Journal of Hematology and Therapeutics
An International Peer-Reviewed Open Access Journal
ISSN 2397-8694


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Hematology and Therapeutics
Volume 2, Issue 1, June 2020, Pages 1–7
Original researchOpen Access
Novel biomarkers for tyrosine kinase inhibitor resistance in chronic myeloid leukemia patients: Bioactive sphingolipids
-
Melis Kartal Yandim1,* PhD, Ilknur Kozanoglu3 MD, Hakan Ozdogu3 MD, Ozden Piskin4 MD, Mehmet Ali Ozcan4 MD, Guray Saydam5 MD, Fahri Sahin5 MD, Ferit Avcu6,7 MD, Ali Ugur Ural8 MD, Ali Unal9 MD and Yusuf Baran1,*
 PhD
PhD
- 1 Department of Molecular Biology & Genetics, Faculty of Science, Izmir Institute of Technology, Izmir, Turkey
- 2 Department of Medical Biology, Faculty of Medicine, Izmir University of Economics, Izmir, Turkey
- 3 Department of Hematology, Faculty of Medicine, Baskent University, Adana, Turkey
- 4 Department of Hematology, Faculty of Medicine, Dokuz Eylul University, Izmir, Turkey
- 5 Department of Hematology, Faculty of Medicine, Ege University, Izmir, Turkey
- 6 Department of Hematology, Faculty of Medicine, Gulhane Military Medical School, Ankara, Turkey
- 7 Department of Hematology, Memorial Ankara Hospital, Ankara, Turkey
- 8 Department of Hematology, Bayındır Hospital, Ankara, Turkey
- 9 Department of Hematology, Faculty of Medicine, Erciyes University, Kayseri, Turkey
*Corresponding author: Prof. Dr. Yusuf BARAN, PhD., Izmir Institute of Technology, Faculty of Science, Department of Molecular Biology and Genetics, Urla, Izmir, Turkey. Tel.: +90 232 7506001; +90 506 1241010. E-mail: ybaran@gmail.com
Received 1 April 2020 Revised 18 May 2020 Accepted 25 May 2020 Published 1 June 2020
DOI: http://dx.doi.org/10.14312/2397-8694.2020-1
Copyright: © 2020 Yandim MK, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Objectives: Tyrosine kinase inhibitor (TKI) resistance is one of the major obstacles in chronic myeloid leukemia (CML) treatment. Ceramide, the central molecule of the sphingolipid metabolism, is synthesized by ceramide synthases (CERS1-6). While ceramide is known to be a pro-apoptotic molecule, glucosylceramide and sphingosine 1-phosphate converted from the ceramide are anti-apoptotic. In this study, we aimed to determine the potential roles of bioactive sphingolipids in terms of predicting drug resistance and prognosis in chronic myeloid leukemia patient samples. Materials and methods: Expression levels of CERS1-6, GCS, SK1, and BCR/ABL genes of 66 CML patients that are newly diagnosed, TKI-resistant or TKI-sensitive were analyzed by qRT-PCR. Results: CERS1-6 genes were expressed higher in the patients treated with TKIs than that of the patients newly diagnosed and TKI-resistant. However, expression levels of anti-apoptotic GCS and SK1 genes were significantly higher in TKI-resistant and blastic phase patients than that of other patients. Additionally, BCR/ABL expression levels were higher in newly diagnosed and TKI-resistant patients. Conclusion: Our results suggest that expression levels of bioactive sphingolipid genes might be novel markers for determination of drug resistance in CML patients. More importantly, they might be used as novel targets for more effective treatment of resistant CML patients.
Keywords: chronic myeloid leukemia; drug resistance; tyrosine kinase inhibitor; bioactive sphingolipids; ceramide
IntroductionTop
Chronic myeloid leukemia (CML) is characterized by Philadelphia (Ph) chromosome that codes for BCR/ABL oncoprotein, having perpetual tyrosine kinase activity [1]. Accordingly, tyrosine kinase inhibitors (TKIs), such as imatinib, nilotinib, and dasatinib are used as frontline therapy in CML [2]. Nonetheless, many patients can develop resistance through several mechanisms [3, 4].
Bioactive sphingolipid family is a member of membrane lipids. They have many important regulatory and structural roles in lipid bilayer, and are important in several cellular events, including proliferation, senescence, apoptosis, inflammatory responses, and migration [5]. Ceramide is the key member of sphingolipid family. Increased levels of ceramide cause growth inhibition, induction of apoptosis and differentiation, alteration of telomerase activity and telomere length, and also senescence. However, increased levels of other important sphingolipids, such as sphingosine 1-phosphate (S1P) and glucosylceramide (GC), converted from the ceramide by the activity of the enzymes, sphingosine kinase-1 (SK1) and glucosylceramide synthase (GCS), respectively, contribute to transformation, induction of cell proliferation, angiogenesis, mobility, and also development of multidrug resistance in several cancer cells [6]. Ceramide is synthesized de novo by CERS genes (CERS1-6) in mammals. Each of these six genes contributes to the generation of different lengths of ceramide [7].
