Journal of Radiology and Imaging
An International Peer-Reviewed Open Access Journal
ISSN 2399-8172


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Radiology and Imaging
Volume 1, Issue 5, November 2016, Pages 33–38
Original researchOpen Access
MRI system scoring for differentiation of malignant versus benign stricture of the common bile duct
- 1 Department of Radiology, Ospedale San Bortolo, v.le F. Rodolfi, 36100 Vicenza, Italy
- 2 Department of Gastroenterology, Ospedale San Bortolo, v.le F. Rodolfi, 36100 Vicenza, Italy
- 3 US Army Health Clinic, v.le della Pace, Vicenza, Italy
*Corresponding author: Sergio Savastano, MD, Department of Radiology, Ospedale San Bortolo, v.le F. Rodolfi 37, 36100 Vicenza, Italy. Tel.: +39 0444 75 3490; Fax: +39 0444 75 3839; E-mail: sergio.savastano@ulssvicenza.it
Received 20 July 2016 Revised 25 September 2016 Accepted 10 October 2016 Published 18 October 2016
DOI: http://dx.doi.org/10.14312/2399-8172.2016-8
Copyright: © 2016 Savastano S, et al. Published by NobleResearch Publishers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
The purpose of this study was to evaluate the ability of magnetic resonance imaging (MRI) to differentiate between malignant and benign strictures of the common bile duct by using a scoring system. MRI scans of 34 patients with indeterminate biliary stricture on prior imaging were reviewed; non-enhanced MRI, magnetic resonance cholangiopancreatography (MRCP), diffusion weighted imaging, and contrast-enhanced MRIs were considered for analysis. Ten radiologic findings were significant for malignancy: luminal irregularity, post-contrast conspicuity, high signal intensity on diffusion weighted MRI, luminal stenosis asymmetry, T2-weighted MRI signal intensity, indistinct outer border, abrupt stenosis, bile duct dilatation ≥ 12.5 mm, stenosis length ≥ 10.5 mm, and wall thickness ≥ 4.5 mm. Since none of these findings was pathognomonic for malignancy, a simple system scoring based on statistically significant findings was developed, where each of the above findings counts for one point. A score of 6 or higher was found only in patients with a malignant stricture of the common bile duct; 70% of patients with a benign stenosis had a score of 1, or 2, and all patients with benign lesions had scores of less than 5. This MRI scoring system can assist in the differential diagnosis of common bile duct stricture with high accuracy; to be widely applicable, the MRI score needs to be validated in a prospective patient population.
Keywords: MRI system scoring; malignant; benign stricture; bile duct
IntroductionTop
Despite rapid advances in diagnostic imaging modalities, noninvasive etiologic diagnosis of obstructive jaundice may be still challenging in some cases. Noninvasive imaging is traditionally required to recognize dilatation of the biliary tree, the level of obstruction, and, whenever possible, the cause of the obstruction. In this setting, magnetic resonance imaging (MRI) plays a pivotal role thanks to an unsurpassed precision in soft tissue contrast differentiation, multiplanar imaging capability, direct visualization of biliary and pancreatic fluids with magnetic resonance cholangiopancreatography (MRCP), identification of hypercellular lesions by means of diffusion weighted imaging (DWI), and hemodynamic analysis with contrast-enhanced MRI (CE-MRI) [1-5]. Moreover, MRI demonstrates high accuracy in local staging of patients with a malignant stenosis [1-3, 5].
The high sensitivity of MRCP in detecting strictures of the common bile duct [1] may not translate to a concomitant improvement in specificity and positive predictive value [6, 7]; therefore, a comprehensive MRI approach to the diagnosis of bile duct stenosis is necessary to minimize indeterminate cases, by pointing out MRI findings characteristics of malignancy [1, 2]. In particular, the etiology of small periductal lesions and of lesions of the common bile duct wall have been difficult to distinguish noninvasively, despite improvements in MRI technology [8, 9].
The purpose of this retrospective study is to evaluate the ability of MRI to differentiate between malignant and benign stenoses of the common bile duct by means of a practical scoring system based on objective imaging findings.
Materials and methodsTop
The present retrospective study was approved by our institutional ethics committee review board. The radiologic information system of our hospital was reviewed for all MRI investigations of the biliary system performed between January 2011 and December 2014. Patients with an indeterminate stricture of the common bile duct, i.e., not definitively diagnosed by abdominal sonography or computed tomography scans, were enrolled in the study. Patients with a prior history of biliary surgery and/or biliary radiologic or endoscopic procedures, common bile duct stones, acute pancreatitis, chronic pancreatitis, or overt neoplasms of the pancreatic head previously detected with ultrasound and/or computed tomography, were excluded from the study. MRI examinations were reviewed by a senior radiologist with 15 year experience in abdominal MRI, and unaware final diagnoses.
