Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 4, Issue 4, September 2016, Pages 31–37
Original researchOpen Access
Newly isolated compounds from West African Sorghum bicolor leaf sheaths Jobelyn® show potential in cancer immunosurveillance
- 1 Department of Pharmacology, Lagos State University, College of Medicine (LASUCOM), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 2 Department of Hematology and Blood Transfusion, Lagos State University Teaching Hospital (LASUTH), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 3 Department of Physiology, Lagos State University, College of Medicine (LASUCOM), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos Nigeria
- 4 Department of Pharmaceutical Chemistry, Obafemi Awolowo University, Ile-Ife, Osun State 220005, Nigeria
- 5 Health Forever Products Inc., Lagos, Nigeria
*Corresponding author: Samira BL .Makanjuola, PhD., Department of Pharmacology, Lagos State University, College of Medicine (LASUCOM), 1-5 Akinjobi Way, Ikeja, G.R.A Ikeja, Lagos, Nigeria. Tel.: +234 80 96633500; E-mail: sbatistalobo@hotmail.com
Received 15 June 2016 Revised 10 August 2016 Accepted 19 August 2016 Published 27 August 2016
DOI: http://dx.doi.org/10.14312/2052-4994.2016-6
Copyright: © 2016 Makanjuola SBL, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Jobelyn®, a West African pharmaceutical product derived from Sorghum bicolor leaf sheaths has been shown to possess strong anti-tumour and anti-inflammatory properties. This study aims to evaluate the expression of cell surface markers CD69 on activated natural killer (NK) cells; natural killer T (NKT) cells; and T cells from human peripheral blood mononuclear cells (PBMC) upon treatment with Jobelyn® fractions using flow cytometry. Blood was collected from 3 donors, PBMC were isolated and plated with each specific fraction: crude extracts (J); ethyl acetate (JE); n-butanol (JB); secondary compounds from JE (JE5; JE6); purified and semi-purified compounds from JE5 (P8 and P9) at specific concentrations (2.5-500 µg/ml). For the crude extracts, JE was the most active showing significant expression of CD69 on NK-(P < 0.001), T- (P < 0.0001), and NKT- treated cells (P < 0.0001). Secondary compound, JE5, of JE also showed significant CD69 expression on NK- (P < 0.018) and T-treated cells (P < 0.027), but not on NKT-treated cells (P > 0.084). Similarly, the semi-purified compound P8, from JE5 showed increased expression of CD69 on NK- (P0.023); T- (P < 0.001), and NKT-treated cells (P < 0.007). Evidence of innate effector cells activation by ethanolic extracts of Jobelyn® suggests that this variety of Sorghum may be able to mediate direct cell cytotoxicity supporting the control and clearance of a number of tumour cells.
Keywords: CD69; NK cells; T cells; NKT cells; innate immunity; flow cytometry
IntroductionTop
Sorghum bicolor is a plant that has been cultivated in southern Africa for over 3000 years. This is a cane like grass, up to 6 m tall with large branched clusters of grains. The individual grains are around 3-4 mm in diameter and vary in colour from pale yellow through reddish brown depending on the soil where it is cultivated. The leaves resemble those of maize, and they sometimes roll over forming sheaths. The West African region is home to a unique variety of Sorghum bicolor, which accumulate high levels of 3-deoxyanthocyanin pigments in their non-grain tissue [1]. The intensively colored leaf sheaths of this wild variety of Sorghum bicolor are found within the Nigerian flora and have been formulated into a commercial pharmaceutical product under the name Jobelyn®.
Since Sorghum bicolor accumulates large quantities of the rare 3-deoxyanthocyanin pigments [2, 3], it has gained interest as potential natural food colorants. However, recent reports demonstrate that the West African Sorghum possesses anti-tumour effects [4] as well as chemo-preventive [5] and anti-inflammatory properties [6, 7].
Anti-tumour potentials of Sorghum were shown through inhibited proliferation of esophageal and colon cancer cell lines as a result of 3-deoxyanthocyanidins induced activity of the phase II protective enzyme, quinine reductase [5]. Whereas, a study carried out on the aqueous and ethanol extracts of Sorghum leaf sheaths from South-West Nigeria has demonstrated activation of human CD3+CD56- T cells through up-regulation of CD69 expression suggesting that Sorghum is capable of supporting immune defenses [7].
Separate studies on the structure of 3-deoxyanthocyanidins in the West African Sorghum identified several peaks including an unusual fused-ring apigeninidin derivative; large quantities of apigenin and most interestingly, an apigeninidin-apigenin condensation product [8]. Such observations correlate with previous reports on the co-existence of apigeninidin and apigenin in specific Sorghum varieties [9]. The characterization of apigenin and apigeninidin derivatives in the West African Sorghum is interesting as these anthocyanin’s pigments are popular as health-promoting phytochemicals capable of inducing cytotoxicity on human cancer cells [4].
