Journal of Diabetology and Endocrinology
An International Peer-Reviewed Open Access Journal
ISSN 2398-0281


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Diabetology and Endocrinology
Volume 1, Issue 1, March 2016, Pages 1–4
Case reportOpen Access
Disuse osteoporosis: A better understanding of pathophysiology may lead to potential therapies
- 1 Fertility Associates Auckland, Level 3, Ascot Central, 7 Ellerslie Racecourse Drive, Remuera, Auckland 1051, New Zealand
- 2 Department of Reproductive Endocrinology, Womens’ Health Division, Level 5, Building 4, Greenlane Clinical Centre, Greenlane, Auckland, New Zealand
- 3 Department of Obstetrics and Gynaecology, University of Auckland, 85 Park Road, Grafton Auckland, New Zealand
- 4 Faculty of Medical and Health Sciences, University of Auckland, 85 Park Road, Grafton Auckland, New Zealand
- 5 Department of Medicine, University of Auckland, 85 Park Road, Grafton Auckland
- 6 Department of Endocrinology, Auckland Hospital and Greenlane Clinical Centre, Private Bag 92189, Auckland Mail Centre, Auckland 1142, New Zealand
*Corresponding author: Dr. Stella Milsom, Fertility Associates Private Bag 28910, Remuera, Auckland 1541, New Zealand. Tel.: +6495209520; Fax: +6495209521; E-mail: s.milsom@auckland.ac.nz
Received 9 December 2015 Revised 4 February 2016 Accepted 15 February 2016 Published 28 February 2016
DOI: http://dx.doi.org/10.14312/2398-0281.2016-1
Copyright: © 2016 Milsom S, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Amputation results in mechanical unloading of a previously weight bearing limb with subsequent focal decrease in bone mineral density (BMD), and the development of disuse osteoporosis. We report the case of a 44-year-old woman with a right above knee amputation sustained three decades ago, who was subsequently diagnosed with severe osteoporosis in the right proximal femur. We discuss the relative failure of established antiresorptive therapies to improve BMD above the site of the amputation, and the possibility that the underlying pathophysiology of disuse osteoporosis is different from osteoporosis associated with aging and menopause. A review of current literature suggests that an alteration in the secretion of sclerostin and parathyroid hormone (PTH) during mechanical unloading may be partially responsible for the development of disuse osteoporosis. Specific anabolic agents such as monoclonal antibody to sclerostin or recombinant PTH may be more effective than antiresorptive therapies to improve BMD and reduce fracture risk in patients with disuse osteoporosis.
Keywords: mechanical loading; disuse osteoporosis; sclerostin; parathyroid hormone
IntroductionTop
Osteoporosis is a common and well-recognized condition characterized by low bone mass and an increased risk of fragility fractures. Bisphosphonates are widely accepted as the first line therapy for the management of osteoporosis across the world. They have been consistently shown to significantly reduce osteoporotic fracture risk associated with aging and menopause [1, 2]. However, these potent therapies appear to be relatively ineffective in the setting of osteoporosis related to the disuse of a peripheral limb [3]. We report the case of a 44-year-old Caucasian women with localized osteoporosis in the setting of an above knee amputation, and discuss the literature on potential therapies for management of osteoporosis in this setting.
Case reportTop
A 44-year-old premenopausal Caucasian woman was referred to the Endocrinology clinic in the year 2000 for assessment of her bone mineral density. This was on a background of a right sided above knee amputation to the level of the mid femoral shaft secondary to severe trauma after a motorbike accident at age 15. Her hip anatomy was unaffected by the accident, as noted on serial imaging. She wore an above knee prosthetic leg for up to 10 hours each day, and used axillary crutches at other times. She had no other significant past medical history and there was no family history of osteoporosis. We obtained informed consent from the patient for publication of the article.