MR examinations of the upper abdomen were performed with a 1.5T magnet (Avanto, Siemens Healthcare, Erlangen, Germany) or a 3T magnet (Skyra, Siemens Healthcare, Erlangen, Germany) using phased-array body surface coils. MRI studies were displayed on a PACS workstation and the following sequences were reviewed: coronal HASTE T2-weighted imaging; in-phase and out-of phase GE T1 sequence; axial HASTE T2-weighted imaging without and with fat suppression; DWI; radial thick slice fat suppressed HASTE T2-weighted MRCP and paracoronal thin slice fat sat 3D T2-weighted MRCP; unenhanced and contrast-enhanced dynamic spoiled 3D GE sequence at 20 sec, 50 sec and 180 sec after intravenous injection of gadobenate dimeglumine (Multihance; Bracco, Milano, Italy). The paramagnetic agent (1 mg/kg body weight) was administered with a power injection through an 18-20 gauge catheter inserted in the antecubital fossa.
Categorical and ordinal variables scrutinized were as follows: (a) magnetic field strength; (b) evidence of the double duct sign; (c) luminal irregularity; (d) tapering versus abrupt narrowing of the stricture; (e) asymmetry of the stenosis; (f) the signal intensity (SI) on T2-weighted images relative to liver, expressed as low, iso-, or high; (g) the SI on DWI at b800, expressed as low or iso-high; (h) the stenosis length; (i) wall thickness of the stenosis; (j) the diameter of the common bile duct; (k) conspicuity of the lesion evaluated on equilibrium phase images of dynamic CE-MRI; (l) blurring of outer margins of the stenosis, estimated on portal phase of enhanced MRI.
SI on T2-weighted and DW images were subjectively evaluated since the very small size of lesions precluded a reliable measurement of the region of interest; for the same reason the apparent diffusion coefficient was not estimated.
Endoscopic ultrasonography (EUS) had been performed in all cases, with a dedicated system (Hitachi Avius, Japan) using longitudinal probes equipped with a 2.8 mm or 3.8 mm operator channel (Pentax EG-3870UTK e PentaxEG-3270UK slim); etiologic diagnostic examinations were completed, when technically feasible, by fine needle aspiration (FNA), accomplished with a 25G needle or fine needle biopsy (FNB) with a micro-histology needle (Cook Echo tip Ultra HD o Echo tip pro core 25G). All EUS procedures were performed by one of us (LC) in patients who had fasted for 24 h; patients were under deep sedation, in the left lateral decubitus position.
Final etiologic diagnosis was made on the basis of EUS-FNA/FNB, or on histology of surgical or postmortem specimens; whenever EUS-FNA/FNB was not technically feasible, or when negative for malignancy, MRI follow up studies were performed for at least six months to track the behavior of the lesion.
Statistical analysis was performed with χ2 and Fisher tests for comparing frequency of categorical variables in benign versus malignant cases. Wilcoxon-Mann-Whitney test was used for correlation of continuous variables, whose accuracy for malignancies was evaluated estimating the area under the characteristic receiver operator curve (ROC); D2 test was also calculated to identify cut-off values for malignancy. Univariate regression analysis, sensitivity and specificity values of all significant variables were finally calculated. A scoring system based on MRI findings significant for a malignancy was considered for both groups of patients. A p value ≤ 0.05 was considered statistically significant; statistical analysis was performed with SPSS® software package (IBM® SPSS® Statistics release 20.0, USA).
ResultsTop
During the period under consideration 1016 subjects underwent MRI studies of the biliary tree. Among these subjects, 34 patients (13 men, 21 women, mean age: 67 years, range: 19-85 year) met the inclusion criteria for the study; 12 of these patients (34%) patients had a prior history of cholecystectomy. The mean interval time between MRCP and EUS was 37 days (range: 1-60 days).
The final diagnoses included 20 cases of benign pathology and 14 cases of malignancy. Three cases of benign biliary stricture were characterized with cytology/histology (one each of cholangitis, mucosal hyperplasia of the papilla of Vater and primary sclerosing cholangitis); all these cases were followed up with MRI for a mean period of 201 days. The remaining 17 patients diagnosed with benign biliary stricture who did not undergo EUS-FNA/FNB had a mean follow up period of 270 days (range: 187-1053 days); the final diagnoses were: secondary cholangitis in 12 patients and Odditis in five patients.
The malignant biliary strictures included four cholangiocarcinomas of the common bile duct, four ductal carcinomas of the pancreatic head, three ampullary adenocarcinomas, one periampullary duodenal carcinoma, one neuroendocrine tumor of the pancreas, and one pancreatic intraductal papillary mucinous neoplasm. Diagnosis was confirmed by surgical pathology in four patients, EUS-FNA/FNB and surgical pathology in nine patients, and EUS-FNA and autopsy in one patient.