In line with this, the present study aims to evaluate the ability of Sorghum bicolor Jobelyn® in providing innate immunity against tumour cells through the activation NK cells as well as NKT cells and T cells. Activated NK cells, T cells and NKT cells will be evaluated for CD69 expression following treatment of human peripheral blood with several Sorghum fractions in order to assess for their ability to activate the innate immunity.
Materials and methodsTop
Sorghum bicolor leaf sheaths grown in West Africa were harvested, processed, packaged and supplied under the trade name Jobelyn® by Health Forever Product Inc. Lagos, Nigeria and Hains Herbal Products LLC, USA. Sorghum bicolor is approved as a food supplement by the National Agency for Food and Drug Administration Control (NAFDAC) of Nigeria. This study was approved by the Lagos State University Teaching hospital (LASUTH) Ethics Committee.
Sample extraction
The dried powdered leaf sheath of Sorghum bicolor was extracted with 50% aqueous ethanol for 24 h following vigorous agitation. The extract was subsequently filtered through cotton wool and concentrated in vacuo to give the crude extract designated as J. This was then dissolved in 80% aqueous ethanol and partitioned into ethyl acetate (EtOAc) and n-butanol successfully. The EtOAc, n-butanol and aqueous fractions were concentrated in vacuo to give fractions labeled JE, JB and JA, respectively (Figure 1).
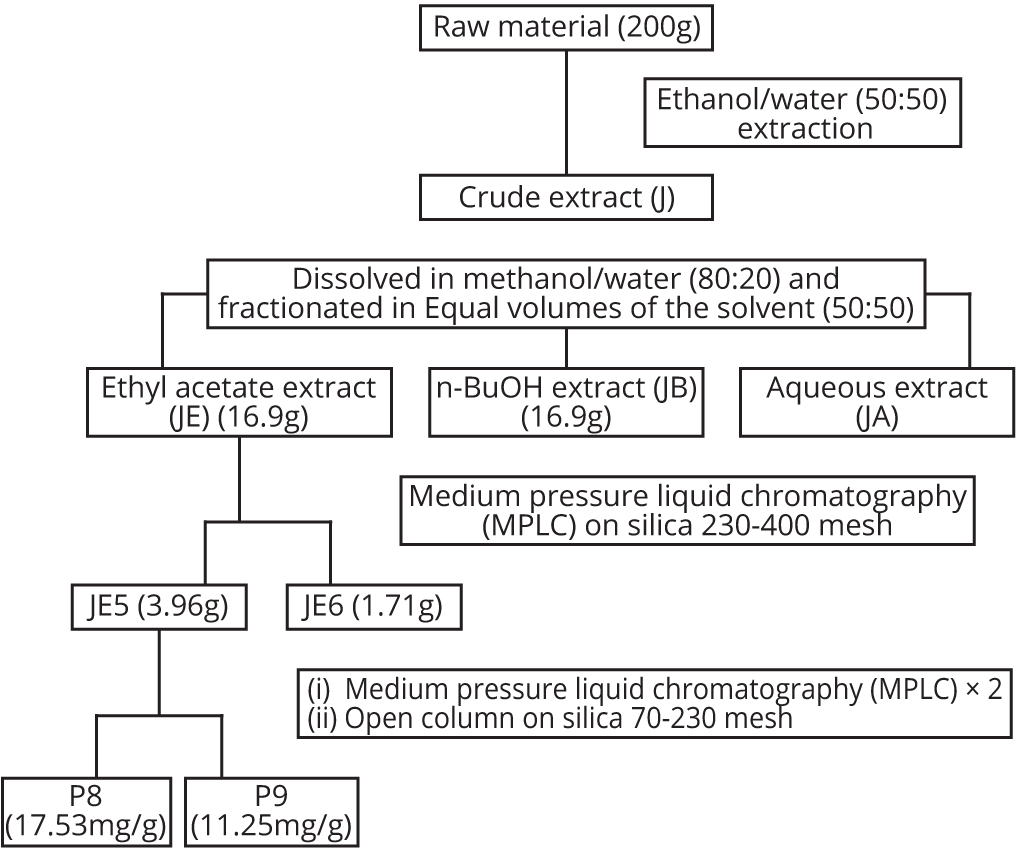
Liquid and thin layer chromatography separation (JE5 to JE6)
Adopting a Medium Pressure Liquid Chromatography (MPLC) technique, JE was sub-fractioned on a silica gel (230-400 mesh) column using a gradient of n-hexane (200 ml), a doubling gradient of 100 ml each of EtOAc-n-hexane, EtOAc, doubling gradient of methanol (MeOH)-EtOAc and finally MeOH. Fractions of 15 ml each were collected in test tubes and monitored by thin layer chromatography (TLC) with normal phase silica gel using ether/hexane (96:4) and 20% EtOAc in ether as the solvent systems. The fractions which had the same TLC characteristics were bulked as appropriate and concentrated in vacuo to give 2 major fractions labelled JE5 and JE6 (Figure 1).