Bone density examination (DXA Lunar DPX-IQ) revealed severe osteoporosis in the right proximal femur (Table 1). Screening tests for secondary causes of osteoporosis other than disuse, including celiac disease, calcium, phosphate, 25-hydroxyvitamin D and parathyroid hormone levels were normal. Therapy was commenced with etidronate (400mg daily for 14 days followed by Calcium Carbonate 500mg daily for 76 days in a repeating cycle) and monthly cholecalciferol 1.25mg. She tolerated the oral etidronate well and her compliance was excellent. The more effective bisphosphonate, alendronate, (70 mg weekly) was substituted for etidronate when national funding for alendronate became available in New Zealand in 2002.
| 2001 | 2002 | 2004 | 2008 | 2011 | |
| L1-L4 | 1.07 | 1.07 | 1.08 | 1.08 | NA |
| Lt neck | 0.83 | 0.85 | 0.9 | NA | 0.87 |
| Rt neck | 0.45 | 0.45 | 0.43 | NA | 0.23 |
| Lt total | 0.92 | 0.95 | 0.96 | 0.96 | 0.96 |
| Rt total | 0.41 | 0.39 | 0.36 | 0.26 | NA |
| Treatment | Etidronate | Alendronate | Alendronate | Zolendronate MHT |
MHT |
Abbreviations: Lt = left hip; Rt = right hip; NA = not available; MHT = menopausal hormone replacement therapy.
Unfortunately, in 2004 (aged 48 years), she fell on to her living room floor while attempting a transfer without crutches or her artificial limb and sustained a comminuted right intertrochanteric hip fracture, managed by closed reduction and a minimally invasive plate and screw fixation (Figure 1). Post-operative recovery was uncomplicated.
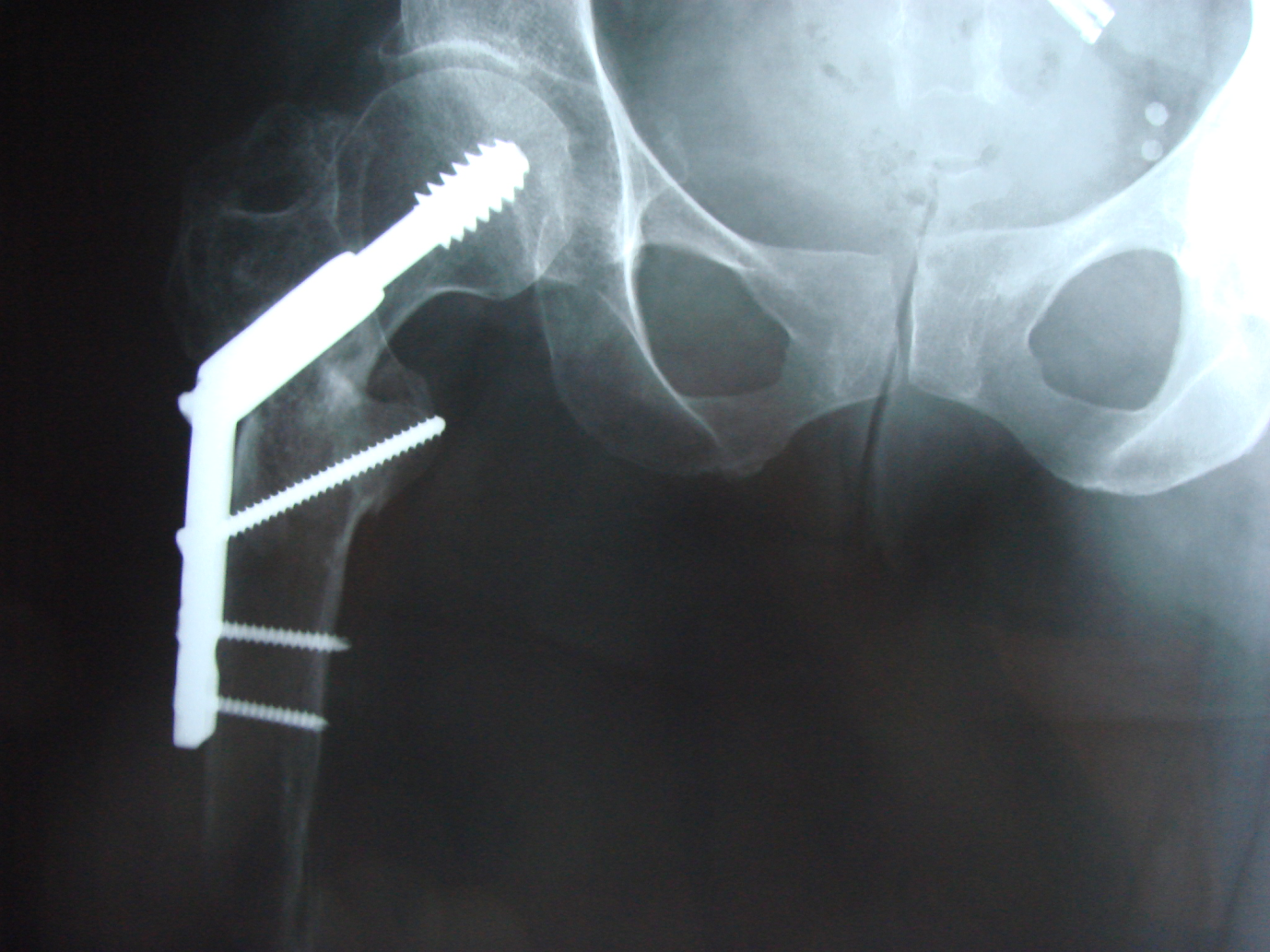
Further decision making around management concentrated on the apparent relative lack of response to an effective bisphosphonate in the presence of a low impact serious fracture in a relatively young patient. In addition, the presence of metal artifact in the right hip area has rendered BMD measurements in this site difficult to interpret.