Our previous studies confirmed the roles of bioactive sphingolipids in TKI-resistance and –sensitivity of CML cell lines [8, 9]. In order to verify our previous results, in this study, we aimed to determine the potential roles of bioactive sphingolipids in CML patient samples, rather than in cell lines. Expression levels of bioactive sphingolipid genes and the BCR/ABL oncogene were determined and compared in newly diagnosed; TKI-treated; and TKI-resistant CML patients’ bone marrow samples. Additionally, the relation between expression levels of bioactive sphingolipid genes and disease prognosis of CML patients was also determined.
Materials and methodsTop
Chemicals
Total RNA isolation kit was obtained from Macherey-Nagel (Germany). Reverse transcriptase (Moroney Murine Leukemia Virus Reverse Transcriptase) and primers were obtained from Fermentas (USA). DyNAmo HS SYBR Green qPCR Kit was obtained from Thermo Fisher Scientific (USA). NH4Cl and KHCO3 included in lysis buffer were obtained from Sigma (USA).
Ethical approval
Approval for the study was given by Ege University, Clinical Research Ethics Committee due to the use of bone marrow samples from chronic myeloid leukemia patients. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (Ege University, Clinical Research Ethics Committee) and with the 1964 Helsinki declaration, and its later amendments or comparable ethical standards.
Informed consent statement
All participants’ rights were protected and written informed consents were obtained before the procedures, according to the Helsinki Declaration.
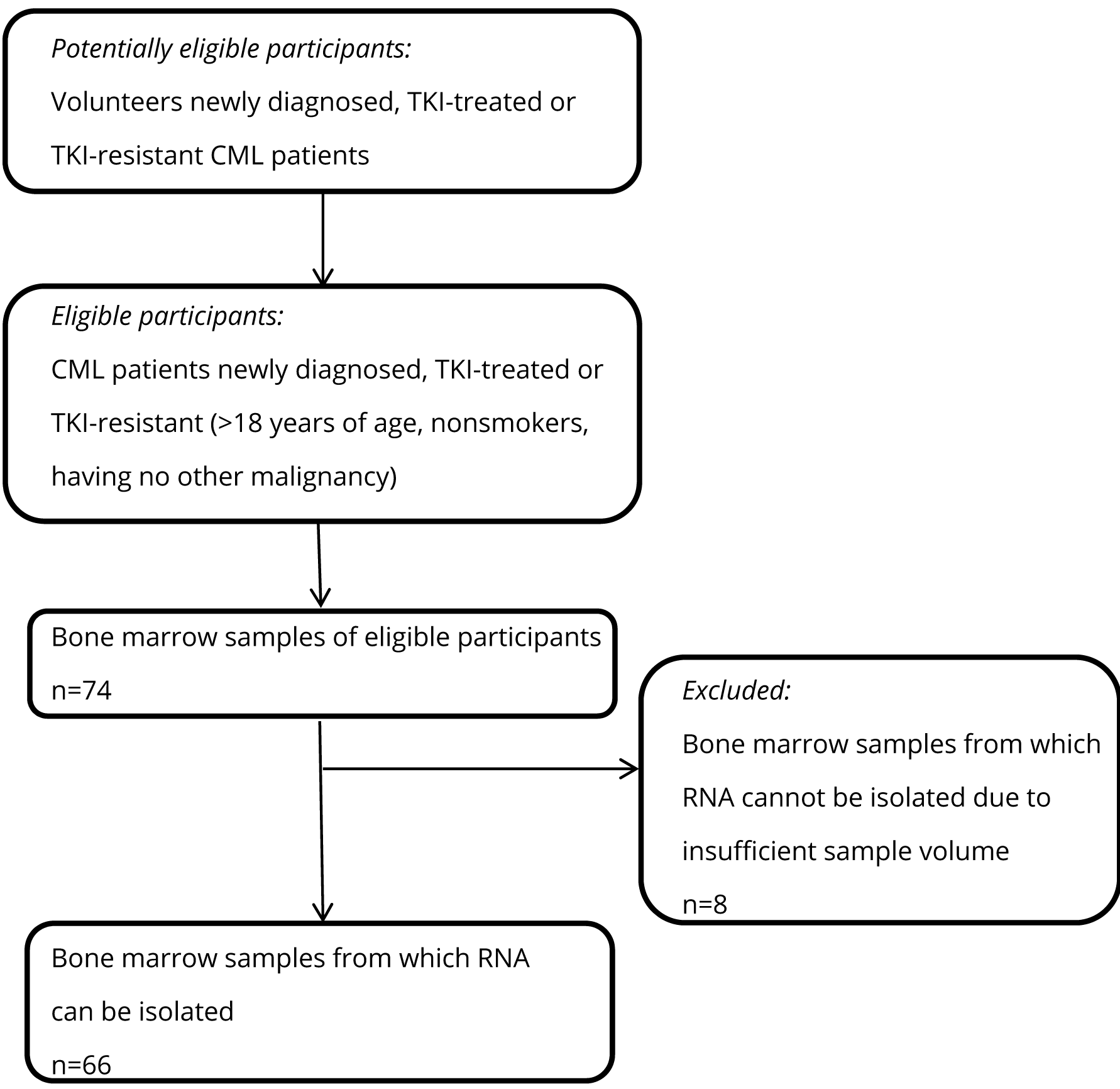
Patient samples
Bone marrow samples were acquired from 66 CML patients at Ege University Hospital, 9 Eylul University Hospital, Baskent University Hospital, Bayındır Hospital, Gulhane Military Medical School, and Erciyes University Hospital. 33 of the patients were newly diagnosed; 19 were TKI (imatinib, nilotinib, and dasatinib)-treated and showed hematological response; 11 were TKI-resistant; and 3 were at blastic phase of the disease. TKI-resistance was assessed according to minimum hematological response. If the patient did not show hematological response to the TKI therapy, the patient was accepted as TKI-resistant.
All patients, 44 males and 22 females, were informed about the study and signed the consent form. The median age of the patients was 52 (Table 1). A description of the patient samples used in this study is provided in Figure 1.
