Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 1, Issue 9, November 2013, Pages 215-219
Case reportOpen Access
Therapeutic response to a new enzyme-targeting radiosensitization treatment (KORTUC-SC) for patients with chemotherapy-resistant supraclavicular lymph node metastasis
-
Aoyama N1,*
 ,
Ogawa Y1,
Kubota K1,
Ohgi K1,
Kataoka Y1,
Miyatake K1,
Tadokoro M1,
Yamanishi T1,
Ohnishi T1,
Hamada N1,
Kariya S1,
Tamura T1,
Nogami M1,
Nishioka A1,
Onogawa M2 and
Miyahara M2
,
Ogawa Y1,
Kubota K1,
Ohgi K1,
Kataoka Y1,
Miyatake K1,
Tadokoro M1,
Yamanishi T1,
Ohnishi T1,
Hamada N1,
Kariya S1,
Tamura T1,
Nogami M1,
Nishioka A1,
Onogawa M2 and
Miyahara M2
- 1 Department of Diagnostic Radiology and Radiation Oncology, Kochi University, Kochi 783-8505, Japan
- 2 Department of Pharmacy, Medical School, Kochi University, Kochi 783-8505, Japan
*Corresponding author: Dr. Nobutaka Aoyama, Department of Diagnostic Radiology and Radiation Oncology, Medical School, Kochi University, Oko-cho, Nankoku-shi, Kochi 783-8505, Japan, Tel.: 088-880-2294; Fax: 088-880-2368. E-mail: jm-aoyama_nobutaka@kochi-u.ac.jp
Received 8 July 2013 Revised 26 September 2013 Accepted 7 October 2013 Published 16 October 2013
DOI: http://dx.doi.org/10.14312/2052-4994.2013-32
Copyright: © 2013 Aoyama N, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
We have developed a new radiosensitizer containing hydrogen peroxide and sodium hyaluronate, part of a method we call “KORTUC II”. This study aimed to evaluate the safety and effectiveness of KORTUC II specifically for patients with chemotherapy-resistant supraclavicular lymph node metastasis (KORTUC-SC). Twelve patients were enrolled after providing fully informed written consent. Most of the patients underwent PET-CT examinations prior to and 3-8 months after KORTUC-SC, and every 6 months thereafter if possible. The patients’ age ranged from 58 to 77. The radiotherapy regimen was 4 Gy/ fraction, 3 fractions/ week, for 4 weeks, and the total dose was 48 Gy. The injection of 3 to 6 mL of the agent was started from the fifth radiotherapy fraction, and it was performed twice a week under ultrasonographic guidance. Therapeutic effects were evaluated by PET-CT examinations performed before and after KORTUC-SC treatment. The patients were well tolerated with minimal adverse effects. Of the 12 patients, 4, 5, 2 and 1 patient showed complete response, partial response, stable disease and progressive disease, respectively. The overall survival rate was 83% at 1 year and 75% at 2 years. The mean duration of follow-up at the end of February 2011 was 35.1 months. Based on the PET-CT studies, remarkable therapeutic effects of the KORTUC II treatment were clearly identified. The treatment outcomes were satisfactory. Well-designed, prospective, randomized, clinical trials are needed to confirm the therapeutic efficacy of KORTUC-SC.
Keywords: hydrogen peroxide; metastasis; radiosensitizer; sodium hyaluronate; supraclavicular lymph node; ultrasonographic guidance
IntroductionTop
Linear accelerator (Linac)-generated high-energy X-rays and electron beams that are generally used for radiotherapy are low-linear energy transfer (LET) forms of radiation, and have a relatively weak biological effect. High-energy X-rays and electron beams have minimal effects on tumors that contain many hypoxic cancer cells and/or large amounts of antioxidative enzymes. Examples of such tumors include malignant melanoma, various types of sarcomas, glioblastoma multiforme, and most relatively large tumors more than several centimeters in their long diameter. Thus, effective treatment of these tumors requires strategies to re-oxygenate hypoxic tumor cells and to inhibit antioxidative enzymes. The concepts underlying this new enzyme-targeting radiosensitization treatment (KORTUC II) are shown in Figure 1 [1]. The aim is to formulate an efficient low-LET radiosensitizer that has minimal irritation, can be safely injected into the human body, and can delay or reduce degradation of hydrogen peroxide and preserve oxygen concentration in the tumor tissue for more than 24 hours.