Categorical variables (Table 1) identified significantly more frequently in malignant strictures were the following: luminal irregularity, asymmetrical stenosis, abrupt stenosis, signal intensity on T2-weighted images, post-contrast conspicuity, and indistinct outer border. Ordinal variables (measured lengths in mm) all differed significantly between malignant and benign strictures (Table 2). The ROC of continuous variables and corresponding thresholds for malignant stenosis are reported in Table 3. The luminal irregularity and the high signal on DW-MRI showed the highest odd ratios at logistic regression analysis, as well as the highest sensitivity for diagnosis of malignant stricture (Tables 4 and 5).
| Benign stricture | % | Malignant stricture | % | p | |
| Magnetic field strength (1.5T vs 3T) | 3/17 | 15/85 | 4/10 | 28.6/71.4 | NS |
| Double duct sign | 5 | 25 | 7 | 50.0 | NS |
| Luminal irregularity | 3 | 15 | 13 | 92.9 | < 0.01 |
| Stenosis asymmetry | 5 | 25 | 12 | 85.7 | < 0.01 |
| Abrupt stenosis | 4 | 20 | 11 | 78.6 | < 0.01 |
| T2W SI | |||||
| Hypointense | 12 | 60 | 2 | 14.3 | 0.013 |
| Iso/hyperintense | 8 | 40 | 12 | 85.7 | |
| SI in DWI b800 (n = 24) | |||||
| Hypointense | 14 | 87.5 | 1 | 12.5 | < 0.01 |
| ±Hyperintense | 2 | 12.5 | 7 | 87.5 | |
| Enhancement (n = 27) | |||||
| Arterial phase (iso/hyperintense) | 9/5 | 64.2/35.8 | 7/6 | 53.8/46.2 | NS |
| Portal ph (iso/hyperintese) | 10/4 | 71.4/29.6 | 6/7 | 46.2/53.8 | NS |
| Equilibrium phase (iso/hyperintense) | 7/7 | 50.0/50.0 | 2/11 | 15.4/84.6 | NS |
| Post-contrast conspicuity (fair/good) (n = 27) | 11/3 | 78.6/21.4 | 1/12 | 7.7/92.3 | < 0.01 |
| Indistinct outer border (n = 27) | 2.0 | 7.4 | 11.0 | 84.6 | < 0.01 |
Abbreviations: SI: signal intensity; NS: not significant.
| MRI findings | Benign stricure | Malignant stricture | P |
| Bile duct dilatation (mm ± ds) | 11.5 ± 5.1 | 15.21 ± 5.32 | < 0.01 |
| Stenosis length (mm ± ds) | 8.1 ± 5.1 | 15.35 ± 7.02 | < 0.01 |
| Stenosis wall thickness (mm ± ds) | 3.6 ± 1.46 | 9.1 ± 6.5 | < 0.01 |
Abbreviations: sd: standard deviation.
| MRI findings | Area under ROC curve | se | Cut-off (mm) |
| Bile duct dilatation | 0.759 | ± 0.085 | 12.5 |
| Stenosis length | 0.814 | ± 0.082 | 10.5 |
| Stenosis wall thickness | 0.824 | ± 0.079 | 4.5 |
Abbreviations: se: standard error.
| MRI findings | Odds Ratio (95% CI) | p |
| Luminal irregularity | 73.6 (6.8-792.0) | < 0.01 |
| DW-MRI hyperintensity | 49.0 (3.8-637.0) | < 0.01 |
| Post-contrast conspicuity | 44.0 (4.0-488.0) | < 0.01 |
| Indistinct outer border | 33.0 (3.9-276.0) | < 0.01 |
| Stenosis asymmetry | 18.0 (2.9-109.0) | < 0.01 |
| Abrupt stenosis | 14.7 (2.7-78.9) | < 0.01 |
| Stenosis length ≥ 10.5mm | 12.4(7.5-20.6) | < 0.01 |
| T2 Iso/hyperintenisity | 9.0 (1.6-51.4) | 0.01 |
| Wall thickness ≥ 4.5mm | 7.1 (3.3-14.1) | < 0.01 |
| Bile duct dilatation ≥ 12.5mm | 6.8 (4.4-10.4) | < 0.01 |
Abbreviations: CI: confidence interval.
| MRI findings | Sensitivity | IC 95% | Specificity | IC 95% |
| Luminal irregularity | 93 | 66-99 | 85 | 62-97 |
| Post-contrast conspicuity | 92 | 64-99 | 78 | 49-95 |
| DW-MRI hyperintensity | 87 | 47-98 | 87 | 62-98 |
| Stenosis asymmetry | 86 | 57-98 | 75 | 51-91 |
| T2 Iso/hyperintenisity | 86 | 57-98 | 60 | 36-81 |
| Indistinct outer border | 85 | 54-98 | 86 | 57-98 |
| Abrupt stenosis | 78 | 49-95 | 80 | 56-94 |
| Bile duct dilatation ≥ 12.5mm | 71 | 42-91 | 75 | 51-91 |
| Stenosis length ≥ 10.5mm | 71 | 46-95 | 81 | 58-94 |
| Wall thickness ≥ 4.5mm | 61 | 32-86 | 71 | 42-91 |
Abbreviations: CI: confidence interval.