Alendronate, temporarily discontinued during the period of fracture healing, was resumed in 2005, together with calcium and Vitamin D supplementation. Additionally, menopausal hormone replacement therapy (MHT) (1 mg beta estradiol daily and a Mirena intrauterine progesterone secreting device) was prescribed primarily for severe menopausal symptoms, but with the possibility that MHT could be beneficial for her osteoporosis. Parenteral bisphosphonate (IV Zolendronate 5mg) was administered in 2008. The patient has subsequently declined further IV or oral bisphosphonate therapy but continued menopausal hormone therapy. She has returned to previous mobility, with instructions to use crutches or her artificial limb at all times and there have been no further fractures to date.
DiscussionTop
Osteoporosis is a systemic skeletal disease associated with a significant risk of fragility fractures. Osteoporosis is usually recognised in the setting of ageing, menopause, and low body mass index, and in association with a number of secondary conditions. However, osteoporosis with subsequent increase in fracture risk in bone affected by amputation of a weight bearing limb has attracted little attention until recently. Our patient demonstrates the severe osteoporosis that may be associated with the amputation of a weight bearing limb and relative ineffectiveness of established antiresorptive therapies such oral and parenteral bisphosphonate therapy, and hormone replacement therapy. BMD in sites other than the right hip area has improved with therapy, whereas there has been no response in BMD in the right hip, and even an apparent deterioration post fracture, despite the expected artefactual increase in BMD that is normally seen with metal in this site. She has additionally sustained a low impact hip fracture subsequent to therapy being initiated. This woman’s apparent unresponsiveness to conventional osteoporosis therapy raises the possibility that osteoporosis associated with an amputation is associated with a different underlying pathophysiology from osteoporosis related to aging and menopause.
Amputation leads to mechanical unloading of a previously weight bearing limb. Mechanical unloading is also observed in pathological immobilisations associated with neurological or muscular diseases (such as stroke and spinal cord injury), space travel or prolonged bed rest. It has been well documented that absence of skeletal mechanical loading leads to a decrease in bone mass, called disuse osteoporosis [4]. A clinical and radiological survey of bone and joint changes in 42 amputees found that osteoporosis in the amputated limb is common, being noted in 88% of amputees [5]. It has also been shown that the total and cortical bone mineral density is significantly lower at the end of the residual limb compared with the intact limb for both above- and below-knee amputees [6]. Furthermore, a longitudinal study of hemiplegic patients with stroke found that even a two month delay in return to ambulation is associated with a significant loss of bone mineral content, especially on the paretic side [7].