Repeated column chromatographic separation of P8 and P9
JE5 was subjected to repeated column chromatography on Sephadex LH-20 column using n-hexane (100 ml), a doubling gradient of EtOAc-n-hexane and the EtOAc or a doubling gradient of MeOH- EtOAc and then MeOH, followed by open column chromatography on silica (70-230 mesh) with a gradient of ether in hexane to produce two further fractions (monitored on a thin layer plate to be relatively pure components) which were concentrated in vacuo and labelled P8 and P9, respectively (Figure 1).
Mass spectroscopy analysis of semi- and purified compounds
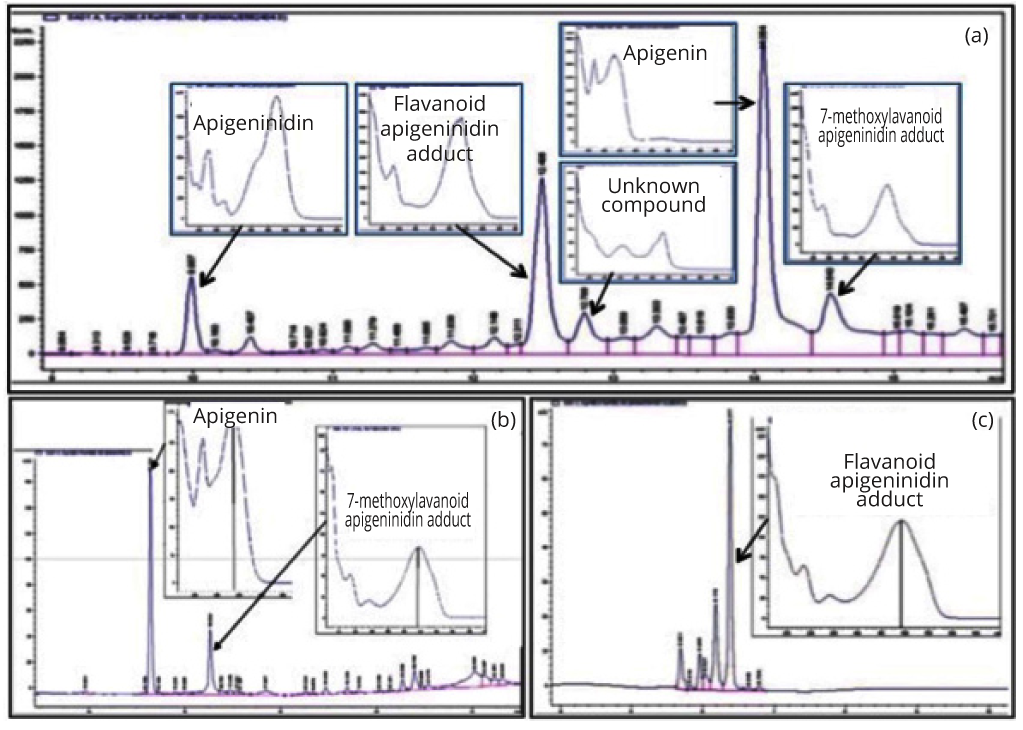
The absorbance profiles of JE5, P8 and P9 isolated from HPLC fractions showed five major peaks for JE5, two major peaks in P8 and a single peak in P9. The fractions were further analysed using an MDS Sciex API QStar Pulsar mass spectroscopy with electrospray ionization (AB SCIEX, Foster City, California, USA) under the following conditions: (1) ions source gas, 40 arbitrary units (au); (2) curtain gas flow rate, 20 au; (3) gas used, nitrogen; (4) ion spray voltage, 4500 V; (5) detector (MCP), 2150 V; ND (6) syringe pump flow, 6 µl/min. Identification of the compounds was based on matching HPLC retention profile, UV-Vis spectra analysis and MS data with authentic standards. The unknown compounds were fragmented at 33% relative collision energy , sheath gas flow rate of 20 au, and auxillary gas flow rate of 10 au, to further aid in structure determination. This procedure has been described recently by Geera and colleagues [8]. Briefly, the mass spectroscopy data from one of the peaks in P8 was identified as apigenin when matched against apigenin standard. The second compound in P8 and the single compound in P9 were initially identified as dimeric flavonoid molecules, differing from each other by one methyl group. Using both UV-Vis data and LC elution profiles, the two compounds were suggested to be 3-deoxyanthocyanidin dimers. Based on the structure, the second compound in P8 was identified as 7-methoxyflavone-apigeninidin adduct while that in P9 was identified as flavonoid-apigeninidin adduct [8]. To date four of the five major compounds in JE5 have now been characterized including apigenin and apigeninidin as well as 7-methoxylflavanoid apigeninidin and flavonoid-apigeninidin adducts, one compound remains unknown as described in Table 1 and Figure 2.