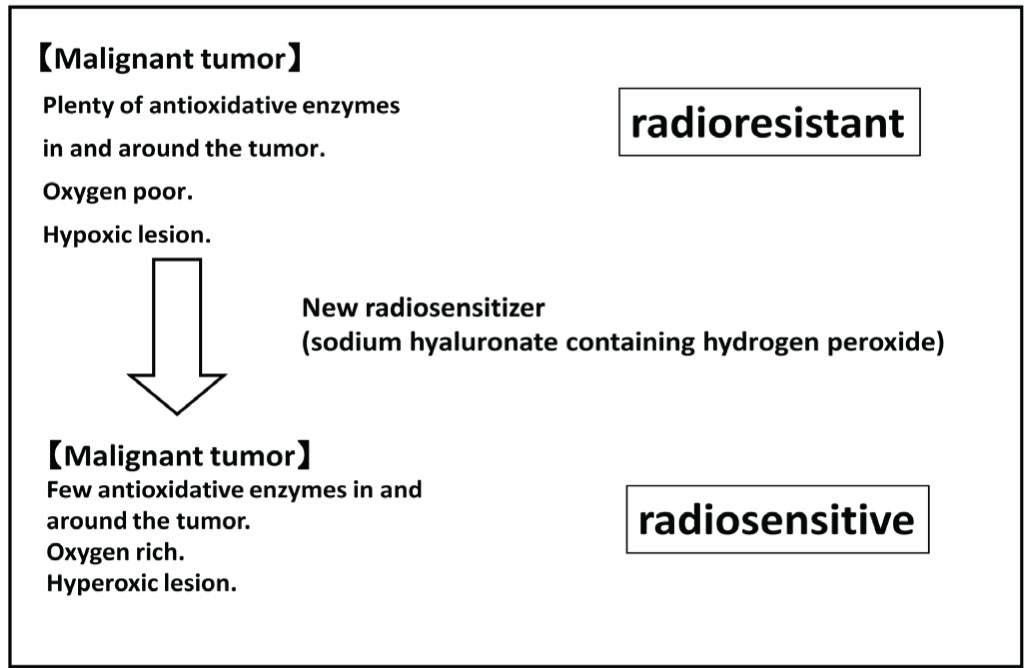
Figure 1 The mechanism of action of the new radiosensitizer (KORTUC II).
After much experimentation, it was found that various side effects (e.g., irritation of the skin, mucous membranes, and other tissues) related to the use of hydrogen peroxide could be reduced by combining hydrogen peroxide with sodium hyaluronate [2].
Moreover, co-administration of sodium hyaluronate with hydrogen peroxide reduced the tendency of hydrogen peroxide to readily give off oxygen in the presence of large amounts of antioxidative enzymes such as peroxidase and catalase. Thus, even when injected into the human body, its rapid degradation due to the action of antioxidative enzymes was significantly reduced, enabling the radiosensitizing effect to be exerted more safely and efficiently and for a longer duration.
Based on these experimental results, we developed Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas (KORTUC) II, a new radiosensitizer containing hydrogen peroxide and sodium hyaluronate for intratumoral injection into various types of tumors that are not superficially exposed [3]. The purpose of this study was to evaluate the safety and effectiveness of KORTUC II specifically for patients with chemotherapy-resistant supraclavicular lymph node metastasis under a method named KORTUC-SC (KORTUC II for chemotherapy-resistant supraclavicular lymph nodes metastasis).
Materials and methodsTop
This study was performed at Kochi Medical School Hospital from September 2006 to March 2010. A total of 12 patients (2 males and 10 females) with chemotherapy-resistant supraclavicular lymph node metastasis were enrolled in the study after providing fully informed written consent. The chemotherapy regimens administrated for the patients before the radiosensitization treatment were described below: Paclitaxel weekly (Pt.2, Pt.12), Cyclophosphamide + Methotrexate + Fluorouracil monthly (Pt.3), Epirubicin + Cyclophosphamide monthly (Pt.4), Trastuzumab + Vinorelbine ditartrate monthly (Pt.5), Fluorouracil + Cyclophosphamide + Epirubicin hydrochloride monthly (Pt.7), Trastuzumab + Docetaxel hydrate monthly (Pt.10), Gemcitabine hydrochloride weekly (Pt. 11). For four patients (Pt.1, Pt.6, Pt.8 and Pt. 9), the other types of chemotherapy regimens were administrated.
