Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 1, Issue 8, October 2013, Pages 194-200
Original researchOpen Access
A retrospective study of survival in breast cancer patients undergoing deuterium depletion in addition to conventional therapies
- 1 HYD LLC for Cancer Research and Drug Development, Budapest, H-1124, Hungary
- 2 Department of Public Health, Medical School, University of Pécs, Pécs, 7624, Hungary
*Corresponding author: Gábor Somlyai, HYD LLC for Cancer Research and Drug Development, Budapest, H-1124, Hungary, Tel: +36-1-381-0765/ +36-1-365-1660; Fax: +36-1-365-1661. E-mail: gsomlyai@hyd.hu
Received 26 May 2013 Revised 6 September 2013 Accepted 16 September 2013 Published 25 September 2013
DOI: http://dx.doi.org/10.14312/2052-4994.2013-29
Copyright: ©2013 Krempels K, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
There is increasing evidence that the heavy isotope of hydrogen, deuterium (D), has a pivotal role in cell signalling and that its depletion through the replacement of normal drinking water with deuterium-depleted water (DDW) results in tumour necrosis. The impact of D–depletion on breast cancer outcome was studied retrospectively. The normal daily water intake (150 ppm D) of 232 breast cancer patients was replaced with DDW (65-105 ppm D) for at least 91 days, without altering conventional treatment regimens. According to staging at initial diagnosis, patients with early stage breast cancer (n=158) achieved a median survival time (MST) of 217 months (18.1 years), compared with 52 months (4.3 years) in patients with advanced disease (n=74). The MST is pending in the subgroup of patients who were in remission at the start of DDW treatment; only one out of 48 patients died during the cumulative follow-up period of 221.1 years. Although single DDW treatment was effective, an outstandingly long MST of 24.4 years was attained in the subgroup of 53 patients who were treated with DDW at least twice. In comparison with published data, DDW treatment in combination with or as an extension of conventional therapies noticeably prolonged MST in certain subgroups of breast cancer patients. D-depletion may also be a highly effective therapy for preventing the recurrence of breast cancer. Furthermore, the method is safe and can be easily integrated into standard treatment regimens for breast cancer.
Keywords: deuterium depletion; deuterium-depleted water; breast cancer; retrospective study; median survival time; early stage breast cancer; advanced disease
IntroductionTop
Breast cancer is a major health problem worldwide and the second most common cause of cancer-related deaths in women. Although population screening by mammography can ensure early detection of breast cancer and the rapid development of therapeutic options (i.e. surgery, radiotherapy, hormonal therapy, chemotherapy, more recently, targeted cancer drugs) provides the possibility of multidisciplinary treatment, the morbidity of breast cancer is still high [1] . Trends in mortality and morbidity did not show a decline in the United States between 2005 and 2009 [2] . Similarly, age-standardised incidence rates for breast cancer in females in Great Britain did not decrease in the past 7 to 8 years [3] . Long-term follow-up studies showed that the probability of relapse ranges from 30 to 85% depending on tumour stage [4] . Median survival time (MST) in advanced breast cancer defined by different clinical trials is 12 to 31 months [5] . Combined chemotherapy and targeted biological therapies are the mainstay of treatment for metastatic breast cancer and the goal in such cases is to maintain quality of life, ameliorate symptoms, and delay progression [5] .
A wide range of information is available regarding the differences in physicochemical properties of deuterium (D) and protium (H), the two stable isotopes of hydrogen [6, 7, 8]. The possible role of naturally occurring deuterium (D) and the impact of a deficit of deuterium on cell proliferation have been verified in numerous biological systems [9, 10, 11]. The in vitro growth rate of different tumorous cell lines was significantly inhibited in culture media prepared with deuterium-depleted water (DDW), and the administration of DDW as drinking water resulted in complete or partial tumour regression in mice xenotransplanted with MDA-MB-231 or MCF-7 human breast adenocarcinomas or PC-3 human prostate tumours [9, 10]. The apoptosis-triggering effect of DDW was demonstrated both in vitro [10] and in vivo [11] . D-depletion also exerts an impact on protooncogenes and tumour suppressor genes. Induction of the expression of c-myc, Ha-ras, and p53 genes by carcinogen exposure was significantly inhibited when experimental animals were given DDW to drink [12] . The registered oral formulation of the deuterium-depleted veterinary anticancer drug Vetera-DDW-25 AUV has been used successfully in the treatment of household pets, mainly dogs and cats with spontaneous malignancies. Siniak et al. also confirmed the anticancer effect of DDW and suggested its application in an expedition to Mars to reduce the risk of cancer as a result of the severe radiation exposure [13] . The anticancer effect of D-depletion was also confirmed in a double-blind, randomised, 4-month-long, human phase II clinical trial on prostate cancer [14] . Extended follow-up and retrospective evaluation of 91 prostate cancer patients suggested that the method might reduce the mortality rate of prostate cancer because it delayed disease progression and prolonged MST [14] . Lung cancer patients undergoing DDW treatment in addition to conventional treatments achieved longer MSTs, and DDW was also successfully applied in lung cancer complicated by brain metastases [15] .
The aim of the present retrospective study was to investigate the impact of D-depletion on the outcome of breast cancer. The daily water intake of 232 breast cancer patients was replaced with DDW (105-65 ppm D) for at least 91 days, without altering the conventional treatment regimens. The primary end point of the study was MST. To obtain data comparable to historical controls, subgroups were created based on initial staging of the disease.
Materials and methodsTop
DDW production
DDW was produced from ordinary water containing a natural amount of D (150 ppm), equivalent to (16.8 mmol/L) by fractional distillation to decrease the D-concentration to 105-65 ppm. The production of DDW is based on the different volatility of normal water (H2O) and heavy water (D2O). At the boiling point of normal water the steam in equilibrium with the liquid phase contains approximately 2.5% less deuterium. By repeating these evaporation steps, which are carried out in distillation towers for industrial quantities, the deuterium content of water can be decreased commensurate with the tray number of the distillation tower. D-concentration was determined by mass spectrometry (Finnigan delta plus XP, using BTW XV standards) with ±1 ppm precision [16].