| Sample | HPLC peak ID | Compound ID | λmax (nm) | [M+H]+ | Major fragments[M+H]+ |
| P8 | 1 | Apigenin | 335 | 271 | - |
| 2 | 7-methoxylavanoid-apigeninidin adduct | 479 | 523 | 384, 269, 255 | |
| P9 | Flavanoid-apigenidin adduct | 480 | 509 | 384, 255 | |
| JE-5 | 1 | Apigeninindia | 475 | 255 | - |
| 2 | Flavanoid-apigenidin adduct | 480 | 509 | 284, 255 | |
| 3 | unknown | 480 | 509 | Not fragmented | |
| 4 | Apigenin | 335 | 271 | - | |
| 5 | 7-methoxylavanoid-apigeninidin adduct | 479 | 523 | 384, 269 |
To prepare sterile liquid fractions of the product suitable for the addition to live cell cultures, 0.5 gm of the leaf sheath powder was added to 5 ml physiological saline for the aqueous extraction method and to 5 ml 50% ethanol for the ethanol extraction method, in 15 ml vials. The vials were vortexed and placed on a rocker at room temperature for 1 h. Solids were removed by centrifugation at 900 g for 10 min, and the resulting liquid extracts were filtered through a 0.22 micron cellulose acetate fiber to produce sterile stock solution, from which serial dilutions were made in physiological saline. The serial dilutions added to live cell cultures were such that more than 0.5% of the ethanol present in the cell cultures. This dose of ethanol was considered to have no physiological effect in the assay, as verified by previous control tests [7].
PBMC preparation for flow cytometry
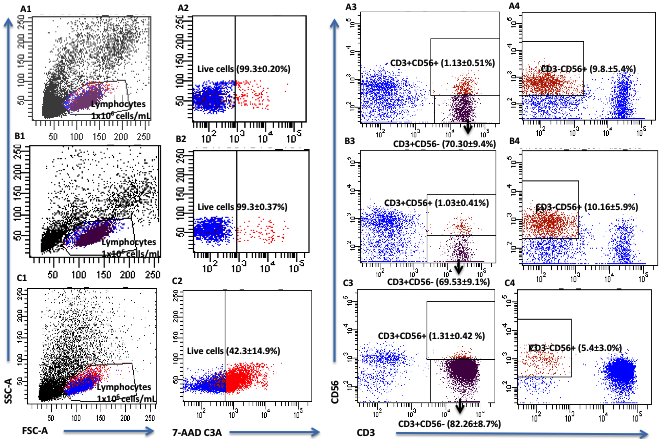
PBMC from three donors question: Were these normal healthy adults? If so, text should read accordingly were isolated and counted (Guava PCA and Cytosoft software version 6.0.3) and re-suspended at a concentration of 1 × 106 cells/ml in RPMI media with 10% FBS. The cells were plated, 1ml per well and then each compound at one of the four specified concentrations: J, JE, JB and JA (500, 100, 10, 5 µg/ml); JE5 and JE6 (250, 100, 50, and 20 µg/ml); and P8 and P9 (100, 20, 5, 2.5 µg/ml) were added to the appropriate well. The cells were incubated with the compound for 18-20 h and then transferred to V-bottom 96-well plates, washed, and re-suspended in PBS buffer containing 1% bovine serum albumin and 0.02% NaN3; monoclonal antibodies were added and incubated in the dark at room temperature for 10 minutes. The monoclonal antibodies were CD3-PerCP (red), CD56-PE (orange) and CD69-FITC (green). The amounts used for each monoclonal antibody was previously titrated to provide optimal staining. The cells were washed and re-suspended in 0.05 ml PBS buffer containing 0.02% NaN3 and transferred into 5 ml falcon tubes each containing 0.4 ml of 1% formalin in PBS. Samples were collected on a FACS Canto (BD) instrumentation with a FACS Diva (BD) software (v6.1.3) according to various combinations of surface markers: NK (CD3-CD56+), NKT (CD3+CD56+) and T (CD3+CD56-). The combination of the surface markers were then followed by evaluation of the expression level (percent and or Median Fluorescent intensity) of the CD69 activation marker on each cell type. Analysis was performed using the service provider Flow Contract Site (Kirkland, WA, USA).