Patients’ age ranged from 58 to 77 years (average 65 years). Radiation therapy to supraclavicular lymph node metastases consisted of 4 Gy/ fraction, 3 fractions/ week, for a total 12 fractions, and the total radiation dose was 48 Gy. The appropriate energy level of electron beams was selected for individual patients based on the depth of the target lesion. The injection of 3 to 6 mL of the agent was initiated at the fifth radiation session and was performed twice a week under ultrasonographic guidance. The total number of injections was 5-6. The ethics committee of the Medical School of Kochi University approved KORTUC II for radiation therapy to supraclavicular lymph node metastases. Patient data and therapeutic effects are shown in Table 1.
| S. No. | Age (y)/ sex | Original disease | Target site | SUVmax of FDG accumulation (pretreatment→post) | Response |
| 1 | 75/F |
Hepatocellular carcinoma |
Left supraclavicular node |
(7.8→2.3) |
PR |
| 2 | 63/F |
Breast cancer |
Right supraclavicular node |
(4.9→5.7) |
PD |
| 3 | 56/F |
Breast cancer |
Right supraclavicular node |
(10.7→2.5) |
CR |
| 4 | 66/F |
Breast cancer |
Left supraclavicular node |
(7.2→3.4) |
PR |
| 5 | 61/F |
Breast cancer |
Left supraclavicular node |
(5.7→2.2) |
CR |
| 6 | 64/M |
Thyroid cancer |
Left supraclavicular node |
(11.1→10.6) |
SD |
| 7 | 63/F |
Carcinoma of unknown primary site |
Right supraclavicular node |
(6.9→1.5) |
CR |
| 8 | 61/M |
Hypopharyngeal cancer |
Right supraclavicular node |
(8.9→no data) |
PR |
| 9 | 63/F |
Uterine cancer |
Right supraclavicular node |
(no data→4.1) |
PR |
| 10 | 58/F |
Breast cancer |
Right supraclavicular node |
(9.3→3.5) |
PR |
| 11 | 77/F |
Pancreatic cancer |
Left supraclavicular node |
(12.5→11.4) |
SD |
| 12 | 73/F |
Breast cancer |
Left supraclavicular node |
(no data→2.1) |
CR |
Assessment of therapeutic response
Most of the 12 patients underwent positron emission tomography-computed tomography (PET-CT) examination before and 3-8 months after KORTUC-SC to evaluate the functional therapeutic response [4, 5]. By PET-CT examination, the SUV-max of the FDG accumulation decreased remarkably. The therapeutic effect was evaluated good. The final therapeutic effect of the lesion was assessed according to the Revised RECIST guideline (version 1.1) [6], and patient monitoring and tumor assessment were performed once a month. Subsequently, patients underwent PET-CT, CT, or MRI examinations every 6 months if possible. Treatment-related complications were assessed in detail to evaluate the feasibility of the approach according to the Common Terminology Criteria for Adverse Events (CTCAE criteria Version 4.0). Patients were followed for at least 12 months.
Formulation example
One syringe (2.5 mL) of hyaluronic acid preparation with a 1% w/v concentration of sodium hyaluronate (ARTZ Dispo, Seikagaku Corporation, Tokyo, Japan) was used. This contained 25 mg of sodium hyaluronate, 2.5 mg of L-methionine, sodium chloride, potassium phosphate, crystalline sodium dihydrogen phosphate, and an isotonizing agent. The preparation was a colorless, transparent, viscous, aqueous solution having a pH of 6.8 to 7.8, a specific osmotic pressure of 1.0 to 1.2 (relative to physiological saline), and an average molecular weight of 600,000 to 1.2 million. To this, 0.5 mL of a 3% w/v solution of hydrogen peroxide (Oxydol, Ken-ei Pharmaceutical Co. Ltd., Osaka, Japan) was added immediately before use, and the solution was mixed well to prepare the radiosensitizer. This final sensitizer had a sodium hyaluronate concentration of 0.83% and a hydrogen peroxide concentration of approximately 0.5%.
ResultsTop
The treatment was well tolerated, with a minimum of adverse effects. Four (33.3%) patients showed local pain or flare at the injection site as an acute phase complication, but they were cured by local treatment within a few days; they were evaluated as having Grade I complications. Three (25%) patients showed fibrosis and sclerosis of the skin and subcutaneous tissue at the treatment site as Grade II chronic phase complications, and they are continuing to be followed.