| Patient characteristics | Number of patients |
| Median age (years) | 52 |
| Females | 22 |
| Males | 44 |
| Newly diagnosed | 33 |
| Treated with IMA | 14 |
| Treated with NIL | 2 |
| Treated with DAS | 3 |
| IMA-resistant | 6 |
| NIL-resistant | 1 |
| DAS-resistant | 1 |
| NIL- & DAS-resistant | 1 |
| IMA- & NIL-resistant | 2 |
| Blastic phase | 3 |
| Total # of patients | 66 |
Isolation of bone marrow mononuclear cells
Mononuclear cells from the patient bone marrow samples were isolated by lysis buffer including 155 mM NH4Cl and 10 mM KHCO3. 10 ml lysis buffer was added onto bone marrow sample in a sterile falcon tube, and shaken by orbital shaker for 10 min. Then, bone marrow sample in the falcon tube was centrifuged at 8000 g for 5 min, and supernatant was removed. The pellet was homogenized with 2 ml lysis buffer, centrifuged again at 8000 g for 5 min, and then this step was repeated. Afterwards, the pellet was washed by 2 ml PBS, and centrifuged at 8000 g for 5 min. After the centrifugation, supernatant was removed, and the pellet was homogenized, and isolated bone marrow mononuclear cells were counted by hemocytometer.
Total RNA isolation and cDNA synthesis
Total RNAs of patient bone marrow mononuclear cells were extracted using a Ribolock RNA isolation kit (Macherey-Nagel, Germany) as described by the manufacturer. 1µg of each total RNA was reverse transcribed using reverse transcriptase (RevertAid Reverse Transcriptase, Thermo Fisher Scientific, USA). After 10 min of incubation at 25°C, and 60 min at 42°C, the reactions were stopped at 70°C for 10 min.
Quantitative real time polymerase chain reaction (QRT-PCR)
cDNAs were used in quantitative RT-PCR to measure the mRNA expression levels of CERS1-6, SK1, GCS, BCR/ABL and β-actin. The expression levels of β-actin were used as internal positive control. All of the experiments were conducted as triplicate in two independent sets. The primer sequences used in this study are listed in the table (Table 2). 2µl of the 1:5 diluted reverse transcriptase reaction product was amplified using these primers by qRT-PCR for 40 cycles, and their expression levels were analyzed by assuming the Ct level of β-actin as reference according to the 2-dCt calculation method.