Statistical analysis
Analysis of variance on the CD69 expression between the different crude extracts was performed using one way ANOVA. Student’s t-test was performed for CD69 expression comparisons between the secondary compounds and semi-purified compounds. Levels of statistical significance between data sets were significant if the P-value was less than 0.05, highly significant if P < 0.01, and a very high significance if P < 0.001.
ResultsTop
The effect of Jobelyn® fractions on activation of NK cells, T cells, and NKT cells using the cell surface marker CD69
Three monocyte subtypes including CD3+ (T cells), CD3+CD56+ (NKT cells) and CD3-CD56+ (NK cells) were identified using flow cytometry analysis of lymphocyte populations from whole blood (Figure 3). The treatment of PBMC cultures with primary extracts showed that JE was the most active for all three cell types (NK, NKT, T cells) and JA was the least active test compound. JE-treated CD3-CD56+ NK cells showed approximately a 2-fold increase in CD69 expression compared to JA at the highest concentration of 500 µg/ml (P = 0.001). JE-treated CD3+ T cells showed approximately 2-fold increase in CD69 expression compared to J and JB and a 3-fold increase compared to JA (P = 0.0001). JE-treated CD3+CD56+ NKT cells also showed approximately 2-fold increase in CD69 expression compared to J and JA and a 3-fold increase compared to J (P = 0.0001). Such results suggest that the ethanol-based fraction of Jobelyn® (JE) is the most effective in activating NK cells, T cells and NKT cells (Figure 4, Table 2).
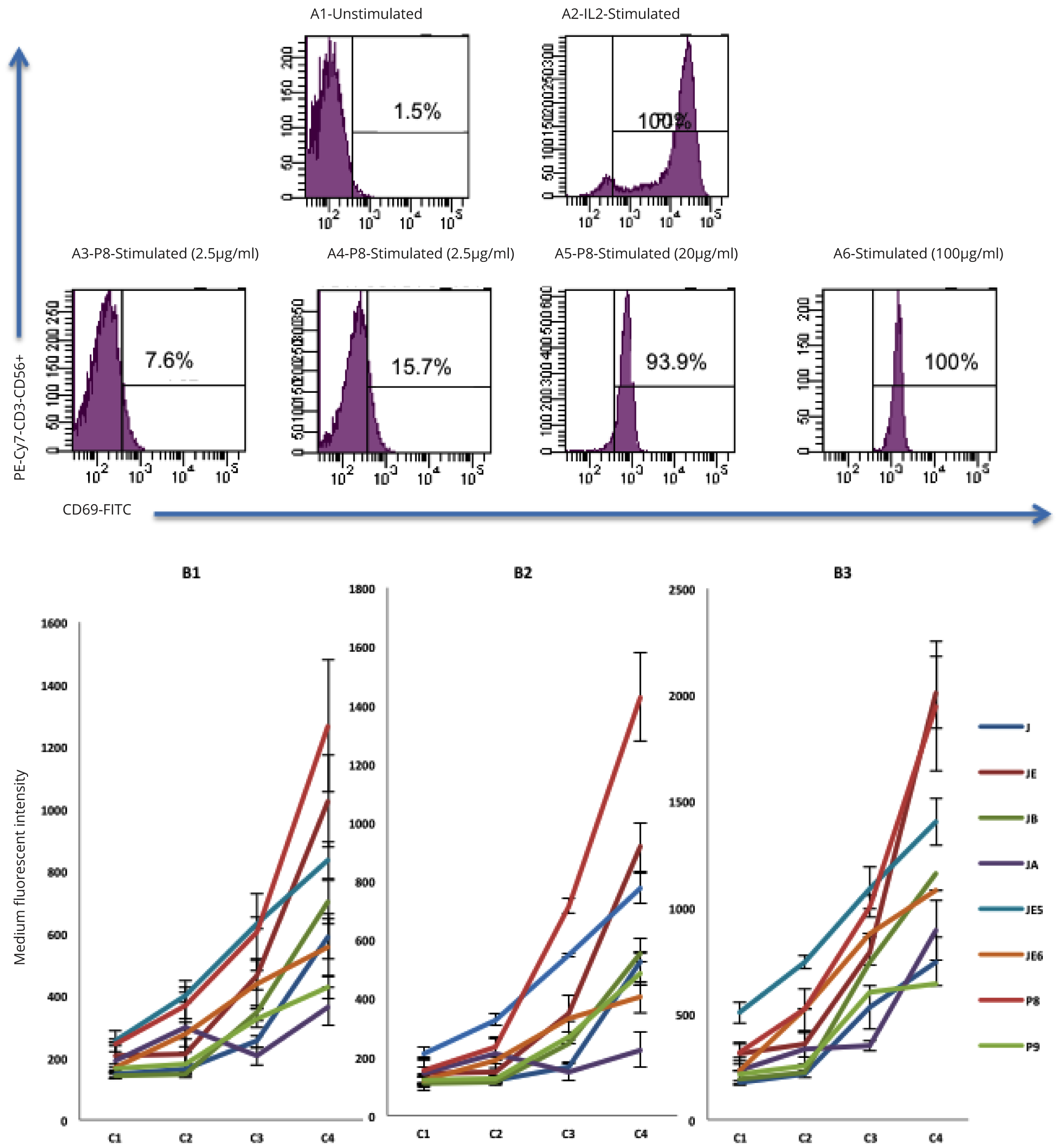
| Compound ID | CD69 CD3-CD56+ NK cells |
CD69 CD3+CD56- T cells |
CD69 CD3+CD56+ NKT cells |
|||
| Mean (MFI) | P-value | Mean (MFI) | P-value | Mean (MFI) | P-value | |
| J | 590 | 0.001 | 526 | 0.0001 | 7465 | 0.0001 |
| JE | 1024 | 916 | 2011 | |||
| JB | 700 | 555 | 1157 | |||
| JA | 364 | 224 | 890 | |||
| JE5 | 833 | 0.018 | 646 | 0.027 | 1400 | 0.084 |
| JE6 | 554 | 403 | 1080 | |||
| P8 | 1266 | 0.023 | 1427 | 0.001 | 1448 | 0.007 |
| P9 | 426 | 485 | 638 | |||
The secondary extract category included JE5 and JE6 derived from JE. JE5 showed greater CD69 expression (P = 0.018) on NK cells compared to JE6. Equally, the expression of CD69 on T cells following JE5 treatment was significantly higher compared to JE6 (P = 0.027). JE5 also showed an increase of CD69 expression compared with JE6 on NKT cells, but this was not significant (P = 0.084). Overall JE5 was the most effective in inducing NK, T and NKT cells stimulation (Figure 4, Table 2).
Semi-purified compounds P8 showed a 3-fold increase (P = 0.023) in the expression of CD69 on compared to P9 on NK cells. Equally, P8-treated T cells showed over a 2-fold increase (P = 0.001) in expression of CD69 compared to P9. P8-treated NKT cells also showed a significant increase (P = 0.007) in CD69 expression compared to P9. The semi-purified compound P8 is the most effective in inducing stimulation of NK cells, T cells and NKT cells stimulation (Figure 4, Table 2).