Main principles of administration of DDW
Per os (PO) DDW treatment supplemented and did not replace conventional therapy. The aim of the treatment was to reduce D–concentration in the patient’s body by replacing the normal water intake with DDW as described previously [14] . DDW treatment started at 105 ppm D, and was gradually decreased to 85 ppm and 65 ppm (with each step 20 ppm lower than the previously applied preparation) every 1 to 3 months, for a total treatment period of 6 to 10 months. DDW treatment was discontinued for 2 to 3 months and started again for repeated 4- to 6-month treatment durations. Over time, the treatment periods were shortened, although each had to be at least 3 months long, and the breaks were extended to 4-6 months. DDW-dose (DdU, Deuterium depletion unit) was calculated according to the following formula [14] :
|
General characterisation of patients
According to the study protocol, eligible patients were women who were diagnosed with histologically confirmed breast cancer and had been consuming DDW for at least 91 days. The study included 232 patients who met the inclusion criteria (Table 1). The median age of the study population was 50 years. The patients started DDW treatment at different times after diagnosis. The median length of time between diagnosis and start of DDW treatment (DG_to_DDWstart) was 13.2 months. The standard deviation (SD) of the average length of time shows that in some cases several years elapsed until inclusion in the study whereas other patients were enrolled after only a few months. The median duration of DDW treatment was 14.1 months (LoDDW) and the cumulative duration of DDW treatment was 474 years. The cumulative length of total follow-up (from diagnosis to the end of follow-up) was 1,346 years and the median follow-up period (DG_to_Flwup) was 49.4 months (4.1 years), although the SD of the average indicated a wide range of values. Thirty-five patients (15%) were followed for a period longer than 10 years. The cumulative length of follow-up from the start of DDW treatment was 654 years (DDWstart_to_Flwup), and the median follow-up time was 18.5 months.
| Variable+ | Median | Average | SD | Cumulative time |
| Age | 50 y |
51 y |
11 y |
N/A |
| Dg_to_DDWstart | 13.2 m |
35.8 m |
53.6 m |
692.8 y |
| LoDDW | 14.1 m |
24.5 m |
29.8 m |
474.2 y |
| Dg_to Flwup | 49.4 m |
69.7 m |
62.3 m |
1346 y |
| DDWstart_to_Flwup | 18.5 m |
33.8 m |
38.4 m |
654 y |
Abbreviations: DG_to_DDWstart = time from diagnosis to the start of DDW treatment; LoDDW = length of DDW treatment; DG_to_Flwup = follow-up time from diagnosis; DDWstart_to_Flwup = follow-up time from the start of DDW treatment; SD = standard deviation; m = months; y = years
Study design and data collection
The aim of the study was to investigate the effect of DDW on the outcome of breast cancer. We evaluated 232 patients who were continuously enrolled between February 1993 and April 2011. Conventional forms of therapy were administered in 16 hospitals and clinics in Hungary (listed in the acknowledgements), where the follow-up examinations also took place. The findings of the follow-up examinations, as well as the applied D-concentration and DDW-dose, were recorded. Patient documentation was retrospectively evaluated and data collection was closed in January 2012. Patients were aware of, and provided access to, available information regarding DDW treatment. Patients were divided into subgroups at two separate points in the study. First, two groups of patients were formed based on the staging at diagnosis: Group A1 (n=74), composed of patients with advanced breast cancer with distant metastasis or locally advanced disease, and Group E1 (n=158), composed of patients with early stage breast cancer whose disease did not spread beyond the axillary lymph nodes. The second time point was when patients entered the study and started DDW treatment. By that time a high percentage of patients had received conventional therapies, which resulted in complete remission in some cases but most patients had progressive disease when they started DDW treatment. Three subgroups were formed based on status at the start of treatment: Group A2 (n=129), composed of advanced breast cancer patients; Group E2 (n=55), composed of patients with early-stage breast cancer, and Group R (n=48), composed of patients in remission. MST calculations were performed both from the time of first diagnosis and from the initiation of DDW treatment. Data of 74 breast cancer patients, who were in advanced stage at the time of diagnosis and therefore had limited life expectancy, were evaluated with respect to a dose-response relationship.
Statistical evaluation
The primary endpoint of the study was survival and the Kaplan-Meier method was applied to calculate survival curves. Statistical analysis was performed using MedCalc Statistical Software, Version 12.3.0. Because the study was performed retrospectively and patients started DDW treatment at different time points after diagnosis, MST was computed both from the time of diagnosis (MST from DG) of breast cancer and from the start of DDW treatment (MST from DDWstart). The dose-response relationship in the subgroup of breast cancer patients with advanced disease was tested using one-way analysis of variance (ANOVA) and the Bonferroni post hoc test. The mean difference is significant at 0.05 level.
ResultsTop
Median survival in breast cancer patients undergoing DDW treatment in addition to conventional treatment regimens
We calculated the MST of all enrolled breast cancer patients using the Kaplan-Meier method although we were aware of the heterogeneity of the study population regarding staging both at the time of diagnosis and at the start of DDW treatment. The overall survival time was 148 months (12.3 years) from the diagnosis of breast cancer and 89 months (7.4 years) from the start of DDW treatment. To determine whether these MSTs are noteworthy, a literature search was performed to determine the stage of breast cancer that results in MSTs of the same length. We found that the chance of long-term survival increases primarily in the subset of patients whose disease is detected at an early stage [17]. Considering that a high percentage (55.6%) of the 232 breast cancer patients enrolled in the present study were in the most advanced stage of disease at start of DDW treatment, we presume that DDW treatment contributed to their extended survival.
Evaluation of the subgroups showed that according to staging of the disease at the first diagnosis, patients with early-stage breast cancer achieved an MST of 217 months (18.1 years), compared with 52 months (4.3 years) for other patients with advanced cancer. MSTs from the start of DDW treatment (DDWstart_to_Flwup) are presented in Figure 1. The MST was 89 months (7.4 years) in the group of patients with early stage breast cancer, while patients in advanced stage achieved an MST of 39 months (3.3 years). It is outstanding that MST calculations are still pending in patients who started DDW treatment in remission; only one out of these 48 patients died during the cumulative follow-up period (DDWstart_to_Flwup) of 221.1 years (median follow-up time: 32 months). A striking difference can be seen between the subgroups, underlying the fact that staging is a major determining factor in the survival of breast cancer. Moreover, all these MSTs are long in comparison to other clinical studies on breast cancer outcome.