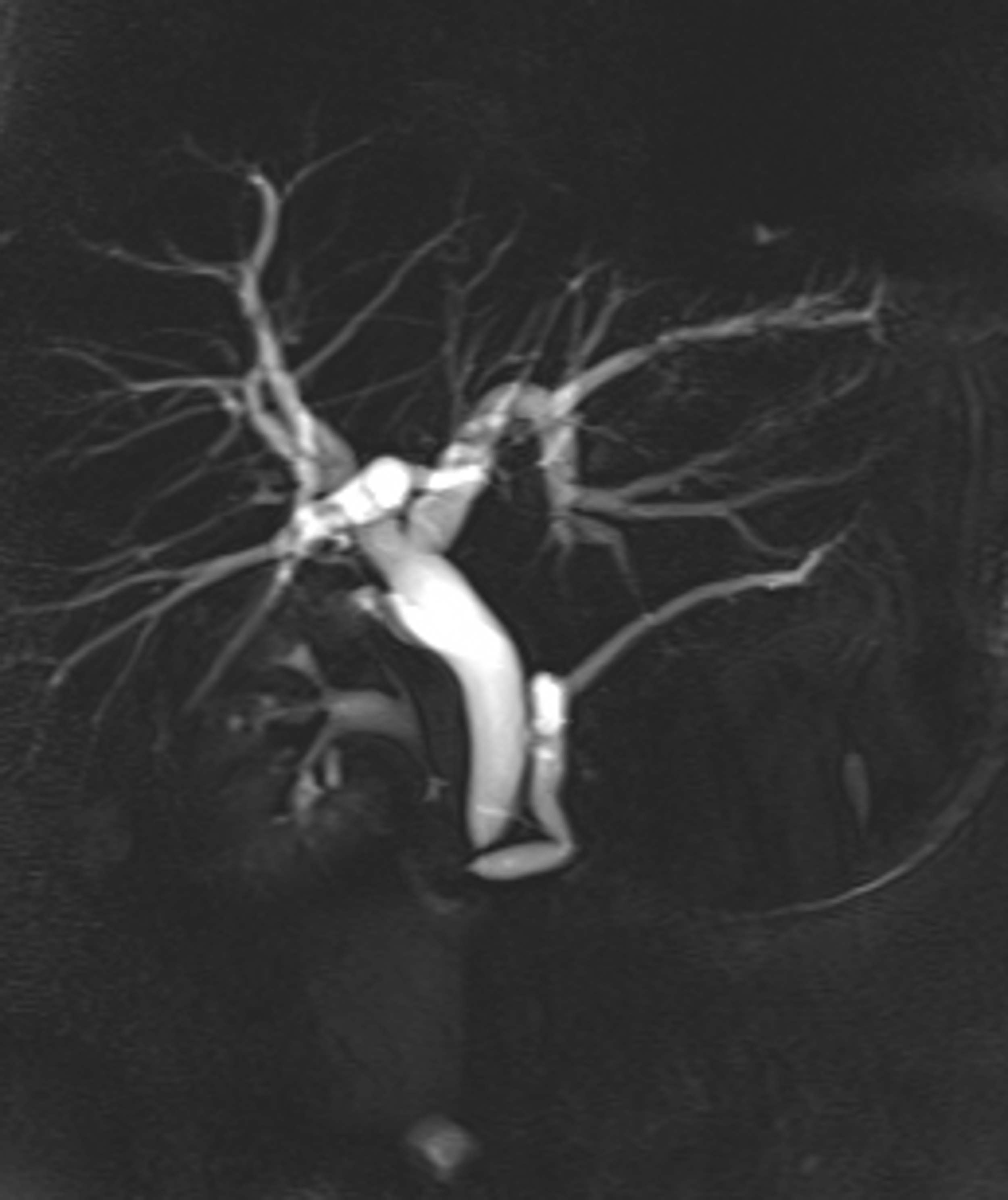
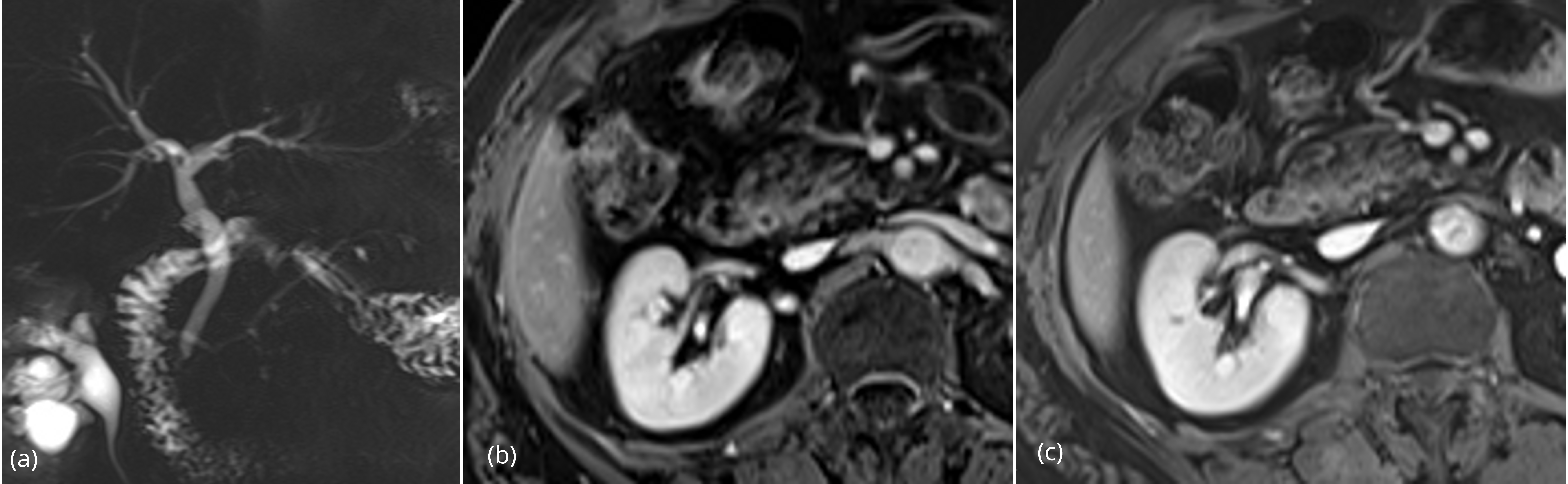
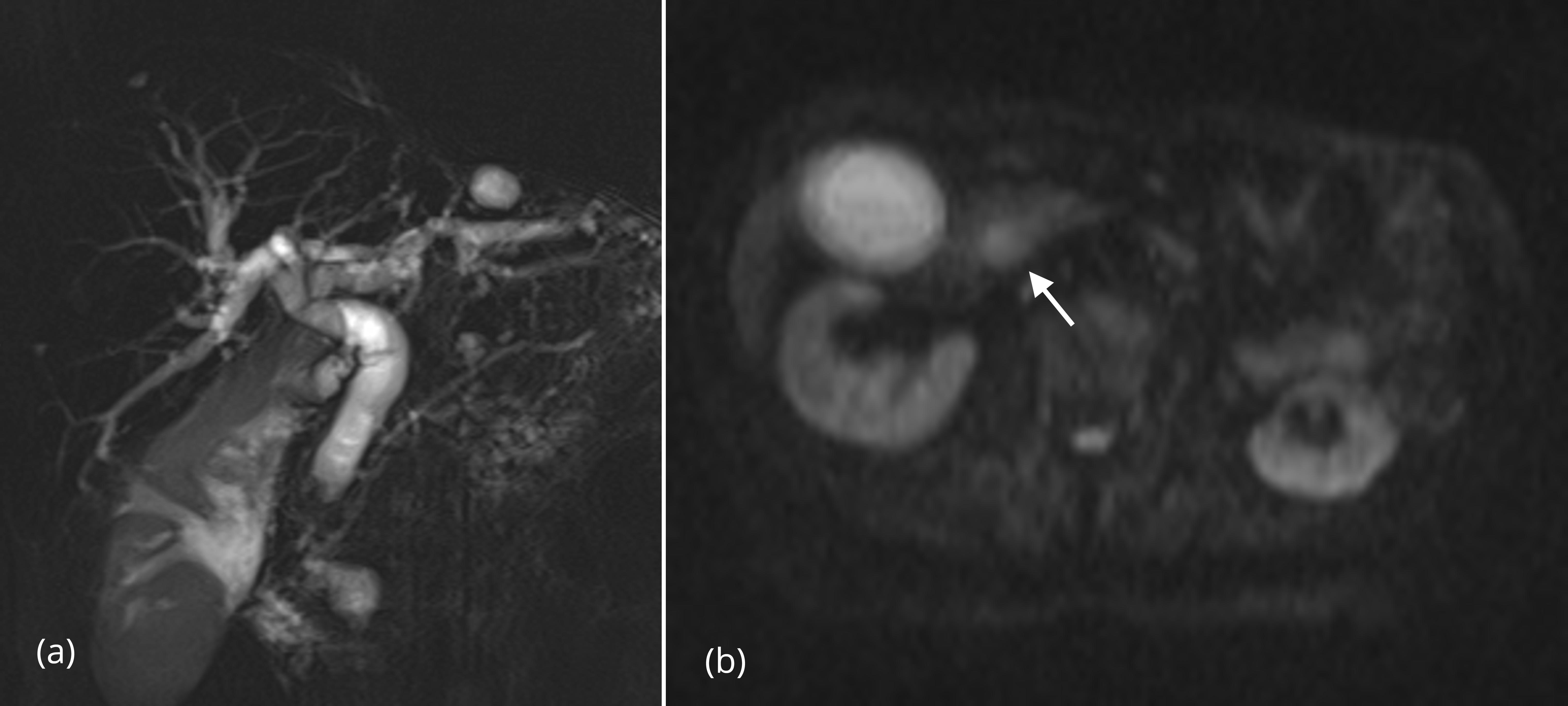
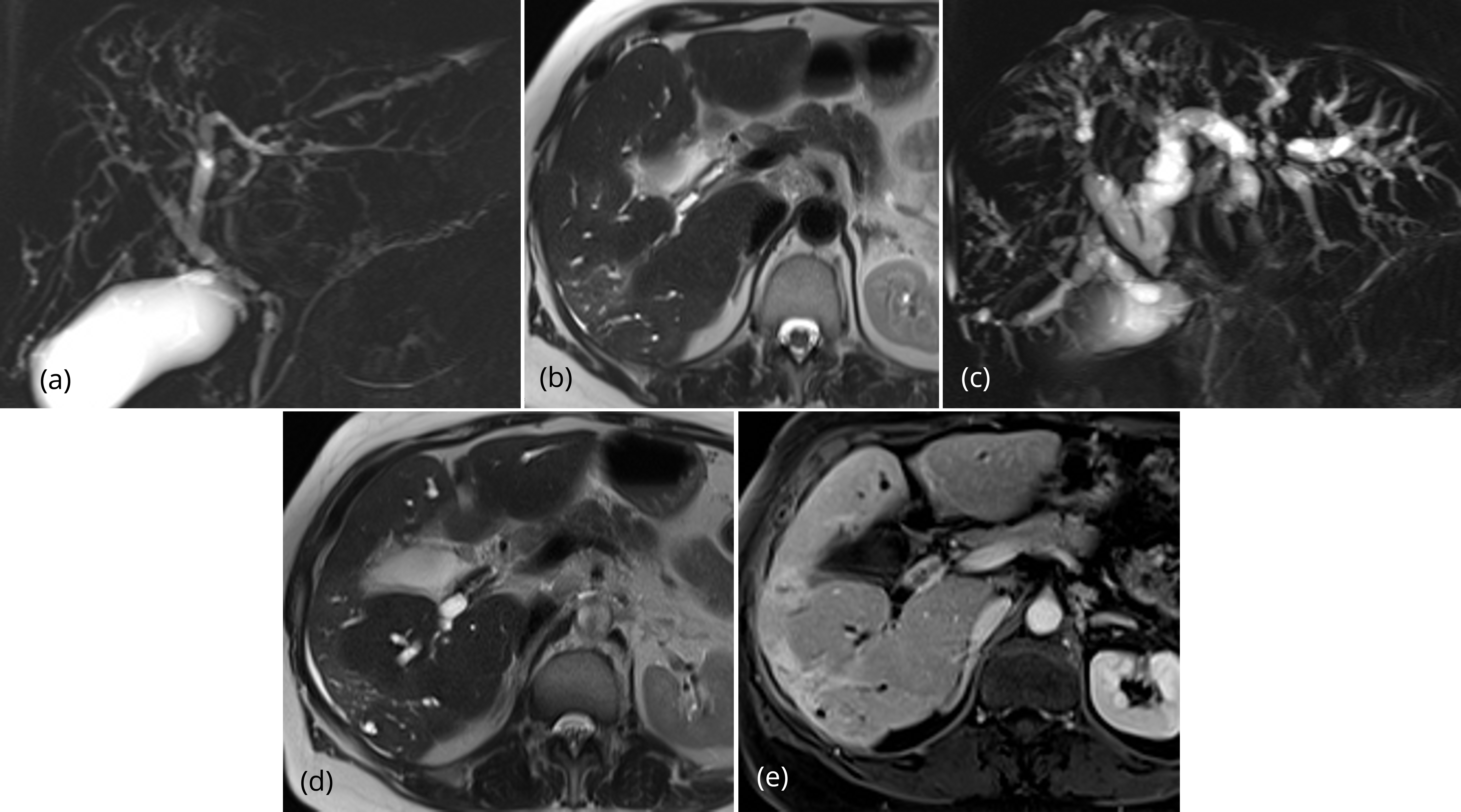
Summation of the ten significant findings permitted a simple scoring system that was highly discriminative for malignant versus benign strictures. In the 20 patients with benign lesions, all but one case totaled four or fewer points; 15/20 (75%) of patients with benign lesions had two or fewer points (Figures 1, 2). A single patient with a benign stricture had a total score of 5 points. Conversely, 12 of 14 patients with a histologically-proven malignant stricture had a score of 6 or higher; the other two patients had a score of 4 points or higher (Figures 3, 4). A score of 6 or greater was diagnostic of a malignant stricture (p < 0.01) (Figure 5) with a ROC area of 0.996; sensitivity of 85.7%, specificity of 100%, positive predictive value of 100% and negative predictive value of 90.9%.