DiscussionTop
The impact of Sorghum bicolor Jobelyn® on the immune response has been previously described by Benson and colleagues [7]. The study showed activation of CD3+CD56+ NKT cells by Sorghum ethanol extract and activation of CD3-CD56+ NK cells by Sorghum aqueous extract. The selective properties of water-based and ethanol-based have prompted this study as it is evident that Sorghum contains multiple and potentially synergistic, active compounds [7].
In keeping with previous studies, here we were able to demonstrate that the Jobelyn® ethanol extracts (JE) not only activates CD3+CD56+ NKT cells but also CD3-CD56+ NK cells and CD3+CD56- T cells. Similarly, JE5, a secondary extract from JE also showed good expression of CD69 on activated T cells, NK cells and NKT cells. Moreover, P8, semi-purified compounds of JE5, showed major CD69 expression on treated T cells, NK cells and NKT cells.
The NK cells in the blood circulation are characterized as CD3-CD56+ lymphocytes, and are predominately negative for the activation of marker CD69. In the presence of suitable stimuli, CD69 expression will increase correlating with increased cytotoxic capacity [10]. CD69 is one of the earliest cell surface markers induced in resting T lymphocytes and NK cells with detectable surface expression of CD69 antigen occurring within 3 hours of stimulation and peaking in activated T lymphocytes and NK cells after 12 h [11]. CD69 expression has served as a marker to evaluate T lymphocytes and NK cells activation as it was found to be associated with expression of CD69 on high percentage of cells [12].
The CD3- CD56+ NK cells in the blood constitute a minor, yet highly potent effector cell population among NK cells [13]. Among the CD56+ CD3- NK cell surface antigens that are involved in cell activation are CD25, the high affinity interleukin-2 receptor α chain (IL-2Rα) [14, 15]; CD16, the low-affinity receptor for the Fc portion of immunoglobulin G that triggers antibody-dependent cellular cytotoxicity of NK cells [16]; and CD69, a cell surface molecule present on activated NK cells that triggers their spontaneous cytotoxicity [10]. The cytotoxic mechanisms of NK cells are crucial to many processes such as defending against pathogens and tumour [17, 18]. Equally, NKT cells represent a subset of T lymphocytes that share receptors with both conventional T cells and NK cells suggesting its crucial role in both pathogen recognition and lysis of target cells [19]. It is indeed encouraging that by characterizing the active compounds of Jobelyn®, we were able to identify its involvement in the immune modulation of NK cells as well as NKT cells and T cells and its possible relevance in processes associated with the control of tumour growth as NK cells mediate immediate short-lived responses by constitutively expressing a lytic machinery that directly lysis cancer cells [20].