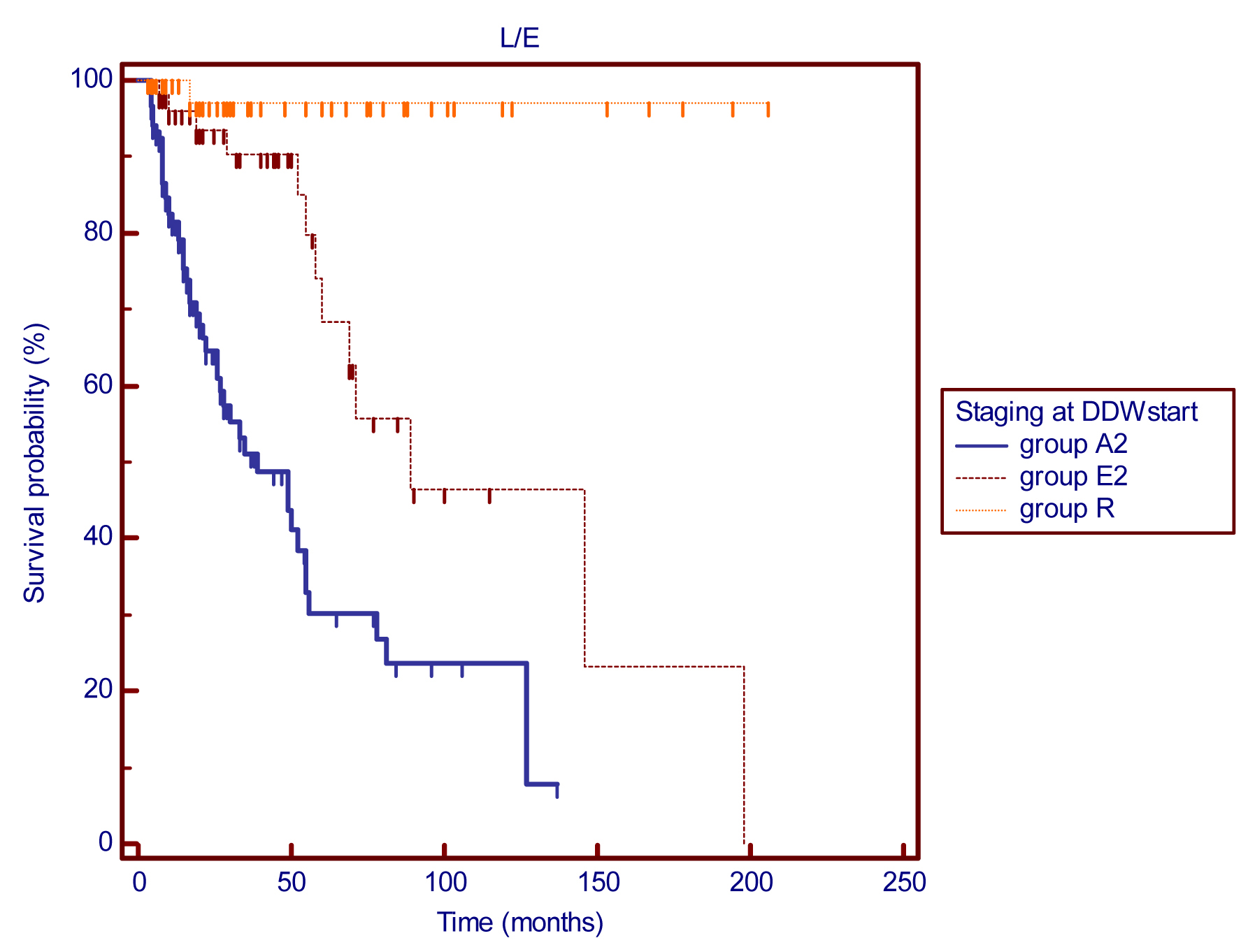
Figure 1 Kaplan-Meier graphs of survival from the start of DDW treatment for 232 patients with different stages of breast cancer: MSTs were 39 months for group A2 (n=129) and 89 months for group E2 (n=55). The MST for group R (48 patients) was non-calculable as only one death in a 221.1-year cumulative follow-up period was recorded, p (between groups) <0.0001.
Abbreviations: A2 = advanced disease; E2 = early-stage breast cancer; R = patients in remission; DDWstart = start of DDW treatment.
Analysis of patients with advanced disease at the start of DDW treatment
Considering all available parameters, our aim was to find the key parameter with the greatest impact on the efficacy of DDW treatment. For this purpose, we evaluated the subgroup of 74 advanced cancer patients comparing to historical control [4, 5]. Descriptive statistics of the subgroup showed that the median time from diagnosis of advanced stage to the start of DDW treatment was 181 days (5.9 months), with a standard deviation (SD) of 424.5 days (13.9 months). The median length of DDW treatment (LoDDW) was 402 days (13.2 months), with SD of 569.26 days (18.7 months).
Analysis of MSTs in subgroups defined by the response to combined treatment with DDW and conventional therapies
Following treatment with DDW in addition to conventional therapies, 16 cases (21.6%) showed a complete response (CR) and 27 (36.5%) showed a partial response (PR). No change (NC) was observed in 12 (16.2%) patients and progressive disease (PD) was recorded in 19 (25.7%). Using the Kaplan-Meier method, MSTs from the start of DDW treatment were 52, 48, 26, and 13 months in the CR, PR, NC, and PD subgroups, respectively [p (among groups) <0.001] (Figure 2). A significant correlation was detected between MSTs and the response to DDW therapy.
