjak M, Koff A, et al. (1999) Prostate cancer cell cycle regulators: response to androgen withdrawal and development of androgen independence. J Natl Cancer Inst 91:1869–1876. Article Pubmed
[32] Niu Y, Altuwaijri S, Lai KP, Wu CT, Ricke WA, et al. (2008) Androgen receptor is a tumor suppressor and proliferator in prostate cancer. Proc Natl Acad Sci USA 105:12182–12187. Article Pubmed
[33] Knudsen KE, Arden KC, Cavenee WK (1998) Multiple G1 regulatory elements control the androgen-dependent proliferation of prostatic carcinoma cells. J Biol Chem 273:20213–20222. Article Pubmed
[34] Soloway MS, Hardeman SW, Hickey D, Raymond J, Todd B, et al. (1988) Stratification of patients with metastatic prostate cancer based on extent of disease on initial bone scan. Cancer 61:195-202. Article Pubmed
[35] FuruyaY, Nozaki T, Nagakawa O, Fuse H (2002) Low serum testosterone level predicts worse response to endocrine therapy in Japanese patients with metastatic prostate cancer. Endocr J 49:85–90. Article Pubmed
[36] Imamoto T, Suzuki H, Yano M, Kawamura K, Kamiya N, et al. (2008) The role of testosterone in the pathogenesis of prostate cancer. Int J Urol 15:472–480. Article Pubmed