Osteoporosis associated with an amputation and other models of disuse appears to be a local phenomenon [8], occurring around sites of mechanical unloading. Under conditions of normal mechanical loading, osteocytes produce multiple signaling molecules, which stimulate bone formation [9]. The canonical Wnt pathway, known to have an important role in bone formation, appears to play a critical role with data from both in vitro and in vivo studies demonstrating an up-regulation of the Wnt pathway after mechanical loading [10]. Sclerostin, a gene product of the SOST gene is a signaling molecule expressed by osteocytes, which is a negative regulator of canonical Wnt signaling. The canonical Wnt-induced responses in mesenchymal cells are critically dependent on LRP5 and LRP6 receptor signaling and sclerostin has been shown to interact with these receptors to inhibit the Wnt pathway [11-13]. The exact molecular mechanism, by which sclerostin exerts its antagonistic effects on Wnt signaling in bone forming osteoblasts, remains unclear. Mechanical loading in rodents is associated with decreased sclerostin levels [14] whereas the mechanical unloading of wild-type mice up-regulates sclerostin and decreases Wnt/β-catenin signalling [11]. In addition, SOST knock-out mice have been found to be resistant to bone loss induced by unloading [11]. Human studies in postmenopausal women immobilized after a single episode of stroke have shown high sclerostin levels, with a negative correlation with bone formation markers [15]. This data supports the hypothesis that, osteocytes increase sclerostin secretion in response to mechanical unloading which in turn inhibits the canonical Wnt pathway, resulting in decreased osteoblast activity, and bone formation.
Established therapies for management of osteoporosis including bisphosphonates have not been efficacious in our patient. However, given the putative inhibitory role of sclerostin in mechanical unloading, therapy directed at lowering sclerostin levels is clinically relevant. Anti-sclerostin antibody administration in a murine model of mechanical unloading completely prevented bone loss and moreover, increased bone formation [16, 17]. In a Phase 2 clinical trial of osteopenic postmenopausal women, monoclonal anti-sclerostin antibody therapy achieved greater increases in BMD than either teriparatide or alendronate [18]. There is no data (to our knowledge) of monoclonal anti-sclerostin antibody therapy in humans with disuse osteoporosis but should be considered for trial when clinically available.
Intermittent use of parathyroid hormone (PTH 1-34) is anabolic on bone and is another potential therapy for our patient. PTH levels have been investigated in black bears who maintain bone mineral content despite up to six months of disuse while hibernating. PTH and osteocalcin concentrations are higher post-hibernation relative to pre-hibernation raising the possibility that anabolic actions of PTH may be important in maintenance of bone mineral content during hibernation [19]. Constitutive activation of PTH/PTHrP receptor signalling in osteoblasts, an action likely to mimic PTH administration, suppresses unloading-induced bone loss in Col1a1-caPPR transgenic mice [20]. The anabolic effect of PTH on bone may in part relate to PTH mediated down-regulation of sclerostin expression by osteocytes [21]. In humans, one small study (n=7) has demonstrated that while PTH levels decreased in healthy men exposed to 90 days of bed rest, serum sclerostin levels increased [22]. Taken together, current evidence indicates that PTH may be involved in bone remodeling associated with disuse, and may in part mediate this effect via change in sclerostin levels.
ConclusionTop
Disuse osteoporosis occurs due to unbalanced bone remodeling, with a decrease in bone formation. Current research strongly supports the likelihood that alterations in the levels of sclerostin and possibly parathyroid hormone are involved in the pathogenesis of bone loss related to mechanical unloading. This raises the possibility that anabolic therapies such as monoclonal antibodies to sclerostin or recombinant PTH may be potential therapeutic options for our patient. In the interim, as prolonged bisphosphonate use appears to have been relatively ineffective and may increase our patient’s risk of atypical femoral fracture, which would seriously impair her current independence and mobility, we have not recommended further bisphosphonate treatment.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