The treatment outcome was satisfactory, and the adverse events were within the acceptable range. Of the 12 patients, 4 (33.3%) showed complete response (CR), 5 (41.6%) showed partial response (PR), 2 (16.6%) showed stable disease (SD), and 1 (8.3%) showed progressive disease (PD). The overall survival rate was 83% at 1 year (10/12 patients) and 75% at 2 years (9/12 patients). Representative PET-CT or CT examinations are shown in Figures 2-5. The mean follow-up period of the patients at the end of February 2011 was 35.1 months. 8 patients remain alive.
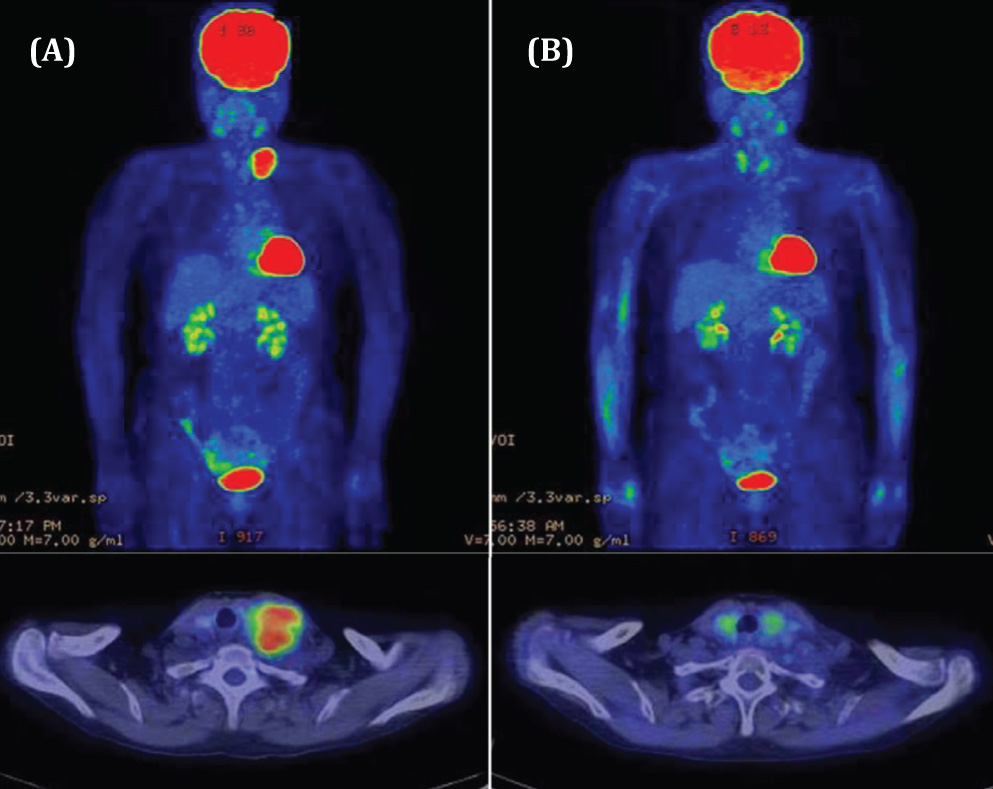
Figure 2 Therapeutic result evaluated by PET-CT examination (Patient 1 in Table 1): A 75-year-old woman with metastasis of hepatocellular carcinoma to the left supraclavicular lymph node (A) FDG-PET, PET-CT just prior to treatment. (B) FDG-PET, PET-CT 3 months following the KORTUC II treatment. The SUVmax of FDG accumulation at the left supraclavicular lymph node has decreased from 7.8 to 2.0. The minor axis of the target lymph node has decreased to 21 mm. The therapeutic effect is assessed as PR (partial response).