In humans, there is evidence that NK cells play a role in immunosurveillance. An eleven-year follow-up survey shows that low NK activity is associated with an increased cancer risk [21]. For example, a decrease of NK cell activity is observed in patients with hereditary colorectal adenocarcinoma [22] and melanoma patients with metastatic disease [23]. Hence the effect of the ethanol-based Jobelyn® compounds on NK cell activity, especially P8, which is a purified compound composed by apigenin and 7-methoxylavanol-apigeninidin adduct [8] is indeed encouraging as it could assist in the control (chemo-protective) and clearance (anti-proliferative) of tumour cells.
Recent discovery of novel NK receptors and their ligands has uncovered the molecular mechanisms that regulate NK cells activation and function. Several activating NK cell receptors and co-stimulatory molecule have been identified to permit these cells to recognize tumour-infected cells. In vitro, NK cell cytotoxicity ability can be enhanced by cytokines such as interleukin-2 [24]. In vitro data using Jobelyn® equally enhanced activity of NK cells showing possible role as co-stimulatory molecule. What remains, is to evaluate the role of ethanol-based compounds of Jobelyn® in vivo and determine whether these extracts have the potential to induce in vivo activation of human NK cells.
It is inevitable to mention that the use of natural products in drug development is likely to provide many lead structures and these can be used as templates for the development of new compounds with enhanced biological properties. Although many disadvantages have been associated in using isolates as they take longer to develop into drugs, side effects and other problems which would require additional management as well as high cost of finished drugs which is usually out of reach to consumers in developing countries. The greatest advantage in the development of traditional medicine with evidence is enormous. The cost is affordable and it is readily available, with fewer side effects and other complications. Such advantages make traditional medicine use common amongst cancer patients, especially in developing countries such as Nigeria where the cost of treatment is deemed high. The reasons for the popularity of traditional medicine use amongst cancer sufferers include improving quality of life, supporting conventional cancer treatment, preventing recurrence, and eventually to prolong life. However, there is limited scientific evidence on the efficacy and safety of natural therapies. This study provides valuable evidence on the effectiveness Sorghum bicolor Jobelyn® in improving the immune response as well as targeting lytic mechanisms associated with NK cells. Although animal studies have been performed describing the high safety profile of Jobelyn® in rats [25], it remains to determine the possible hazards herbal consumption may pose on public health. This requires vigorous clinical trials to establish the efficacy or the presence of adverse effects in such compounds.
ConclusionTop
Jobelyn®, a pharmaceutical product derived from Sorghum bicolor leaf sheath has been fractionated into several compounds including the purified flavonoid-apigeninidin fraction (P9) and the semi-purified 7-methoxyflavone-apigeninidin and apigenin fractions (P8). Interestingly, P8 showed significant potential in inducing in vitro activation of human NK cells suggesting it could support crucial immune defense mechanisms as the ones required in the control and clearance of tumour cells.
Acknowledgement
S.M. performed data analysis and wrote the paper; Dosunmu D and Ajonuma L wrote the paper; Ogundaini A designed and performed fractionation experiments; Okubena O provided Sorghum bicolor extract. The authors would like to thank Flow Contract Site Laboratory, Kirkland USA, for performing flow cytometry experiments and Health Forever Products Inc., Lagos Nigeria for funding this project.
Conflicts of interest
Makanjuola SL, Dosunmu D, Ajonuma L are employees of the Lagos State University College of Medicine (LASUCOM), a State University for Medical Sciences; Ogundaini A is an employee of the Obafemi Awolowo University, a Federal University of Nigeria and Okubena O is an employee of Health Forever Products, Inc. The experiments were performed at Flow Contract Site Laboratory, Kirkland USA, an independent contract research laboratory specialised in natural products research.