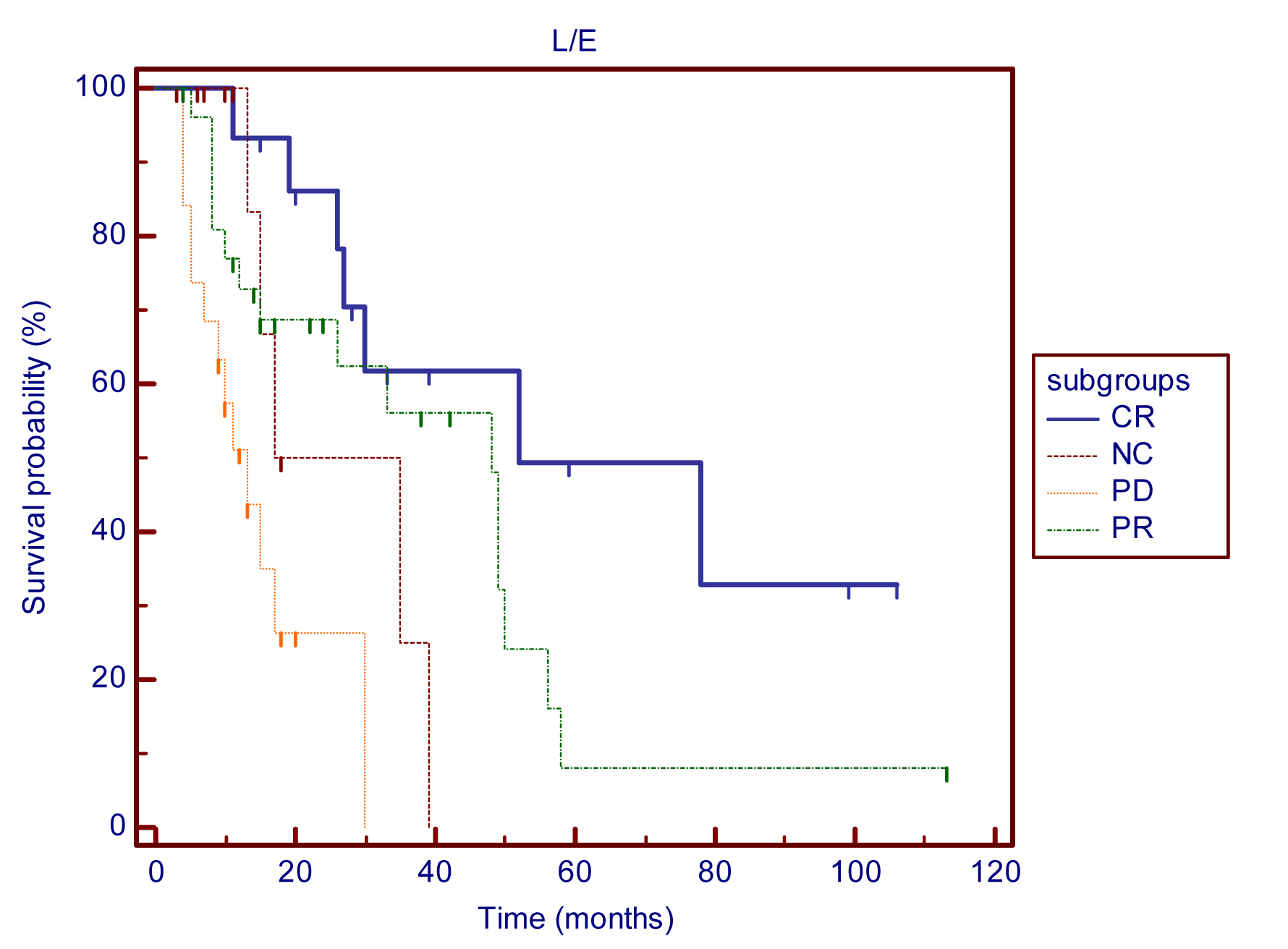
Figure 2 Kaplan-Meier graphs of survival from the start of DDW treatment in CR, PR, NC, and PD subgroups of 74 breast cancer patients with advanced disease: MST in the subgroups was as follows: CR - 52 months (n=16), PR - 48 months (n=27), NC - 26 months (n=12), PD - 13 months (n=19) p (between groups) <0.0001.
Abbreviations: CR = complete response; PR = partial response; NC = no change; PD = progressive disease.
Analysis of the relationship between DDW-dose and response to DDW treatment in breast cancer patients at an advanced stage
Preclinical studies suggested that the main determining factor in the anticancer effect of DDW is the calculated dose of DDW (DdU). The DdU was in the range of 0.02 to 4.71 in the population during the follow-up, and the response to DDW treatment was determined at 85 time points in the 74 patients. Table 2 shows the means of DdU calculated in the CR, PR, NC and PD subgroups. The highest mean was found in subgroup CR, while the lowest value in group NC. Because data showed normal distribution and there was no significant difference among the standard deviations between the subgroups, one-way analysis of variance (ANOVA) and the Bonferroni post hoc test were performed. There was a significant difference between the subgroups (p<0.001) using the one-way ANOVA test (Table 3). Analysis of certain subgroup pairs revealed significant difference in the following cases: subgroup CR and NC (p=0.001) and subgroup CR and PD (p=0.004). The results suggest that higher DDW-dose results in a pronounced response to DDW treatment.
| Response | N | Mean | Std. Deviation | 95% Confidence Interval for Mean | Minimum | Maximum | |
| Lower Bound | Upper Bound | ||||||
| CR | 16 |
1.6879 |
1.02796 |
1.1401 |
2.2357 |
0.45 |
4.71 |
| PR | 29 |
1.2897 |
0.57248 |
1.0720 |
1.5075 |
0.20 |
2.36 |
| NC | 12 |
0.6638 |
0.35056 |
0.4411 |
0.8865 |
0.20 |
1.17 |
| PD | 28 |
0.9289 |
0.66529 |
0.6709 |
1.1868 |
0.02 |
2.27 |
| Total | 85 |
1.1574 |
0.75377 |
0.9949 |
1.3200 |
0.02 |
4.71 |
Abbreviations: DdU = deuterium depletion unit; DDW = deuterium-depleted water; CR = complete response; PR = partial response; NC = no change; PD = progressive disease
Analysis of the effect of early- or late-onset of DDW treatment after diagnosis of breast cancer
In the present retrospective trial the enrollment of patients was continuous and the time period between diagnosis and DDW start was relatively long in most patients (median 13.2 months). Therefore, we investigated whether the length of time from diagnosis to the start of DDW treatment influenced the possible effect of DDW treatment on the outcome of the disease. Patients were divided into two subgroups: group I (n=114) contained patients that joined the study within 1 year after their illness was diagnosed, and group II (n=118) contained patients who started DDW treatment more than 1 year after the first diagnosis. By the time patients entered the trial a higher proportion of patients in group II (79.7%) were staged at advanced disease compared with those in group I (30.7%). Early stage breast cancer was diagnosed in 36.8% of the patients in group I and in 11% of group II. On the other hand, the number of patients who were successfully treated and achieved remission due to conventional treatments was significantly lower in group II than in group I (9.3% versus 31.6%).