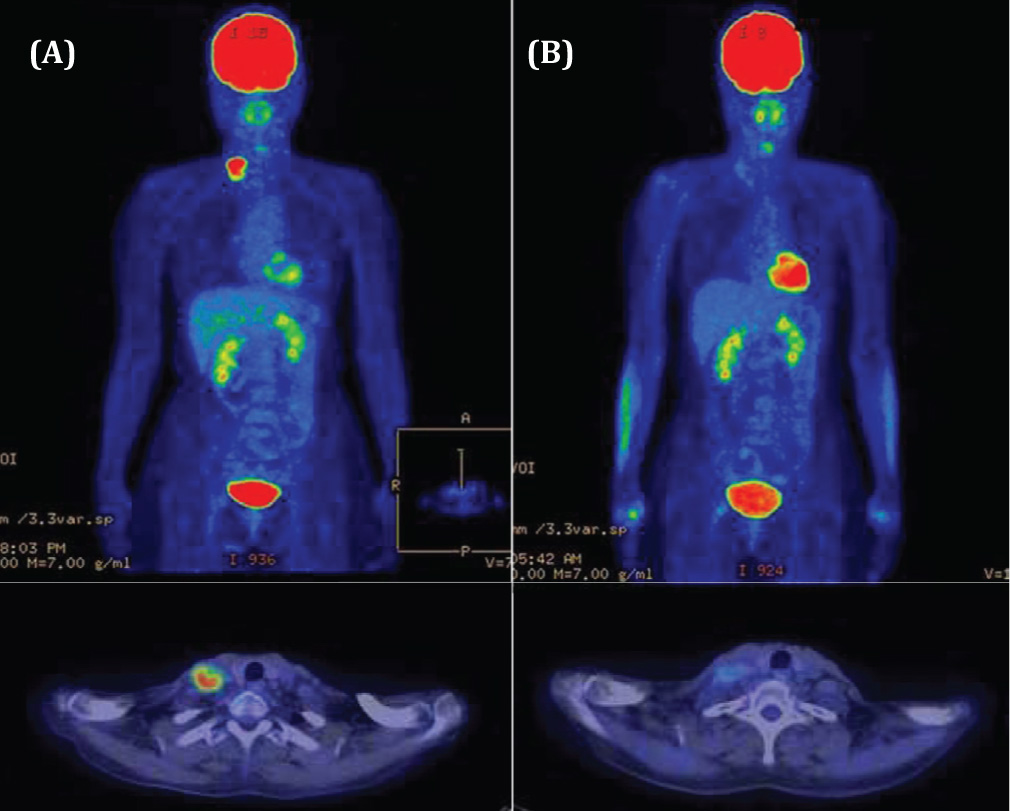
Figure 3 Therapeutic result evaluated by PET-CT examination (Patient 3 in Table 1): A 56-year-old woman with metastasis of breast cancer to the right supraclavicular lymph node (A) FDG-PET, PET-CT just prior to treatment. (B) FDG-PET, PET-CT 1 month following the KORTUC II treatment. The SUVmax of FDG accumulation at the right supraclavicular lymph node has decreased from 10.7 to 2.5. The minor axis of the target lymph node has decreased to unmeasurable size on the CT image. The therapeutic effect is assessed as CR (complete response).
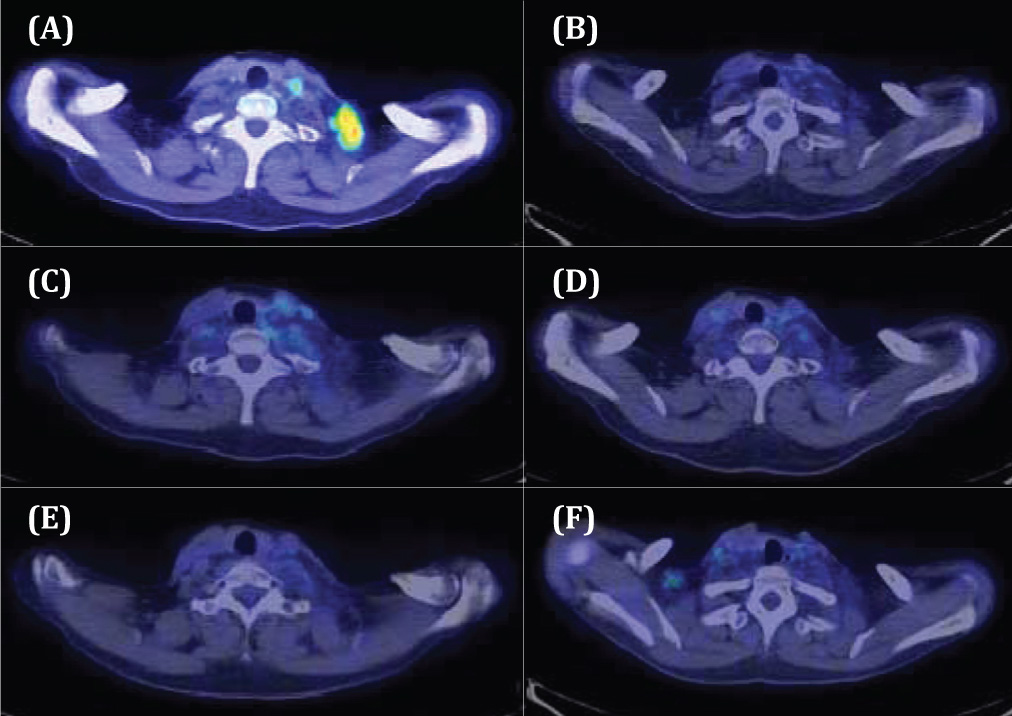
Figure 4 Therapeutic result evaluated by PET-CT examination (Patient 5 in Table 1): A 61-year-old woman with metastasis of breast cancer to the left supraclavicular lymph node (A) PET-CT just prior to treatment. (B) PET-CT 8 months following the KORTUC II treatment. (C) PET-CT 11 months later. (D) PET-CT 14 months later. (E) PET-CT 18 months later. (F) PET-CT 24 months later. The SUVmax of FDG accumulation at the left supraclavicular lymph node has decreased from 5.7 to 2.2. The minor axis of the target lymph node has decreased to unmeasurable size on the CT image. The therapeutic effect is assessed as complete response (CR). Since then, the patient has not experienced recurrence at the treatment site.