ReferencesTop
[1]Kayodé AP, Nout MJ, Linnemann AR, Hounhouigan JD, Berghofer E, et al. Uncommonly high level of 3-deoxyanthocyanidins and antioxidant capacity in the leaf sheaths of dye Sorghum. J Agric Food Chem. 2011; 59(4):1178-1184.Article Pubmed
[2]Mazza G, Brouillard R. Recent developments in the stabilization of anthocyanins in food products. Food Chem. 1987; 25(3):207–225.Article
[3]Awika JM, Rooney LW. Sorghum phytochemicals and their potential impact on human health. Phytochemistry. 2004; 65(9):1199-1221.Article Pubmed
[4]Shih CH, Siu SO, Ng R, Wong E, Chiu LC, et al. Quantitative analysis of anticancer 3-deoxyanthocyanidins in infected sorghum seedlings. J Agric Food Chem. 2007; 55(2):254-259.Article Pubmed
[5]Yang L, Browning JD, Awika JM. Sorghum 3-deoxyanthocyanins possess strong phase II enzyme inducer activity and cancer cell growth inhibition properties. J Agric Food Chem. 2009; 57(5):1797–1804.Article Pubmed
[6]Burdette A, Garner PL, Mayer EP, Hargrove JL, Hartle DK. Anti-inflammatory activity of select sorghum (Sorghum bicolor) brans. J Med Food. 2010; 13(4):879–887.Article Pubmed
[7]Benson KF, Beaman JL, Ou B, Okubena A, Okubena O, et al. West African Sorghum bicolor leaf sheaths have anti-inflammatory and immune-modulating properties in vitro. J Med Food. 2013; 16(3):230-238.Article Pubmed
[8]Geera B, Ojwang LO, Awika JM. New highly stable dimeric 3-deoxyanthocyanidin pigments from Sorghum bicolor leaf sheath. J Food Sci. 2012; 77(5):C566–C572.Article Pubmed
[9]Dykes L, Seitz L M, Rooney W L, Rooney L. W. Flavanoid composition of red sorghum genotypes. Food Chem. 2009; 116(1):313-317.Article
[10]Moretta A, Poggi A, Pende D, Tripodi G, Orengo AM, et al. CD69-mediated pathway of lymphocyte activation: anti-CD69 monoclonal antibodies trigger the cytolytic activity of different lymphoidef fector cells with the exception of cytolytic T lymphocytes expressing T-cell receptor alpha/beta. J Exp Med. 1991; 174(6):1393-1398.Article Pubmed
[11]Testi R, Phillips JH, Lanier LL. T-cell activation via Leu 23 (CD69). J Immunol. 1989; 143(4):1123-1128.Article Pubmed
[12]Clausen J, Vergeiner B, Enk M, Petzer AL, Gastl G, et al. Functional significance of the activation-associated receptors of CD25 and CD69 on human NK-cells and NK-like T-cells. Immunobiology. 2003; 207(2):85-93.Article Pubmed
[13]Rosenberg SA, Lotze MT, Muul LM, Leitman S, Chang AE, et al. Observations on the systemic administration of autologous lymphokine activated killer cells andrecombinant interleukin-2 to patients with metastatic cancer. N Engl J Med. 1985; 313(23):1485-1492.Article Pubmed
[14]Nagler A, Lanier LL, Phillips JH. Constitutive expression of high affinity interleukin 2 receptors on human CD16-natural killer cells in vivo. J Exp Med. 1990; 171(5):1527–1533.Article Pubmed
[15]Siegel JP, Sharon M, Smith PL, Leonard WJ. The IL-2 receptor beta chain (p70): role in mediating signals for LAK, NK, and proliferative activities. Science. 1987; 238(4823):75–78.Article Pubmed
[16]Trinchieri G. Biology of natural killer cells. Adv Immunol. 1989; 47:187-376.Article Pubmed
[17]Smyth MJ, Hayakawa Y, Takeda K, Yagita H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat Rev Cancer. 2002; 2(11):850-861.Article Pubmed
[18]Lodoen MB and Lanier LL. Natural killer cells as an initial defense against pathogens. Curr Opin Immunol. 2006; 18(4):391–398.Article Pubmed
[19]Alter G, Malenfant JM, Altfeld M. CD107a as a functional marker for identification of natural killer cell activity. J Immunol Methods. 2004; 294(1-2):15-22.Article Pubmed
[20]Cheng M, Chen Y, Xiao W, Sun R, Tian Z. NK cell-based immunotherapy for malignant diseases. Cell Mol Immunol. 2013; 10(3):230–252.Article Pubmed
[21]Imai K, Matsuyama S, Miyake S, Suga K, Nakachi K. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: an 11-year follow-up study of a general population. Lancet. 2000; 356(9244):1795–1799.Article Pubmed
[22]Warren RP, Stembridge AM, Gardner EJ. Deficient immune function of peripheral blood mononuclear cells from patients with Gardner syndrome. Clin Exp Immunol. 1985; 60 (3):525–531.Article Pubmed
[23]Jović V, Konjević G, Radulović S, Jelić S, Spuzić I. Impaired perforin-dependent NK cell cytotoxicity and proliferative activity of peripheral blood T cells is associated with metastatic melanoma. Tumori. 2001; 87(5):324–329.Pubmed
[24]Wu J, Lanier LL. Natural Killer cells and cancer. Adv Cancer Res. 2003; 90:127-156.Pubmed
[25]Eniojukan JF, Bolajoko AA. Toxicological Profiles of Commercial Herbal Preperation, Jobelyn. International Journal of Health Research. 2009; 2(4):369-374.Article