| Gene | Primer sequence |
| β-actin | F: 5’-CAGAGCAAGAGAGGCATCCT-3’ |
| R: 5’-TTGAAGGTCTCAAACATGAT-3’ | |
| CERS1 | F: 5’-CACTGCGCGCCTCTTTCG-3’ |
| R: 5’-ATTGTGGTACCGGAAGGCG-3’ | |
| CERS2 | F: 5’- GCTGGAGATTCACATTTTAC-3’ |
| R: 5’-GAAGACGATGAAGATGTTGT-3’ | |
| CERS3 | F: 5’CATGATCTTGCAGGTCCTTCACC-3’ |
| R: 5’-CTCGTCATCACTCCTCACATCC-3’ | |
| CERS4 | F: 5’-GACCTTCTCCTACAGTGCCAAC-3’ |
| R: 5’-GTCGCACACTTGCTGATACTGC-3’ | |
| CERS5 | F: 5’-ATCTTCTTCGTGAGGCTG-3’ |
| R: 5’- ATGTCCCAGAACCAAGGT-3’ | |
| CERS6 | F: 5’- ATCAGGAGAAGCCAAGCACG-3’ |
| R: 5’- AGTAGTGAAGGTCAGTTGTG-3’ | |
| GCS | F: 5’- TCCAGATACGCTTACTGACATGG-3’ |
| R: 5’-TTGAAACCAGTTACATTGGCAGA-3’ | |
| SK1 | F: 5’- TCCTGGCACTGCTGCACTC-3’ |
| R: 5’-TAACCATCAATTCCCCATCCAC -3’ |
Statistical analysis
Statistical significance was determined using one-way analysis of variance for quantitative RT-PCR. P<0.05 was considered to be significant.
ResultsTop
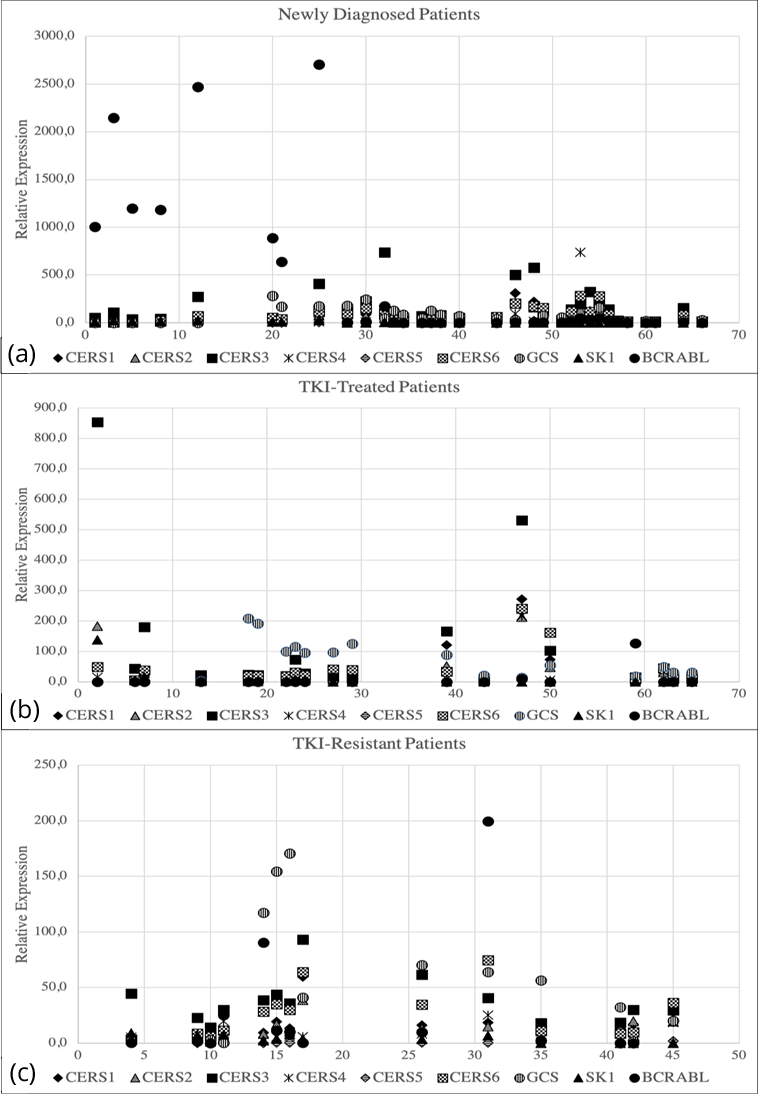
Expression levels of bioactive sphingolipid (CERS1-6, GCS, SK1) and BCR/ABL genes in patient samples
Real-time PCR results were analysed according to the expression levels of beta actin gene of each patient as reference. Cumulatively, expression levels of CERS1-6 genes were significantly higher in some of the newly diagnosed patients; and in the patients TKI-treated and showed minimum hematological response. On the other hand, expression levels of CERS1-6 genes were found significantly lower in the patients treated with TKIs and showed loss in molecular response; TKI-resistant patients; and in the patients at blastic phase, which is the most aggressive phase of the disease (Figure 2). Our data demonstrated that GCS and SK1 genes were highly expressed in some of the newly diagnosed CML patients; TKI- resistant patients; and in the patients at blastic phase. However, expression levels of GCS and SK1 genes were lower in the patients treated with TKIs and showed minimum hematological response; and in some of the newly diagnosed patients (Figure 2). Additionally, our results showed that BCR/ABL gene was highly expressed in some of the newly diagnosed CML patients. On the other hand, BCR/ABL gene expression levels were found lower in some of the patients that showed resistance to nilotinib or imatinib; and in the patients that showed hematological remission (Figure 2).