The MST for group I (n=114) is still pending although 18 deaths were registered during the cumulative follow-up period of 399.8 years, which indicates 1 death per 22.2 years. The MST for group II was 49 months (4.1 years) (Figure 3). Considering that the great majority of patients (79.7%) suffered from advanced disease when they entered the trial, we assume that DDW extended the survival period in this subset of patients.
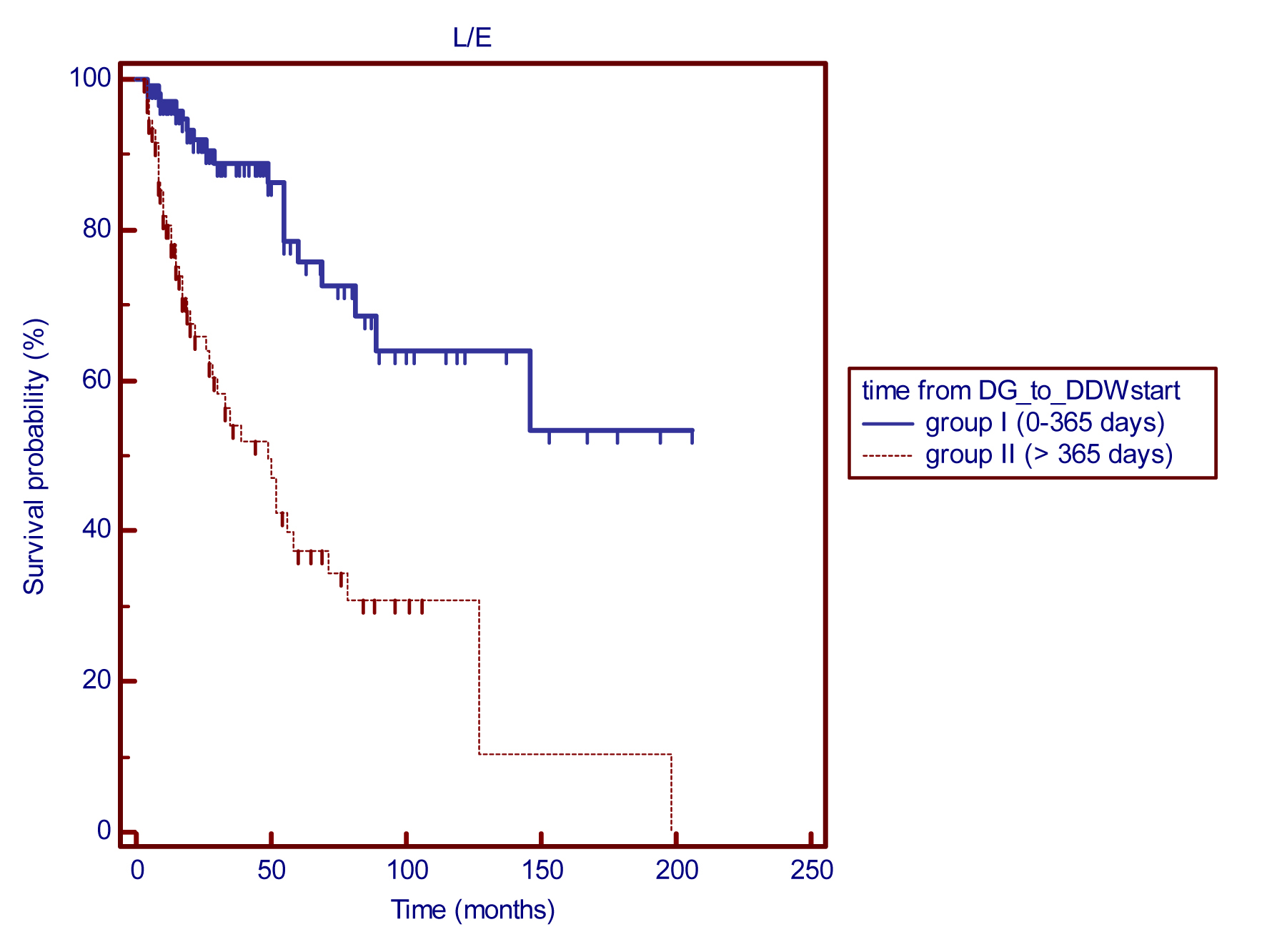
Figure 3 Kaplan-Meier graphs of survival from start of DDW treatment in subgroups of breast cancer patients defined by the length of time from diagnosis to DDWstart: MST for group I is pending (18 deaths/114 patients), cumulative follow-up was 399.8 years, indicating one death in 22.2 years. Group II (118 patients) showed a MST of 49 months, p (between groups) <0.0001.
Abbreviation: DG_to_DDWstart = time from the diagnosis to the start of DDW treatment.
MST in subgroups defined by the frequency of DDW treatments: Single versus repeated DDW treatment
Prevention of relapses and/or development of distant metastases is a crucial aspect of breast cancer, even decades after the first diagnosis and successful treatment. Further subgroups were formed according to the frequency of DDW treatment. A single DDW treatment was administered to 179 patients (group N) whereas 53 patients repeated the cure at least once (group Y). The Kaplan-Meier graphs show that the survival curves have distinct shapes (Figure 4). Patients who were treated with DDW at least twice had an MST of 293 months (24.4 years) whereas those who received a single DDW treatment had an MST of 108 months (9 years).