DiscussionTop
MRI characterization of common bile duct stenosis relies on direct and secondary findings, none of which by themselves are pathognomonic for malignancy [1, 2, 5, 10, 11]. As previously reported [12], MRI diagnosis is not affected by the magnetic field strength.
MRCP provides noninvasive images comparable to those obtained by direct retrograde or transhepatic cholangiography, and can provide indirect evidence of a malignant lesion, such as irregularity of the inner border, abrupt stenosis, and stenosis length. Although these findings are highly sensitive, they are not specific for malignancy [1, 2, 4]. Dilatation of the biliary tree is an epiphenomenon of biliary strictures and depends more on the duration and degree rather than the cause of the stenosis, although such dilatation is more frequently observed in cases of malignancy [1, 4, 13]; therefore this sign has an ancillary value only, and should be always considered as a part of the complete radiologic picture.
Morphologic and signal characteristics of a stenosing biliary lesion may differentiate between benign and malignant pathology. Although high SI on T2-weighted images of the biliary lesion are correlated with malignancy, this finding has a weak predictive diagnostic value. Cholangiocarcinomas may be hyperintense relative to the liver because of increased interstitial fluid [2], but may also show a low signal due to a desmoid component that mimics a fibrotic stenosis; conversely, high signal intensity of a benign stricture may be observed when marked inflammation is present. In any event, evaluation of the SI should be cautiously assessed in most cases, due to the very small tumor size of these infiltrating lesions.
DW-MRI at high b values is a reliable predictor for a malignant bile stricture and can improve sensitivity of MRCP due to its sensitivity in appraising hypercellularity [5, 11, 14–16]; nevertheless care must be taken since inflammatory processes can also be hyperintense on DW-MRI [15, 16]. Moreover DW-MRI may not be discriminatory in cases of well differentiated neoplasms or for small tumors, and image quality may suffer from artifacts that induce signal loss [11].
CE-MRI can improve detection rate of cholangiocarcinomas, which usually hyperenhance on equilibrium phase images [2, 4, 17]. In the present study we did not find any statistical difference of enhancing pattern on CE-MRI between benign and malignant strictures; we believe this is due to heterogeneity of malignant tumors in our series, which included hypovascular tumors, such as cholangiocarcinomas and pancreatic ductal carcinomas, as well as hypervascular neoplasms, such as periampullary and neuroendocrine neoplasms. However, conspicuity of lesions on portal and/or equilibrium phases (hyperenhancing pattern for desmoid tumors and hypoenhancing for hypervascular neoplasms) significantly differs between the two groups in our series. The diagnostic value of CE-MRI is further highlighted by its ability to delineate thickness and outer borders of a bile duct wall or a periductal lesion, both of which are predictors for malignancy [2, 4].
Since these MRI findings are not individually pathognomonic for malignant bile duct stenosis, a constellation of MRI findings may be studied to improve diagnostic reliability. Kim et al. reported that a higher specificity may be reached by combining a large number of MRI findings (maximum six), unfortunately at the expense of a concomitant decrease in sensitivity; they achieved the best performance with a combination of three signs (sensitivity 100%, specificity 87.5%) [2]. In the present study, we created a 10-point score, assigning one point for each MRI finding statistically significant for malignancy, and found a score of six or more signs highly discriminatory for malignant lesions. Conversely, a score of one or two points was typical of benign strictures; in this setting MRI was extremely effective at ruling out a malignant neoplasm, and could allow for noninvasive imaging follow up over a period of time, without the need for biopsy in all cases of common bile duct stenosis. A score of 3 to 5 points falls in the gray zone of diagnostic uncertainty, and suggests that a tissue diagnosis should be obtained expeditiously in these patients.
The present study has some limitations. First, because our patient selection was retrospective, there may be inherent biases that have gone undetected; specifically, heterogeneity of the malignant group can alter evaluation of SI on T2-weighted MRI and DW-MRI, as well as evaluation of enhancing pattern on CE-MRI. Second, statistical analysis may have been affected by (i) the low sample size and (ii) non-uniformity of MRI investigations, since all 34 patients had not undergone all imaging sequences. Third, lack of histologic confirmation in all diagnosed benign lesions promotes uncertainty in the accuracy of the score; we feel this limitation has been mitigated by a sufficiently long follow up period. Finally, since our score is based on a retrospective chart review, to be valuable clinically, it must be validated in a prospective group of patients; we are now enrolling patients prospectively to test the score’s validity.