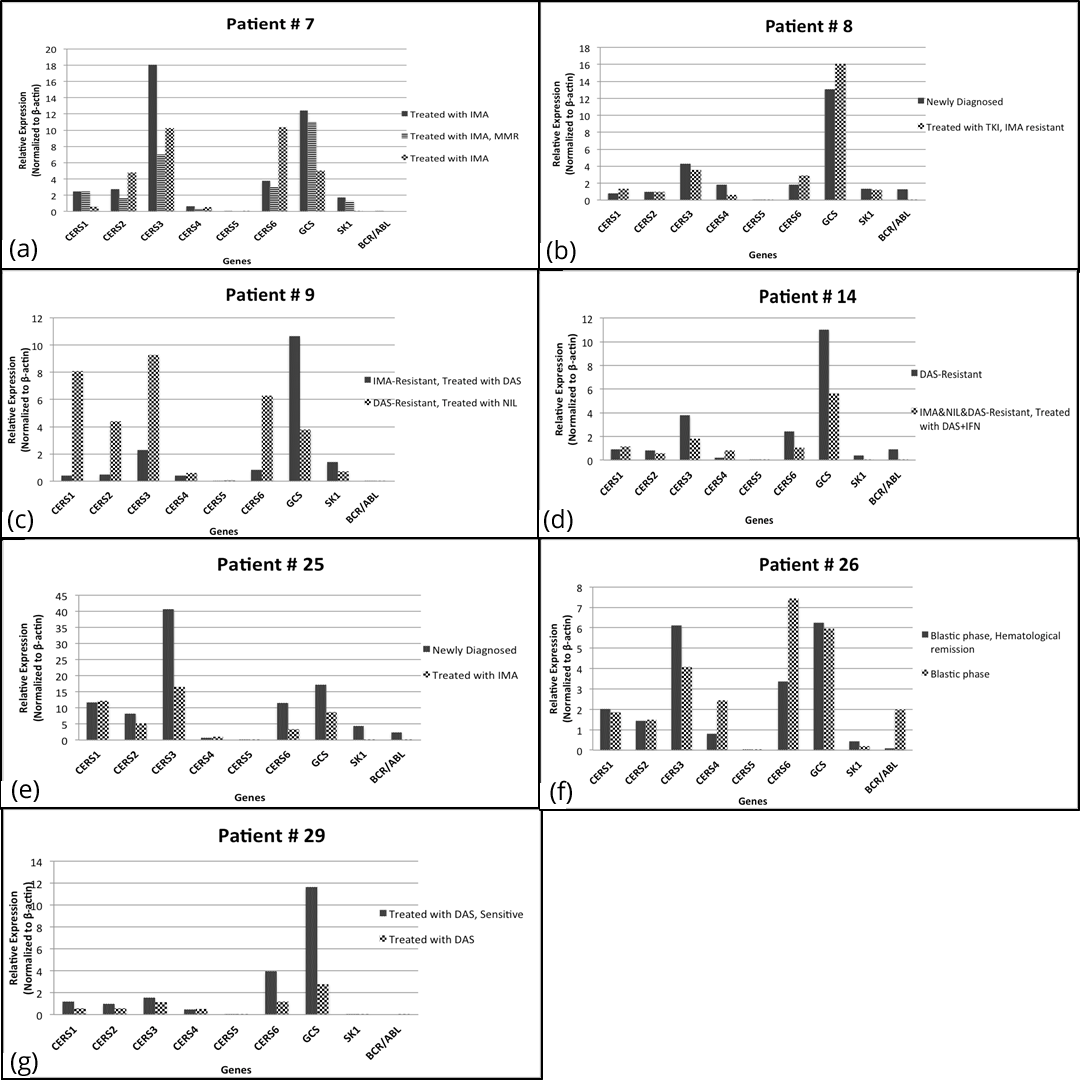
Determination of the relation between the expression levels of bioactive sphingolipid genes and disease prognosis
In order to determine the relation between the expression levels of bioactive sphingolipid genes and disease prognosis, we acquired bone marrow samples from 7 patients (#7, #8, #9, #14, #25, #26, and #29) on multiple occasions, and compared the bioactive sphingolipid expression levels in each sample of the patients during their treatment.