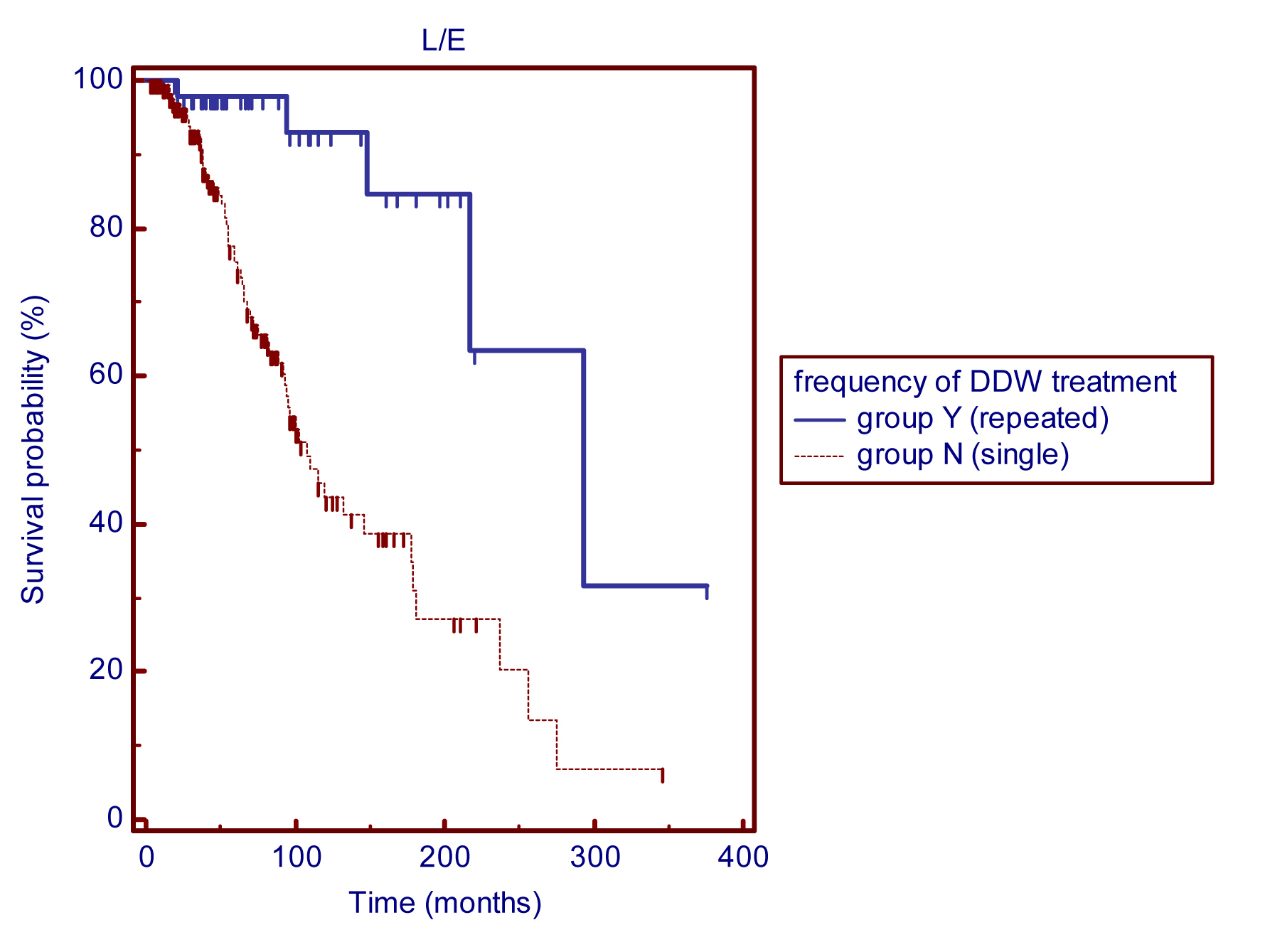
Figure 4 Kaplan-Meier graphs of survival from the diagnosis in breast cancer patients undergoing single (group N) or repeated (group Y) DDW treatment: MST for group Y (n=53, repeated DDW treatment) was 293 months, compared with 108 months for group N (n=179 patients, single DDW treatment), p (between groups) <0.0001.
Survival in breast cancer patients enrolled before and after January 2005
The present retrospective study covers a wide interval from April 1993 to January 2012. During that time there were significant advances in the treatment of breast cancer. The entire study population was divided into two subgroups based on the date of involvement so that we could investigate, whether D-depletion contributed to the longer survival, when it was applied in addition to the recent treatment regimens. The subgroup of patients who entered the study before January 2005 (n=156) showed longer survival in comparison to the historical controls. The MST was 71 months (5.9 years) in the group of patients with early stage (n=34) breast cancer, while patients in advanced stage (n=86) achieved an MST of 35 months (2.9 years) (Figure 5). MST calculations are still pending in patients who started DDW treatment in remission; only one out of these 36 patients died. MSTs in the subgroup of patients enrolled after January 2005 was non-calculable. Survival from start of DDW treatment was extremely long, 8 deaths were registered during the cumulative follow-up period of 118.7 years, despite of the fact, that high proportion of these patients was diagnosed with advanced breast cancer (n=43), 21 patients suffered from primary disease and only 12 patients were in remission.
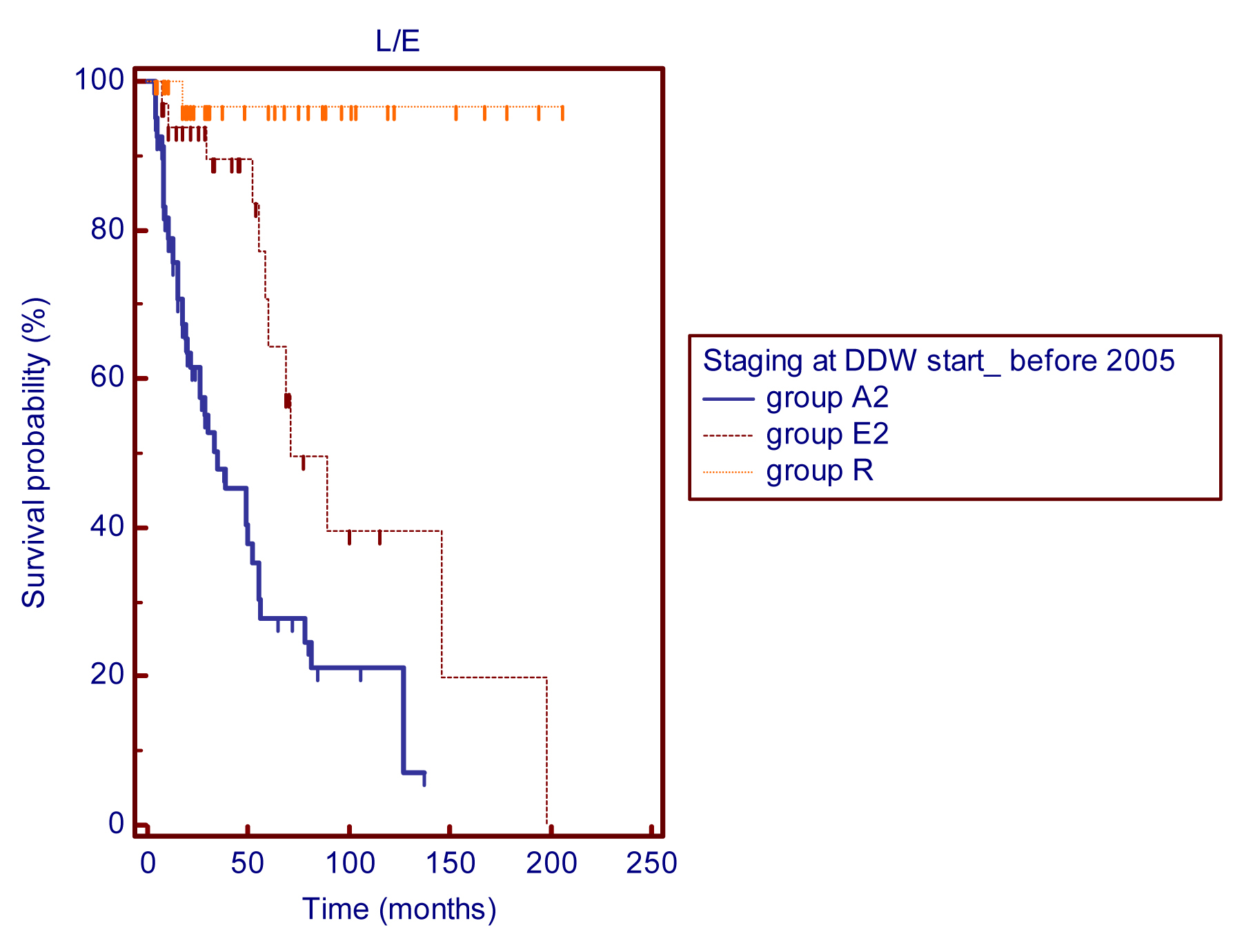
Figure 5 Kaplan Meier graphs of survival from the start of DDW treatment in 156 patients enrolled before 2005: MSTs were 35 months for group A2 (n=86) and 71 months for group E2 (n=34). The MST for group R (n=36) was non-calculable as only one death was recorded, p (between groups) <0.0001.
Abbreviations: A2 = advanced disease; E2 = early-stage breast cancer; R = patients in remission; DDWstart = start of DDW treatment.
DiscussionTop
In vitro and in vivo experiments have demonstrated that naturally occurring deuterium plays a pivotal role in cell growth and a shortage of deuterium in tumour cells induces apoptosis, resulting in partial or complete tumour regression [9, 10, 11, 18]. Clinical studies on lung cancer and prostate cancer confirmed that integration of D-depletion into conventional treatments delayed disease progression, extended MST, was beneficial in the prevention of relapses [14, 19], and reduced the risk of the development of cancer in severe radiation exposure [13] . To gain further information on the application of D-depletion, we retrospectively evaluated records of 232 breast cancer patients who underwent DDW treatment in addition to conventional treatment regimens.
The overall survival rates calculated in the present study suggest that DDW treatment noticeably prolonged MST in comparison with published data [5, 17]. The MST of the entire cohort was 148 months from diagnosis of the disease and 89 months from the start of DDW treatment. Consistent with earlier publications, subgroup evaluation showed that MST correlated with the staging of breast cancer [4, 17] with an MST of 217 months in patients with early disease and 52 months in advanced cases. We were aware that the study population was heterogeneous. All medical records were thoroughly investigated for possible parameters that might influence the outcome, and the subgroup evaluation was performed based on these aspects. It is well known that retrospective approach has limitations but one of the advantages of a retrospective study is their usefulness in addressing rare conditions and it helps to assess the feasibility of prospective studies.