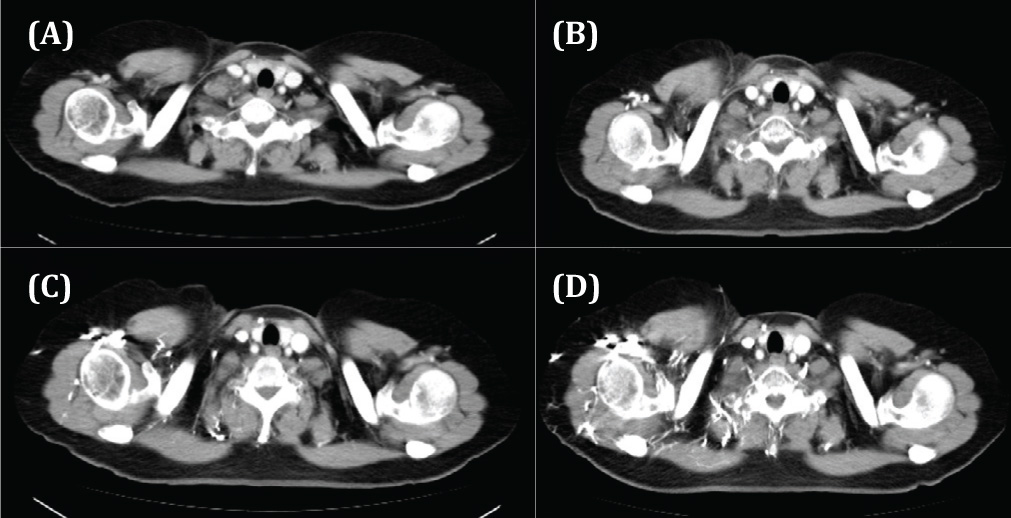
Figure 5 Therapeutic result evaluated by CT examination (Patient 9 in Table 1): A 63-year-old woman with metastasis of uterine cancer to the right supraclavicular lymph node (A) Contrast enhanced CT just prior to treatment. (B) Contrast-enhanced CT 5 months following the KORTUC II treatment. (C) Contrast enhanced CT 20 months following the KORTUC II treatment. (D) Contrast enhanced CT 29 months following the KORTUC II treatment. The minor axis of the right supraclavicular lymph node is 22 mm at pretreatment. After treatment, the right supraclavicular lymph node is decreased to unmeasurable size. The therapeutic effect is assessed as complete response (CR). Since 20 months later, the right supraclavicular lymph node can no longer be identified. The patient has not experienced any further evidence of disease.
DiscussionTop
Advanced diagnostic methods have resulted in earlier detection of recurrent cancers and/or distant metastases when they are still limited to a single site/organ (oligometastasis) [7]. For these cases, radiotherapy is considered to be a reliable method of treatment. In fact, some patients with oligometastasis have been reported to be cured by radiotherapy alone [7-9]. However, radiation therapy is not effective in all patients, particularly when the tumor contains many hypoxic tumor cells or high levels of antioxidative enzymes [10, 11].
Most radiation therapy is performed using X-rays or high-energy electron beams from a Linac; however, these forms of radiation are not ideal in terms of their low-linear energy transfer (LET) characteristics. Almost two-thirds of radiation-induced damage to cancer cells caused by these low-LET radiation sources results from the production of reactive oxygen species (ROS) in irradiated cells [12]. Therefore, oxygen is essential for oxidation of radicals produced by irradiation. However, antioxidative enzymes such as peroxidase and catalase scavenge the radicals produced by low-LET irradiation, resulting in a decreased irradiation effect. Peroxidase and catalase are abundant in most tumor tissues, as demonstrated by the fact that intrinsic peroxidase must be blocked before immunohistochemical staining of fresh tumor tissue [10, 11].