Bone marrow samples of patient #7 were acquired three times when the patient was treated with imatinib (sample 1), when treated with imatinib and showed major molecular response (MMR) (sample 2), and again when treated with imatinib (sample 3). Our results showed that expression levels of CERS2, -5, and -6 increased, but GCS, SK1, and BCR/ABL expression levels decreased during imatinib treatment of patient #7 (Figure 3a). Additionally, we acquired bone marrow samples from patient #8 twice when the patient was newly diagnosed (sample 1), and when showed resistance to imatinib but still treated with imatinib (sample 2). In the patient #8, CERS1, -2, -5, and -6 expression levels increased during imatinib treatment (Figure 3b). When the patient #8 gained resistance to imatinib, GCS expression was found to be increased, whereas SK1 expression was almost unchanged (Figure 3b). Samples of patient #9 were acquired twice when the patient was imatinib-resistant and treated with dasatinib (sample 1), and also when dasatinib-resistant and treated with nilotinib (sample 2) (Figure 3c). Our results showed that although the patient #9 was resistant to imatinib and dasatinib, expression levels of CERS1, -2, -3, -5, and -6 genes significantly increased while GCS, SK1, and BCR/ABL expression levels decreased during nilotinib treatment (Figure 3c). Furthermore, we also acquired bone marrow samples of patient #14 twice when the patient was dasatinib-resistant (sample 1), and when imatinib-, nilotinib-, and dasatinib-resistant but treated with dasatinib in combination with interferon (sample 2). The expression levels of CERS4 increased in this patient during the treatment with dasatinib and interferon combination, while GCS, SK1, and BCR/ABL expression levels decreased (Figure 3d). Moreover, samples of patient #25 were acquired twice when the patient was newly diagnosed (sample 1), and when treated with imatinib (sample 2). When we compared the expression levels with those of the first sample, our results showed that in patient #25, the expression levels of CERS1 and -4 slightly increased, whereas expression levels of the other bioactive sphingolipid genes and BCR/ABL decreased during imatinib treatment (Figure 3e). In patient #26 at blastic phase, expression levels of CERS1, -2, and -5, and also GCS were almost unchanged whereas BCR/ABL expression significantly increased over time (Figure 3f). We acquired bone marrow samples from patient #29 twice when the patient was treated with dasatinib in different time periods. Our results showed that CERS4 expression slightly increased over time during treatment. However, the expression levels of CERS1, -2, -3, -5 and -6 genes decreased to less than half during dasatinib treatment. Expression levels of GCS and SK1 genes also decreased significantly in the same patient (Figure 3g).
DiscussionTop
Our previous studies have indicated that there is a balance between ceramide and S1P in CML cell lines, but if the balance is interrupted and equilibrium is changed to the S1P direction, CML cells start to develop resistance against imatinib [10]. Furthermore, inhibition of GCS and SK1 by PDMP and SK1 inhibitor, respectively, results in ceramide accumulation, and this, in turn, sensitizes the cells to the treatment, and triggers apoptosis in drug-resistant and –sensitive CML cell lines [9, 10]. Additionally, combinations of PDMP, SK1 inhibitor, or ceramide with anticancer agents show synergistic cytotoxic and apoptotic effects in CML cell lines [8, 9, 11, 12]. Moreover, BCR/ABL stability is maintained by SK1 and S1P Receptor-2 by changing protein phosphatase 2A activity [13].