In Group I patients (n=114), who started DDW treatment within 1 year after diagnosis, only 18 deaths were registered (one death in every 22.2 years). The low death rate did not allow the calculation of MST. When DDW treatment was started later than 1 year after diagnosis (Group II, n=118) the MST was 49 months despite the fact that 79.7% of the patients were in advanced stage.
The cumulative follow-up of 1,346 years in the present study allowed us to evaluate whether D-depletion was effective in the prevention of relapses. Our data suggest that DDW treatment provided additional benefits to breast cancer patients and had a significant role in the prevention and/or delay of progression. DDW treatment was also applicable to patients who were in remission after successful conventional treatment (n=48), and the cumulative follow-up period and reported deaths indicated one death per 221.1 years, which is an outstanding result among published data on breast cancer. Further follow-up is needed to assess the exact survival periods for this subpopulation.
Evaluation of the correlation between DDW-dose (DdU) and response to treatment provided evidence on the effectiveness of DDW therapy. The great majority of patients whose disease did not change or progressed took DDW at a low dose and/or at irregular intervals. Additionally, irregular consumption resulted in alterations in the response to the therapy: interruption of DDW consumption resulted in disease progression, but PD or NC converted to PR or CR when DDW treatment was resumed. These facts strongly suggest that in most cases the failure of DDW therapy could be attributed to improper dosage of DDW and/or irregular DDW consumption, rather than the ineffectiveness of DDW treatment. Moreover, patients showing CR took DDW at a higher dose and their MSTs (52 months) were two or three times longer than those of patients who received only conventional treatment (MST, 12-31 months).
The other key issue determining the effectiveness of D-depletion was the frequency of DDW applications (single or repeated treatment). Although single DDW treatment (n=179) extended survival in comparison with published MSTs, repeated DDW treatment resulted in outstandingly long survival periods. Patients who took DDW for only one treatment period (n=126) had an MST of 9 years whereas patients (n=53) who took DDW more than once attained an MST of 24.4 years.
It is crucial to investigate survival data of breast cancer patients from the point of long-term follow-up, because long-term survival and the chance for late onset of recurrence are also characteristic for breast cancer. There are advances in the knowledge about breast cancer, in its molecular basis, in early detection and multidisciplinary treatment options. Our study covers a wide time interval, and this fact raises the question, whether D-depletion could exert its beneficial effects when it is applied in patients undergoing the most recent treatment regimens. MSTs of patients enrolled before 2005 (Figure 5) were noticeably longer compared to historical control, and did not significantly differ from MSTs of the entire population (Figure 1). This fact suggests that D-depletion had significant impact on survival. Patients entering the trial after January 2005 reached excellent survival, which also support the evidence that DDW treatment contributed to longer survival. Extension of the follow-up period is needed to calculate exact MSTs in the subgroups with different stages of breast cancer.