The low-LET radioresistant tumor cells exist mostly in hypoxic regions of the tumor. In the hypoxic state, DNA damage induced by radiation is not oxidized. Moreover, ROS produced in cancer cells by radiation are eliminated by antioxidative enzymes, and this appears to impede apoptosis induced by dysfunction of the mitochondrial membrane potential and/or lysosomal membrane permeability.
Although various radiosensitizing treatments for low-LET radioresistant neoplasms have been developed [13-17], a clinically applicable method has not yet been established. Many studies have examined the efficacy of increasing tissue oxygen concentration to promote oxidation of radicals produced by low-LET irradiation to cancer cells/tissue, but few therapeutic gains have thus far been observed [18-20]. Besides increasing tissue oxygen concentrations, it is important to inactivate antioxidative enzymes, such as peroxidase and catalase, which protect cancer cells from oxidative stress.
Hydrogen peroxide is the only agent that both inactivates antioxidative enzymes and produces oxygen when applied topically to tumor tissue. The method targets intrinsic peroxidase and catalase present in the tumor tissue. Therefore, KORTUC II is considered to be a new enzyme-targeting radiosensitization treatment for almost every type of low-LET radioresistant neoplasm. Indeed, the combined use of sodium hyaluronate and hydrogen peroxide preserved the oxygen concentration in tumor tissue for more than 24 hours, while intratumoral injection of hydrogen peroxide alone resulted in rapid lowering of oxygen concentrations in the tumor tissue.
Radiotherapy using this radiosensitizer can be implemented by first applying the radiosensitizer to the area of the tumor to be irradiated, and then preferably using a Linac to irradiate the affected part with X-rays or electron beams. With respect to use of the agent, it is essential to avoid direct injection of this agent into blood vessels and to confirm even distribution of oxygen micro-bubbles throughout the tumor tissue using ultrasonographic or CT guidance. Hence, KORTUC II can be thought of as a new image-guided radiosensitization method.
Kim et al. pronounced the better therapeutic effect of chemoradiotherapy for the supraclavicular lymph node metastasis 2012 [21]. The reason of the better therapeutic effect is that chemotherapy was effective for all the patients. At the moment, the treatment outcome is satisfactory and the adverse events are within the acceptable range. Since both the number of patients treated with this agent and the observation period length are insufficient, well-designed and randomized clinical trials are needed to establish the therapeutic efficacy of KORTUC-SC.
Since this radiosensitizer is safe and less expensive than other methods, and it can be applied for almost every type of low-LET radioresistant neoplasm, it may have the potential for worldwide and immediate use.
ConclusionTop
Based on the findings of imaging studies, KORTUC II treatment for chemotherapy-resistant supraclavicular lymph node metastasis shows remarkable therapeutic effects. KORTUC II represents a novel form of treatment using this radiosensitizer containing hydrogen peroxide and sodium hyaluronate that is injected intratumorally mainly under ultrasonographic guidance. The treatment was well tolerated with a minimum of adverse effects such as local pain at the injection site. The treatment outcome was satisfactory. The cost of the therapy is far less than that of the molecular-targeting therapy currently used, such as cetuximab. Well-designed, prospective, randomized, clinical trials are needed to establish the therapeutic efficacy of KORTUC-SC.
Funding
This work was partially supported by a grant-in-aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science and Technology.
Conflict of interest
The authors wish to express that they have no conflict of interest.
ReferencesTop
[1] Ogawa Y, Kubota K, Ue H, Tadokoro M, Matsui R, et al . (2011) Safety and effectiveness of a new enzyme-targeting radiosensitization treatment (KORTUC II) for intratumoral injection for low-LET radioresistant tumors. Int J Oncol 39:553-560. Article Pubmed
[2] Ogawa Y, Kubota K, Ue H, Tadokoro M, Matsui R, et al. (2007) Development and clinical application of a new radiosensitizer containing hydrogen peroxide and hyaluronic acid sodium for topical tumor injection- a new enzyme-targeting radiosensitization treatment, KORTUC II (Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, Type II). Strahlenther Onkol 183:100-101.