There are some clinical studies about the potential effects of bioactive sphingolipids in patients with several types of cancer, such as breast, head and neck, and non-small cell lung cancers. In breast cancer patients, it has been reported that C2 and C6 ceramides do not cause high levels of toxicity, and are well tolerated [14]. In another study, comparisons of normal tissues with tumor tissues of 45 patients with head and neck squamous cell carcinoma have been observed to have higher levels of C16:ceramide and C24:ceramide, but significantly lower levels of C18:ceramide in tumor tissues. Additionally, lower C18:ceramide levels have been reported to be related to invasive and metastatic phenotype in these tumor tissues [15]. Ceramide has been also used in cancer immunotherapy. In phase I trials in head and neck cancer, it has been reported that α-galactosylceramide-triggered antigen-presenting cells increase peripheral blood levels of natural killer cells. Natural killer function of these cells also increases significantly. Additionally, it has been reported there is no adverse effect of this treatment [16]. The same cancer immunotherapy approach has been also reported in non-small cell lung cancer. In phase I and II studies with 17 non-small cell lung cancer patients, PBMCs treated with α-galactosylceramide were co-cultured with IL-2 and GM-CSF, and then these cells were intravenously injected. The results showed that 10 out of 17 patients showed higher peripheral blood levels of IFN-gamma producing cells; additionally, their median survival time was longer compared to the other seven patients. In all patients, α-galactosylceramide was tolerated well, without observed adverse effects [17]. In another phase I clinical study, 43 cancer patients with solid tumors were treated with safingol (SK1 inhibitor) alone, and also in combination with cisplatin. The results showed that 37 patients responded to the therapy, 6 patients showed stability in disease, and reversible liver toxicity was observed in a dose-dependent manner. In addition, plasma levels of S1P decreased in response to safingol treatment in a dose-dependent manner [18]. In another study, it has been observed that ceramide levels are significantly lower in CD34 progenitor cells of 30 blast phase CML patients, as compared to healthy counterparts. In addition, these CD34 cells are more resistant to TKI treatment in vitro [19].
In this study, we analyzed and compared the expression levels of bioactive sphingolipid metabolism genes (CERS1-6, GCS, SK1) and BCR/ABL gene in bone marrow samples from 66 CML patients with different disease profiles. In order to assess the effects of the expression levels of bioactive sphingolipid genes in the clinical progress of CML, we acquired bone marrow samples from 7 patients more than once in different time periods. In the light of the results of this study, we concluded that CERS1, -2, -3, -5, and -6 expressions might be important in the achievement of positive responses to TKI treatment. Decreases in expression levels of these genes might lead to the development of resistance against TKIs in CML patients. However, CERS4 gene expression might not be related to the positive responses to TKI treatment in CML. In most CML patients resistant to TKIs, or at the blastic phase, CERS4 was expressed at higher levels. Moreover, GCS might be related to the development of resistance against TKIs. In our results, nilotinib and dasatinib were more effective than imatinib in decreasing GCS expression in CML patients. Furthermore, in most cases, there was an inverse relation between gene expression levels of CERS1 and GCS, SK1, and also BCR/ABL. We also observed that SK1 expression increased especially in imatinib-resistant CML patients. Additionally, dasatinib was more effective in decreasing the expression levels of SK1. On the other hand, although some of the patients expressing high levels of SK1 also expressed higher levels of BCR/ABL, our results showed no strong correlation between SK1 and BCR/ABL expression in CML patients. This correlation might be seen as comparing protein levels. More importantly, acquiring bone marrow samples from CML patients on different conditions in different time periods showed that it might be possible to estimate the disease progression through considering the expression levels of bioactive sphingolipid genes. In the future, protein levels of these bioactive sphingolipids beside of mRNA levels are needed to be compared in more patient samples.
ConclusionTop
These results confirm our previous conclusions that increased intracellular levels of CERS genes trigger TKI-induced cell death, and also GCS and SK1 contribute to the development of drug resistance in CML cell lines. In this study, we showed that expression levels of bioactive sphingolipids might play an important role in the achievement of positive responses, or development of drug resistance to TKI therapy in CML patients. Therefore, expression levels of bioactive sphingolipid genes might be predictive for estimating disease progression. More importantly, targeting bioactive sphingolipid genes, for instance, overexpression of CERS genes or downregulation of GCS and SK1, might be more effective in CML therapy. For this reason, bioactive sphingolipids might be novel targets, as well as prognostic markers for greater effectiveness in CML therapy.
Acknowledgments
We would like to acknowledge the Scientific and Technological Research Council of Turkey (TUBITAK) for financial support for this study with the project number 111S392. Additionally, we would like to thank Biotechnology and Bioengineering Center of Izmir Institute of Technology staff for their help and technical support.
Funding
This work was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) (grant number: 111S392; YB).
Declaration of interest
The authors declare that they have no conflict of interest.