Our data indicate that repeated DDW treatment is highly beneficial for breast cancer patients and might contribute to longer survival and possibly the prevention of disease recurrence. Compared with other published data, DDW treatment in combination with, or as an extension of conventional therapies noticeably prolonged MST in breast cancer patients. The above results are impressive in breast cancer research. All this information is useful in conduction of further prospective trials to clarify the clinical significance of this new treatment modality.
ConclusionTop
D-depletion may be a highly effective therapy both in the treatment of early-stage or advanced breast cancer and in preventing disease recurrence. The method is safe and innocuous; preclinical toxicology studies [20] , prospective and retrospective clinical trials [14, 18] confirmed that absolutely no adverse events occurred during long-term application of DDW at a wide concentration range of 25 to 125 ppm. We conclude that D-depletion offers additional benefits to conventional therapy and can be integrated into standard treatment regimens for breast cancer.
Acknowledgements
The authors wish to acknowledge the clinicians who carried out the conventional forms of cancer therapy and follow-up examinations at the following Hungarian hospitals: Bajcsy-Zsilinszky Hospital, Budapest; Buda-Region MÁV Hospital, Budapest; MÁV Hospital, Budapest; Korányi National Institute of Tuberculosis and Pulmonology, Budapest; National Institute of Oncology, Budapest; Péterfy Sándor Hospital, Budapest; Semmelweis University Clinics, Budapest; St. Imre Hospital, Budapest; St. István Hospital, Budapest; St. Margit Hospital, Budapest; Uzsoki Hospital, Budapest; Szeged City Hospital, Szeged; Semmelweis Hospital, Miskolc; Csongrád Regional Hospital, Szentes; University of Debrecen, Medical and Health Science Center; Regional Hospital, Berettyóújfalu.
ReferencesTop
[1] Boyle P, Ferlay J (2005) Cancer incidence and mortality in Europe, 2004. Article Pubmed
[2] Howlader N, Noone AM, Krapcho M, Neyman N, Aminou R, et al. (2012) SEER Cancer Statistics Review, 1975-2009 (Vintage 2009 Populations), National Cancer Institute. Article
[3] Breast Cancer Incidence Statistics Cancer Research UK . Article
[4] Gonzalez-Angulo AM, Morales–Vasquez F, Hortobágyi GN (2007) Overview of resistance to systemic therapy in patients with breast cancer. Adv Exp Med Biol 608:1-22. Article Pubmed
[5] O'Shaughnessy J (2005) Extending survival with chemotherapy in metastatic breast cancer. Oncologist 10:20–29. Article Pubmed
[6] Katz JJ, Crespi HL (1971) “Isotope Effects in Biological Systems,” In: C. J. Collins and N. S. Bowman, Eds., Isotope Effects in Chemical Reactions, Van Nostrand Reinhold, New York, pp. 286-363.
[7] Rundel PW, Ehleringer JR, Nagy KA (1988) Stable Isotopes in Ecological Research, New York: Springer.
[8] Jancsó G (2003) Isotope Effects. In: Vértes A, Nagy S, Klencsár Z, editors. Handbook of Nuclear Chemistry, Volume 2. Dordrecht, Netherland: Kluwer Academic Publishers pp. 85-116.
[9] Somlyai G, Jancsó G, Jákli G, Vass K, Barna B, et al. (1993) Naturally occurring deuterium is essential for the normal growth rate of cells. FEBS Lett 317:1–4. Article Pubmed
[10] Somlyai G, Laskay G, Berkényi T, JákliGy, Jancsó G (1998) Naturally occurring deuterium may have a central role in cell signalling. In: Heys JR, Melillo DG, editors. Synthesis and applications of isotopically labelled compounds. New York: John Wiley and Sons Ltd, pp. 137-141.
[11] Somlyai G, Laskay G, Berkényi T, Galbács Z, Galbács G, et al. (1998) The biological effects of deuterium-depleted water, a possible new tool in cancer therapy. Journal of Oncology 30:91–94. Article
[12] Gyöngyi Z, Somlyai G (2000) Deuterium depletion can decrease the expression of C-myc Ha-ras and p53 gene in carcinogen-treated mice. In Vivo 14:437–439. Article Pubmed
[13] SiniakIuE, Turusov VS, Grigor'ev AI, Zaridze DG, Gaĭdadymov VB, et al. (2003) Consideration of the deuterium-free water supply to an expedition to Mars. Aviakosm Ekolog Med 37:60–63. Article Pubmed
[14] Kovács A, Guller I, Krempels K, Somlyai I, Jánosi I, Z, et al. (2011) Deuterium depletion may delay the progression of prostate cancer. J Cancer Ther 2:548-556. Article
[15] Gyöngyi Z, Budán F, Szabó I, Ember I, Kiss I, et al. (2013) Deuterium depleted water effects on survival of lung cancer patients and expression of Kras, Bcl2, and Myc genes in mouse lung. Nutr Cancer 65:240-246. Article Pubmed
[16] Prosser SJ, Scrimgeour CM (1995) High-precision determination of 2H/1H in H2 and H2O by continuous-flow isotope ratio mass spectrometry. Analytical Chemistry, 67:1992-1997. Article
[17] Soerjomataram I, Louwman MW, Ribot JG, Roukema JA, Coebergh JW (2008) An overview of prognostic factors for long-term survivors of breast cancer. Breast Cancer Res Treat 107:309-330. Article Pubmed
[18] Cong FS, Zhang YR, Sheng HC, Ao ZH, Zhang SY, et al. (2010) Deuterium-depleted water inhibits human lung carcinoma cell growth by apoptosis. Exp Ther Med 1:277-283. Article Pubmed
[19] Krempels K, Somlyai I, Somlyai G (2008) A retrospective evaluation of the effects of deuterium depleted water consumption on 4 patients with brain metastases from lung cancer. Integr Cancer Ther 7:172-181. Article Pubmed
[20] Török G, Csík M, Pintér A, Surján A (2000) Effects of Different Deuterium Concentrations of the Media on the Bacterial Growth and Mutagenesis. Health Science 44:331-338.