Declaration of interest
There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
ReferencesTop
[1]Liberman UA, Weiss SR, Bröll J, Minne HW, Quan H, et al. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. The Alendronate phase III osteoporosis treatment study group. N Engl J Med. 1995; 333(22):1437–1443.Article Pubmed
[2]Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007; 356(18):1809–1822.Article Pubmed
[3]Li CY, Price C, Delisser K, Nasser P, Laudier D, et al. Long-term disuse osteoporosis seems less sensitive to bisphosphonate treatment than other osteoporosis. J Bone Miner Res. 2005; 20(1):117–124.Article Pubmed
[4]Alexandre C, Vico L. Pathophysiology of bone loss in disuse osteoporosis. Joint Bone Spine. 2011; 78(6):572–576.Article Pubmed
[5]Burke MJ, Roman V, Wright V. Bone and joint changes in lower limb amputees. Ann Rheum Dis. 1978; 37(3):252–254.Article Pubmed
[6]Sherk VD, Bemben MG, Bemben DA. BMD and bone geometry in transtibial and transfemoral amputees. J Bone Miner Res. 2008; 23(9):1449–1457.Article Pubmed
[7]Jørgensen L, Jacobsen BK. Changes in muscle mass, fat mass, and bone mineral content in the legs after stroke: a 1 year prospective study. Bone. 2001; 28(6):655–659.Article Pubmed
[8]Kulkarni J, Adams J, Thomas E, Silman A. Association between amputation, arthritis and osteopenia in British male war veterans with major lower limb amputations. Clin Rehabil. 1998; 12(4):348–353.Article Pubmed
[9]Klein-Nulend J, Bacabac RJ, Bakker AD. Mechanical loading and how it affects bone cells: the role of the osteocyte cytoskeleton in maintaining our skeleton. Eur Cell Mater. 2012; 24:278–291.Article Pubmed
[10]Robinson JA, Chatterjee-Kishore M, Yaworsky PJ, Cullen DM, Zhao W, et al. Wnt/beta-catenin signaling is a normal physiological response to mechanical loading in bone. J Biol Chem. 2006; 281(42):31720–31728.Article Pubmed
[11]Lin C, Jiang X, Dai Z, Guo X, Weng T, et al. Sclerostin mediates bone response to mechanical unloading through antagonizing Wnt/beta-catenin signaling. J Bone Miner Res. 2009; 24(10):1651–1661.Article Pubmed
[12]Li X, Zhang Y, Kang H, Liu W, Liu P, et al. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J Biol Chem. 2005; 280(20):19883–19887.Article Pubmed
[13]Semenov M, Tamai K, He X. SOST is a ligand for LRP5/LRP6 and a Wnt signaling inhibitor. J Biol Chem. 2005; 280(29):26770–26775.Article Pubmed
[14]Robling AG, Niziolek PJ, Baldridge LA, Condon KW, Allen MR, et al. Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/Sclerostin. J Biol Chem. 2008; 283(9):5866–5875.Article Pubmed
[15]Gaudio A, Pennisi P, Bratengeier C, Torrisi V, Lindner B, et al. Increased sclerostin serum levels associated with bone formation and resorption markers in patients with immobilization-induced bone loss. J Clin Endocrinol Metab. 2010; 95(5):2248–2253.Article Pubmed
[16]Li X, Ominsky MS, Warmington KS, Morony S, Gong J, et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J Bone Miner Res. 2009; 24(4):578–588.Article Pubmed
[17]Tian X, Jee WS, Li X, Paszty C, Ke HZ. Sclerostin antibody increases bone mass by stimulating bone formation and inhibiting bone resorption in a hindlimb-immobilization rat model. Bone. 2011; 48(2):197–201.Article Pubmed
[18]McClung MR, Grauer A, Boonen S, Bolognese MA, Brown JP, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014; 370(5):412–420.Article Pubmed
[19]Donahue SW, Galley SA, Vaughan MR, Patterson-Buckendahl P, Demers LM, et al. Parathyroid hormone may maintain bone formation in hibernating black bears (Ursus americanus) to prevent disuse osteoporosis. J Exp Biol. 2006; 209(Pt 9):1630–1638.Article Pubmed
[20]Ono N, Nakashima K, Schipani E, Hayata T, Ezura Y, et al. Constitutively Active Parathyroid Hormone Receptor Signaling in Cells in Osteoblastic Lineage Suppresses Mechanical Unloading-induced Bone Resorption. J Biol Chem. 2007; 282(35):25509–25516.Article Pubmed
[21]Silvestrini G, Ballanti P, Leopizzi M, Sebastiani M, Berni S, et al. Effects of intermittent parathyroid hormone (PTH) administration on SOST mRNA and protein in rat bone. J Mol Histol. 2007; 38(4):261–269.Article Pubmed
[22]Spatz JM, Fields EE, Yu EW, Divieti Pajevic P, Bouxsein ML, et al. Serum Sclerostin increases in healthy adult men during bed rest. J Clin Endocrinol Metab. 2012; 97(9):1736–1740.Article Pubmed