[3] Ogawa Y, Kubota K, Ue H, Kataoka Y, Tadokoro M, et al. (2009) Phase I study of a new radiosensitizer containing hydrogen peroxide and sodium hyaluronate for topical tumor injection: a new enzyme-targeting radiosensitization treatment, Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas, Type II (KORTUC II). Int J Oncol 34: 609-618. Article Pubmed
[4] Wang Y, Zhang C, Liu J, Huang G (2012) Is 18F-FDG PET accurate to predict neoadjuvant therapy response in breast cancer? A meta-analysis. Breast Cancer Res Treat 131:357-369. Article Pubmed
[5] Zucchini G, Quercia S, Zamagni C, Santini D, Taffurelli M, et al. (2013) Potential utility of early metabolic response by 18F-2-fluoro-2-deoxy-D-glucose-positron emission tomography/computed tomography in a selected group of breast cancer patients receiving preoperative chemotherapy. Eur J Cancer 49:1539-1545. Article Pubmed
[6] Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, et al. (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205-216. Article Pubmed
[7] Niibe Y, Kuranami M, Matsunaga K, Takaya M, Kakita S, et al. (2008) Value of high-dose radiation therapy for isolated osseous metastasis in breast cancer in terms of oligo-recurrence. Anticancer Res 28:3929-3932. Article Pubmed
[8] Milano MT, Zhang H, Metcalfe SK, Muhs AG, Okunieff P (2009) Oligometastatic breast cancer treated with curative-intent stereotactic body radiation therapy. Breast Cancer Res Treat 115:601-608. Article Pubmed
[9] Niibe Y, Hayakawa K (2010) Oligometastases and oligo-recurrence: the new era of cancer therapy. Jpn J Clin Oncol 40:107-111. Article Pubmed
[10] Ogawa Y, Nishioka A, Hamada N, Terashima M, Inomata T, et al. (1997) Immunohistochemical study of c-fos-positive lymphocytes infiltrated into human squamous cell carcinomas of the head and neck during radiation therapy and its clinical significance. Clin Cancer Res 3:2301-2307. Article Pubmed
[11] Ogawa Y, Nishioka A, Hamada N, Terashima M, Inomata T, et al. (1997) Expression of fas (CD95/APO-1) antigen induced by radiation therapy for diffuse B-cell lymphoma: immunohistochemical study. Clin Cancer Res 3:2211-2216. Article Pubmed
[12] Jette DC, Wiebe LI, Chapman JD (1983) Synthesis and in vivo studies of the radiosensitizer 4-[82Br]bromomisonidazole. Int J Nucl Med Biol 10:205-210. Article Pubmed
[13] Lawrence TS, Blackstock AW, McGinn C (2003) The mechanism of action of radiosensitization of conventional chemotherapeutic agents. Semin Radiat Oncol 13:13-21. Article Pubmed
[14] Böhm L, Roos WP, Serafin AM (2003) Inhibition of DNA repair by Pentoxifylline and related methylxanthine derivatives. Toxicology 193:153-160. Article Pubmed
[15] Milas L (2001) Cyclooxygenase-2 (COX-2) enzyme inhibitors as potential enhancers of tumor radioresponse. Semin Radiat Oncol 11:290-299. Article Pubmed
[16] Choy H, Milas L (2003) Enhancing radiotherapy with cyclooxygenase-2 enzyme inhibitors: a rational advance? J Natl Cancer Inst 95:1440-1452. Article Pubmed
[17] Overgaard J (1994) Clinical evaluation of nitroimidazoles as modifiers of hypoxia in solid tumors. Oncol Res 6:509-518. Article Pubmed
[18] Gerlach NL, Barkhuysen R, Kaanders JH, Janssens GO, Sterk W, et al. (2008) The effect of hyperbaric oxygen therapy on quality of life in oral and oropharyngeal cancer patients treated with radiotherapy. Int J Oral Maxillofac Surg 37:255-259. Article Pubmed
[19] Overgaard J (2007) Hypoxic radiosensitization: adored and ignored. J Clin Oncol 25:4066-4074. Article Pubmed
[20] Williamson RA (2007) An experimental study of the use of hyperbaric oxygen to reduce the side effects of radiation treatment for malignant disease. Int J Oral Maxillofac Surg 36:533-540. Article Pubmed
[21] Kim JY, Kim JY, Kim JH, Yoon MS, Kim J, et al. (2012) Curative chemoradiotherapy in patients with stage IVB cervical cancer presenting with paraortic and left supraclavicular lymph node metastases. Int J Radiat Oncol Biol Phys 84:741-747. Article Pubmed