ReferencesTop
[1]Cea M, Cagnetta A, Nencioni A, Gobbi M, Patrone F. New insights into biology of chronic myeloid leukemia: implications in therapy. Curr Cancer Drug Targets. 2013; 13(7):711–723.Article Pubmed
[2]O'Brien S, Radich JP, Abboud CN, Akhtari M, Altman JK, et al. Chronic myelogenous leukemia, Version 1.2014. J Natl Compr Canc Netw. 2013; 11(11):1327–1340.Article Pubmed
[3]Milojkovic D, Apperley J. Mechanisms of resistance to imatinib and second-generation tyrosine inhibitors in chronic myeloid leukemia. Clin Cancer Res. 2009; 15(24):7519–7527.Article Pubmed
[4]Roychowdhury S, Talpaz M. Managing resistance in chronic myeloid leukemia. Blood Rev. 2011; 25(6):279–290.Article Pubmed
[5]Kimura S, Ando T, Kojima K. Ever-advancing chronic myeloid leukemia treatment. Int J Clin Oncol. 2014; 19(1):3–9.Article Pubmed
[6]Yandim MK, Apohan E, Baran Y. Therapeutic potential of targeting ceramide/glucosylceramide pathway in cancer. Cancer Chemother Pharmacol. 2013; 71(1):13–20.Article Pubmed
[7]Kartal-Yandim M, Adan-Gokbulut A, Baran Y. Molecular mechanisms of drug resistance and its reversal in cancer. Crit Rev Biotechnol. 2016; 36(4):716–726.Article Pubmed
[8]Teufel A, Maass T, Galle PR, Malik N. The longevity assurance homologue of yeast lag1 (Lass) gene family (review). Int J Mol Med. 2009; 23(2):135–140.Article Pubmed
[9]Gencer EB, Ural AU, Avcu F, Baran Y. A novel mechanism of dasatinib-induced apoptosis in chronic myeloid leukemia; ceramide synthase and ceramide clearance genes. Ann Hematol. 2011; 90(11):1265–1275.Article Pubmed
[10]Camgoz A, Gencer EB, Ural AU, Baran Y. Mechanisms responsible for nilotinib resistance in human chronic myeloid leukemia cells and reversal of resistance. Leuk Lymphoma. 2013; 54(6):1279–1287.Article Pubmed
[11]Baran Y, Salas A, Senkal CE, Gunduz U, Bielawski J, et al. Alterations of ceramide/sphingosine 1-phosphate rheostat involved in the regulation of resistance to imatinib-induced apoptosis in K562 human chronic myeloid leukemia cells. J Biol Chem. 2007; 282(15):10922–10934.Article Pubmed
[12]Kartal M, Saydam G, Sahin F, Baran Y. Resveratrol triggers apoptosis through regulating ceramide metabolizing genes in human K562 chronic myeloid leukemia cells. Nutr Cancer. 2011; 63(4):637–644.Article Pubmed
[13]Can G, Cakir Z, Kartal M, Gunduz U, Baran Y. Apoptotic effects of resveratrol, a grape polyphenol, on imatinib-sensitive and resistant K562 chronic myeloid leukemia cells. Anticancer Res. 2012; 32(7):2673–2678.Article Pubmed
[14]Salas A, Ponnusamy S, Senkal CE, Meyers-Needham M, Selvam SP, et al. Sphingosine kinase-1 and sphingosine 1-phosphate receptor 2 mediate Bcr-Abl1 stability and drug resistance by modulation of protein phosphatase 2A. Blood. 2011; 117(22):5941–5952.Article Pubmed
[15]Jatoi A, Suman VJ, Schaefer P, Block M, Loprinzi C, et al. A phase II study of topical ceramides for cutaneous breast cancer. Breast Cancer Res Treat. 2003; 80(1):99–104.Article Pubmed
[16]Karahatay S, Thomas K, Koybasi S, Senkal CE, Elojeimy S, et al. Clinical relevance of ceramide metabolism in the pathogenesis of human head and neck squamous cell carcinoma (HNSCC): attenuation of C(18)-ceramide in HNSCC tumors correlates with lymphovascular invasion and nodal metastasis. Cancer Lett. 2007; 256(1):101–111.Article Pubmed
[17]Uchida T, Horiguchi S, Tanaka Y, Yamamoto H, Kunii N, et al. Phase I study of alpha-galactosylceramide-pulsed antigen presenting cells administration to the nasal submucosa in unresectable or recurrent head and neck cancer. Cancer Immunol Immunother. 2008; 57(3):337–345.Article Pubmed
[18]Motohashi S, Nagato K, Kunii N, Yamamoto H, Yamasaki K, et al. A phase I-II study of alpha-galactosylceramide-pulsed IL-2/GM-CSF-cultured peripheral blood mononuclear cells in patients with advanced and recurrent non-small cell lung cancer. J Immunol. 2009; 182(4):2492–2501.Article Pubmed
[19]Dickson MA, Carvajal RD, Merrill AH, Gonen M, Cane LM, et al. A phase I clinical trial of safingol in combination with cisplatin in advanced solid tumors. Clin Cancer Res. 2011; 17(8):2484–2492.Article Pubmed