ConclusionTop
MRI investigation of common bile duct stricture requires an accurate technique relying on all available sequences to arrive at a definitive diagnosis. This comprehensive diagnostic approach can benefit from the application of our MRI score, which may narrow uncertainties in differentiating malignant versus benign causes of common bile duct stricture. Low scores rule out malignant lesions, and high scores are pathognomonic for malignant strictures; in the event of scores midway on our scale, we recommend tissue diagnosis to confirm the absence or presence of malignancy.
Conflicts of interest
The authors declare no conflicts of interest.
ReferencesTop
[1]Park MS, Kim TK, Kim KW, Park SW, Lee JK, et al. Differentiation of extrahepatic bile duct cholangiocarcinoma from benign stricture: Findings at MRCP versus ERCP. Radiology. 2004; 233(1):234–240.Article Pubmed
[2]Kim JY, Lee JM, Han JK, Kim SH, Lee JY, et al. Contrast-enhanced MRI combined with MR cholangiopancreatography for the evaluation of patients with biliary strictures: Differentiation of malignant from benign bile duct strictures. J Magn Reson Imaging. 2007; 26(2):304–312.Article Pubmed
[3]Yeh BM, Liu PS, Soto JA, Corvera C, Hussain HK. MR imaging and CT of the biliary tract. Radiographics. 2009; 29(6):1669–1688.Article Pubmed
[4]Yu XR, Huang WY, Zhang BY, Li HQ, Geng DY. Differentiation of infiltrative cholangiocarcinoma from benign common bile duct stricture using three-dimensional dynamic contrast-enhanced MRI with MRCP. Clin Radiol. 2014; 69(6):567–573.Article Pubmed
[5]Park HJ, Kim SH, Jang KM, Choi S, Lee SJ, et al. The role of diffusion-weighted MR imaging for differentiating benign from malignant bile duct strictures. Eur Radiol. 2014; 24(4):947–958.Article Pubmed
[6]Rösch T, Meining A, Frühmorgen S, Zillinger C, Schusdziarra V, et al. A prospective comparison of the diagnostic accuracy of ERCP, MRCP, CT, and EUS in biliary strictures. Gastrointest Endosc. 2002; 55(7):870–876.Article Pubmed
[7]Sai JK, Suyama M, Kubokawa Y, Watanabe S, Maehara T. Early detection of extrahepatic bile-duct carcinomas in the nonicteric stage by using MRCP followed by EUS. Gastrointest Endosc. 2009; 70(1):29–36.Article Pubmed
[8]Topazian M. Endoscopic ultrasonography in the evaluation of indeterminate biliary strictures. Clin Endosc. 2012; 45(3):328–330.Article Pubmed
[9]Victor DW, Sherman S, Karakan T, Khashab MA. Current endoscopic approach to indeterminate biliary strictures. World J Gastroenterol. 2012; 18(43):6197–6205.Article Pubmed
[10]Chung YE, Kim MJ, Kim HM, Park MS, Choi JY, et al. Differentiation of benign and malignant ampullary obstructions on MR imaging. Eur J Radiol. 2011; 80(2):198–203.Article Pubmed
[11]Cui XY, Chen HW, Cai S, Bao J, Tang QF, et al. Diffusion-weighted MR imaging for detection of extrahepatic cholangiocarcinoma. Eur J Radiol. 2012; 81(11):2961–2965.Article Pubmed
[12]Onishi H, Kim T, Hori M, Murakami T, Tatsumi M, et al. MR cholangiopancreatography at 3.0 T: Intraindividual comparative study with MR cholangiopancreatography at 1.5 T for clinical patients. Invest Radiol. 2009; 44(9):559–565.Article Pubmed
[13]Wu DS, Chen WX, Wang XD, Acharya R, Jiang XH. Pancreaticobiliary duct changes of periampullary carcinomas: quantitative analysis at MR imaging. Eur J Radiol. 2012; 81(9):2112–2117.
[14]Cui XY, Chen HW. Role of diffusion-weighted magnetic resonance imaging in the diagnosis of extrahepatic cholangiocarcinoma. World J Gastroenterol. 2010; 16(25):3196–3201.Article Pubmed
[15]Lee NK, Kim S, Kim GH, Kim DU, Seo HI, et al. Diffusion-weighted imaging of biliopancreatic disorders: Correlation with conventional magnetic resonance imaging. World J Gastroenterol. 2012; 18(31):4102–4117.Article Pubmed
[16]Lee NK, Kim S, Seo HI, Kim DU, Woo HY, et al. Diffusion-weighted MR imaging for the differentiation of malignant from benign strictures in the periampullary region. Eur Radiol. 2013; 23(5):1288–1296.Article Pubmed
[17]Li N, Liu C, Bi W, Lin X, Jiao H, et al. MRCP and 3D LAVA imaging of extrahepatic cholangiocarcinoma at 3 T MRI. Clin Radiol. 2012; 67(6):579–586.Article Pubmed



